Region:Global
Author(s):Shubham
Product Code:KRAD0724
Pages:81
Published On:August 2025
By Type:The market is segmented into various types of blood typing methods, including Serological Blood Typing, Molecular Blood Typing, HLA Typing, and Microarray/NGS-based Panels. Each of these methods serves specific purposes in blood transfusion and organ transplantation, with varying levels of accuracy and complexity.
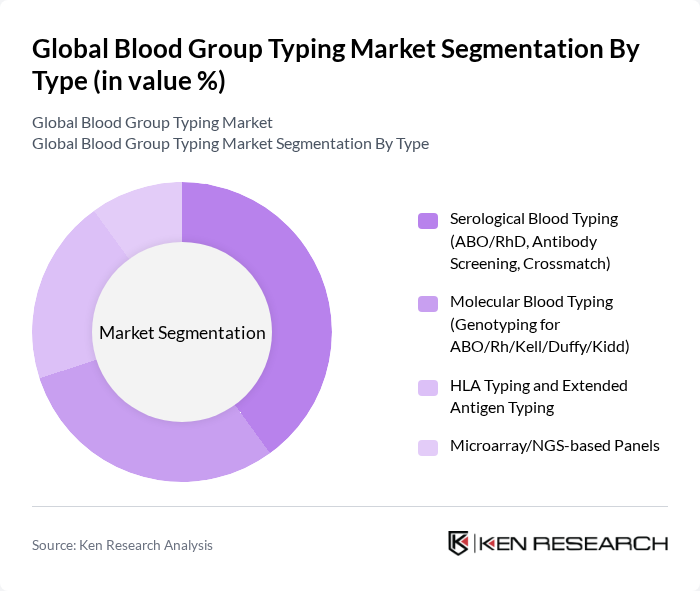
The Serological Blood Typing segment is currently dominating the market due to its established methodologies and widespread acceptance in clinical settings. This traditional method is favored for its reliability and cost-effectiveness, making it the preferred choice for hospitals and blood banks. However, the Molecular Blood Typing segment is gaining traction, particularly for its precision in genotyping, which is crucial for organ transplantation and personalized medicine. The increasing demand for accurate blood typing in complex cases is driving growth in this segment.
By End-User:The market is segmented based on end-users, including Hospitals & Transfusion Services, Blood Banks & Plasma Centers, Independent & Reference Diagnostic Laboratories, and Point-of-Care/Outreach services. Each segment plays a crucial role in the overall blood typing process, catering to different healthcare needs.
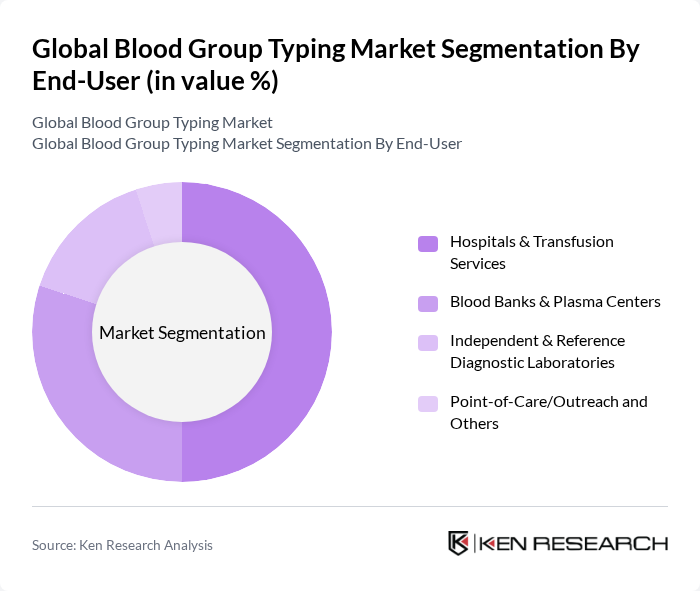
The Hospitals & Transfusion Services segment leads the market, accounting for a significant share due to the high volume of blood transfusions performed annually. These facilities require reliable and rapid blood typing to ensure patient safety during transfusions. Blood Banks & Plasma Centers also contribute significantly, as they are essential for maintaining a steady supply of safe blood products. The growing trend towards point-of-care testing is expected to enhance the outreach segment, although it currently holds a smaller market share.
The Global Blood Group Typing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Grifols, S.A., Bio-Rad Laboratories, Inc., Thermo Fisher Scientific Inc., Abbott Laboratories, QuidelOrtho Corporation, Siemens Healthineers AG, Beckman Coulter, Inc. (a Danaher company), Roche Diagnostics (F. Hoffmann-La Roche Ltd.), Becton, Dickinson and Company (BD), Hologic, Inc., Immucor, Inc. (a Werfen company), DiaSorin S.p.A., Mylab Discovery Solutions Pvt. Ltd., Fujirebio Holdings, Inc., Tecan Group Ltd., Quotient Limited (Alba by Quotient), BAG Diagnostics GmbH (BAG Health Care), BioView Ltd. and Inova Diagnostics (for extended immunohematology) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the blood group typing market in None is poised for significant transformation, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the expansion of healthcare infrastructure will improve access to blood typing services. Additionally, the rise of mobile blood typing units will facilitate on-site testing, further supporting the market's growth trajectory in the future.
| Segment | Sub-Segments |
|---|---|
| By Type | Serological Blood Typing (ABO/RhD, Antibody Screening, Crossmatch) Molecular Blood Typing (Genotyping for ABO/Rh/Kell/Duffy/Kidd) HLA Typing and Extended Antigen Typing Microarray/NGS-based Panels |
| By End-User | Hospitals & Transfusion Services Blood Banks & Plasma Centers Independent & Reference Diagnostic Laboratories Point-of-Care/Outreach and Others |
| By Application | Blood Transfusion & Pretransfusion Testing Organ & Hematopoietic Stem Cell Transplantation (incl. HLA) Prenatal & Neonatal Testing (e.g., HDN risk, NIPT RhD) Disease Diagnosis & Research (Immunohematology, Rare Donor) |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Distribution Channel | Direct Sales to Healthcare Providers Distributors/Channel Partners eProcurement/Online Tenders Others |
| By Product Type | Reagents & Consumables (Anti-sera, Gel Cards, Microplates) Instruments & Analyzers (Automated, Semi-automated) Software & LIMS/Middleware Controls, Calibrators, and Ancillary Supplies |
| By Pricing Model | Reagent Rental/Consumables-led Capital Purchase (Instruments) Subscription/Service & Software Licensing Value-Based/Volume Contracts |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Blood Banks and Transfusion Centers | 120 | Blood Bank Managers, Transfusion Medicine Specialists |
| Clinical Laboratories | 90 | Laboratory Technicians, Quality Control Managers |
| Healthcare Providers | 70 | Physicians, Nurse Practitioners |
| Regulatory Bodies | 40 | Health Policy Analysts, Regulatory Affairs Specialists |
| Research Institutions | 60 | Research Scientists, Academic Professors |
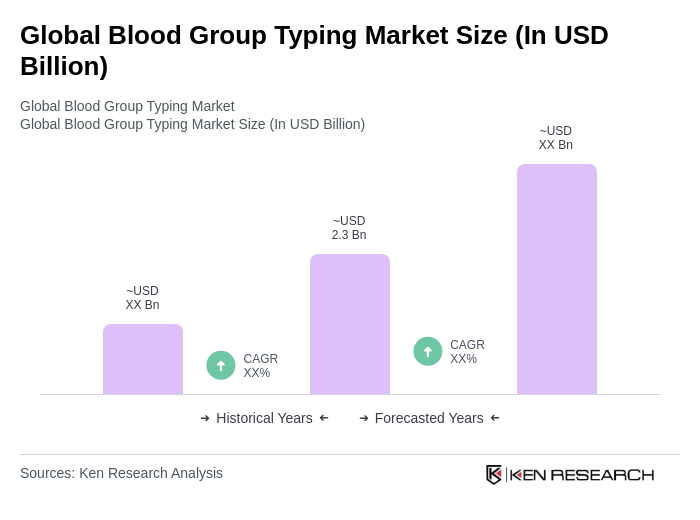
The Global Blood Group Typing Market is valued at approximately USD 2.3 billion, reflecting a significant growth driven by the increasing demand for safe blood transfusions, advancements in technology, and rising surgical volumes and trauma cases.