Region:Global
Author(s):Dev
Product Code:KRAC0381
Pages:84
Published On:August 2025
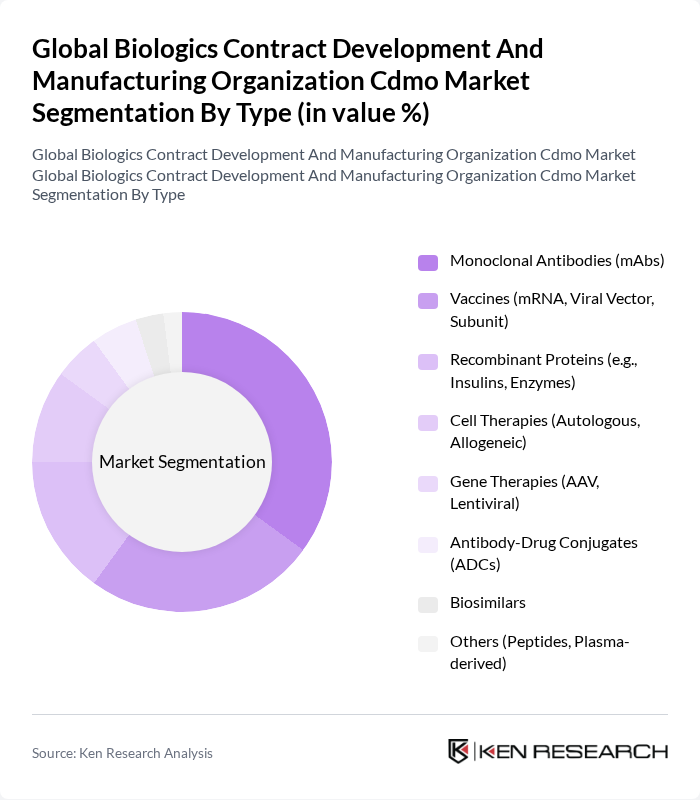
By Type:The market is segmented into various types, including Monoclonal Antibodies (mAbs), Vaccines (mRNA, Viral Vector, Subunit), Recombinant Proteins, Cell Therapies, Gene Therapies, Antibody-Drug Conjugates (ADCs), Biosimilars, and Others. Among these, Monoclonal Antibodies (mAbs) dominate the market due to their widespread application in treating various diseases, including cancer and autoimmune disorders. The increasing focus on personalized medicine and the growing number of mAb approvals contribute to their leading position in the market .
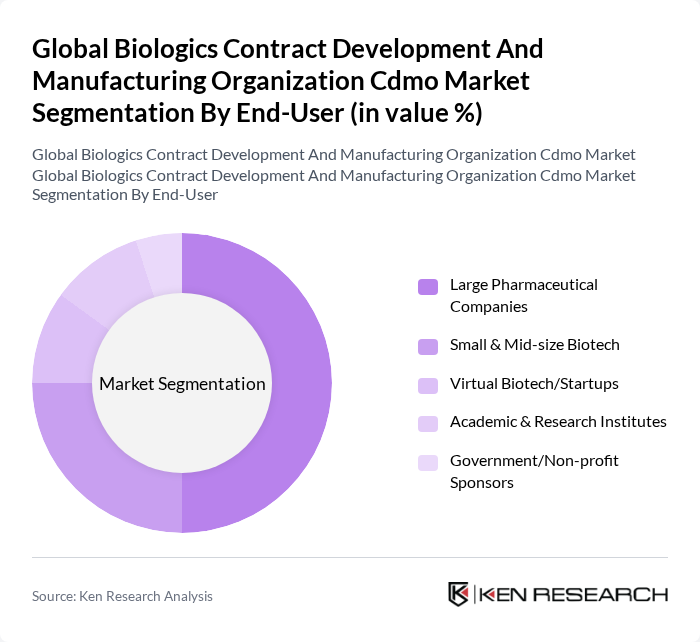
By End-User:The end-user segmentation includes Large Pharmaceutical Companies, Small & Mid-size Biotech, Virtual Biotech/Startups, Academic & Research Institutes, and Government/Non-profit Sponsors. Large Pharmaceutical Companies dominate this segment due to their extensive resources, established market presence, and ability to invest in advanced biomanufacturing technologies. Their need for reliable and scalable production capabilities drives the demand for CDMO services .
The Global Biologics Contract Development And Manufacturing Organization Cdmo Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group Ltd., Catalent, Inc., Samsung Biologics Co., Ltd., WuXi Biologics (Cayman) Inc., Boehringer Ingelheim BioXcellence, FUJIFILM Diosynth Biotechnologies, Thermo Fisher Scientific (Patheon Pharma Services), Recipharm AB, AGC Biologics, CordenPharma International, KBI Biopharma, Inc., Rentschler Biopharma SE, Abzena plc, Evonik Health Care (Biologics & Advanced Therapies), Charles River Laboratories (Viral Vector & CDMO Services) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biologics CDMO market appears promising, driven by the increasing demand for innovative therapies and the expansion of biopharmaceutical companies. As the industry embraces digital transformation, the integration of AI and machine learning in manufacturing processes is expected to enhance efficiency and reduce costs. Additionally, the focus on sustainability will likely lead to the adoption of greener manufacturing practices, positioning CDMOs as key players in the evolving healthcare landscape while addressing environmental concerns.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies (mAbs) Vaccines (mRNA, Viral Vector, Subunit) Recombinant Proteins (e.g., Insulins, Enzymes) Cell Therapies (Autologous, Allogeneic) Gene Therapies (AAV, Lentiviral) Antibody-Drug Conjugates (ADCs) Biosimilars Others (Peptides, Plasma-derived) |
| By End-User | Large Pharmaceutical Companies Small & Mid-size Biotech Virtual Biotech/Startups Academic & Research Institutes Government/Non-profit Sponsors |
| By Application | Therapeutics Diagnostics & Companion Diagnostics Research Use/Preclinical Clinical Supply (Phase I–III) |
| By Service Type | Process & Analytical Development cGMP Manufacturing (Clinical & Commercial) Fill-Finish & Packaging Quality Control/Release & Stability Testing Regulatory & CMC Services Tech Transfer & Scale-up Supply Chain & Cold Chain Logistics |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Bioprocess/Platform | Mammalian Cell Culture (e.g., CHO) Microbial Fermentation (E. coli, Yeast) Single-Use/Disposable Bioprocessing Continuous/Perfusion Processing Viral Vector Manufacturing (AAV, LVV) mRNA & LNP Manufacturing |
| By Pricing/Engagement Model | Fee-for-Service (FFS)/Time & Materials Full-Service/End-to-End (One-Stop CDMO) Dedicated Capacity/Reserved Suites Risk/Revenue-Sharing & Milestone-based |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biologics Manufacturing Processes | 120 | Manufacturing Directors, Process Engineers |
| Regulatory Compliance in Biologics | 90 | Regulatory Affairs Managers, Quality Control Officers |
| Market Trends in Biologics CDMO | 100 | Market Analysts, Business Development Managers |
| Client Relationships in CDMO Sector | 70 | Client Relationship Managers, Procurement Specialists |
| Innovation in Biologics Development | 80 | R&D Directors, Product Development Scientists |
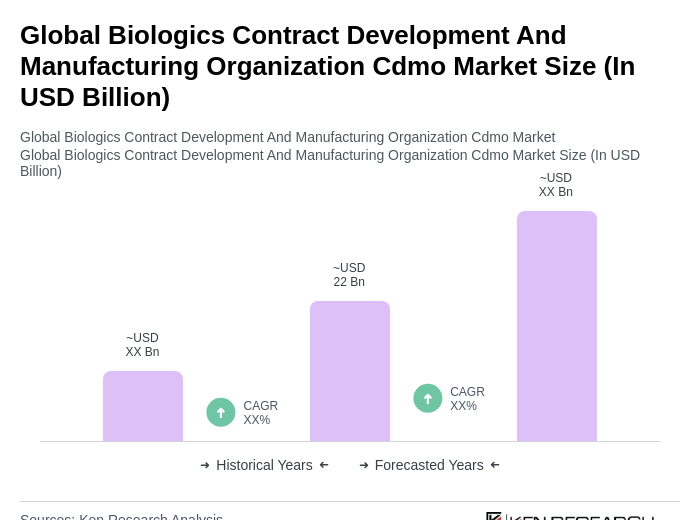
The Global Biologics Contract Development and Manufacturing Organization (CDMO) market is valued at approximately USD 22 billion, driven by increasing demand for biologics and advancements in biomanufacturing technologies.