About the Report
Base Year 2024Listen to the audio summary
Global Clinical Trial Support Services Market Overview
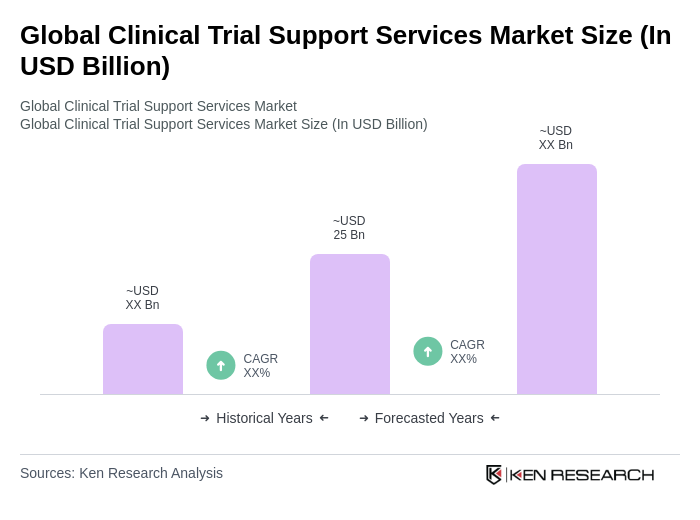
- The Global Clinical Trial Support Services Market is valued at approximately USD 25 billion, based on a five-year historical analysis. This level is supported by multiple industry trackers reporting market sizes in the mid?twenties billion range, reflecting steady demand for outsourced support across patient recruitment, site management, data management, central labs, and logistics .
- Growth is primarily driven by the increasing demand for innovative therapies, the rise in chronic and rare diseases, and the need for faster, cost?efficient drug development processes. Vendors provide services that ensure uninterrupted execution of trials (e.g., patient recruitment, regulatory compliance, data collection), which are increasingly outsourced by pharmaceutical and biotechnology sponsors .
- Key players are predominantly located in North America and Europe, with the United States a major contributor due to its robust healthcare and research infrastructure, high R&D investments, and the concentration of leading pharma and biotech companies conducting most global trials in the region .
- In 2023, the U.S. Food and Drug Administration advanced multiple initiatives to streamline clinical trials with a focus on patient safety, data integrity, and innovative trial designs, including issuing draft and final guidances under mandates such as the Food and Drug Omnibus Reform Act to enable decentralized trials, digital health technologies, and master protocols, with the aim of improving efficiency and access to therapies .
Global Clinical Trial Support Services Market Segmentation
By Type:The market is segmented into various types of clinical trials, including Phase I, Phase II, Phase III, Phase IV, Observational Studies, and Others. Each phase plays a crucial role in the drug development process, with Phase III trials being particularly significant due to their large-scale testing of efficacy and safety. Industry data consistently show that Phase III programs are the most resource?intensive and heavily outsourced, reinforcing their larger share of spend within support services .
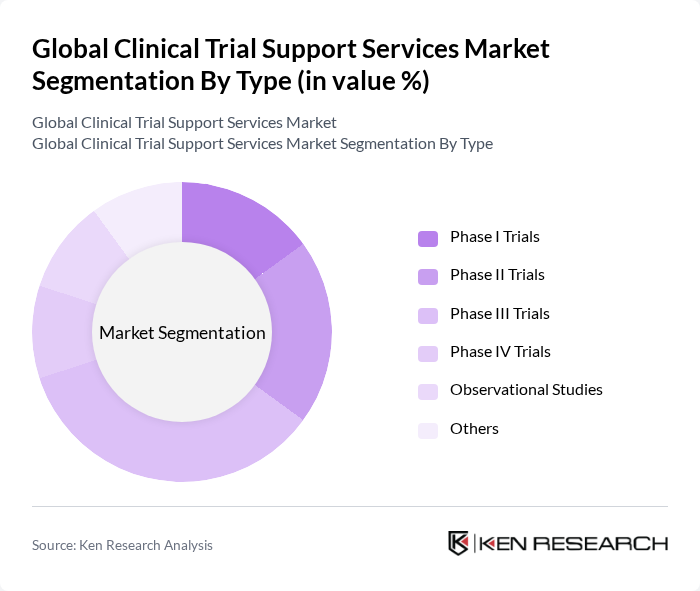
The Phase III Trials segment dominates the market due to its critical role in determining the efficacy and safety of new drugs before regulatory approval. These trials typically involve larger patient populations across multiple geographies, necessitating extensive site management, centralized services, and complex data operations—areas where specialized support services are essential .
By End-User:The market is segmented by end-users, including Pharmaceutical Companies, Biotechnology Firms, Academic Institutions, and Contract Research Organizations (CROs). Each plays a vital role in the clinical trial ecosystem, with pharmaceutical companies being the largest contributors as primary sponsors of global clinical trials. Industry sources indicate a majority of trials are commercially sponsored, aligning with higher outsourcing to support service providers .
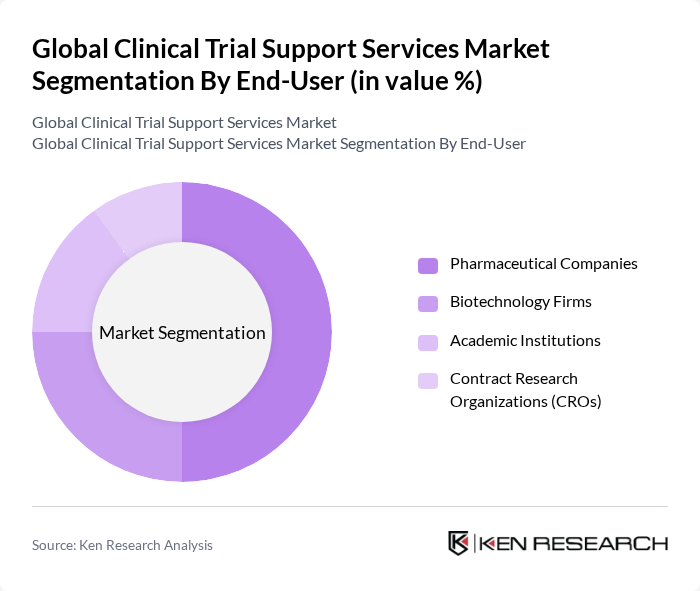
Pharmaceutical Companies dominate the market as they are the primary sponsors of clinical trials, with extensive pipelines and high R&D spend driving demand for external support services to manage scale, timelines, and regulatory requirements. Reports note that around 60% of trials are industry?sponsored, underscoring pharma’s leading role in outsourcing support functions .
Global Clinical Trial Support Services Market Competitive Landscape
The Global Clinical Trial Support Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as IQVIA Inc., Labcorp (Laboratory Corporation of America Holdings), Parexel International Corporation, ICON plc, Charles River Laboratories International, Inc., Medpace Holdings, Inc., Syneos Health, Inc., PPD, Inc. (Thermo Fisher Scientific), WuXi AppTec Co., Ltd., KCR S.A., Celerion, Inc., Worldwide Clinical Trials, Clario (formerly ERT and Bioclinica), Medidata Solutions, Inc. (a Dassault Systèmes company), Advarra, Inc., Veristat, LLC, Caidya, Eurofins Scientific SE, CTI Clinical Trial and Consulting Services, Advanced Clinical contribute to innovation, geographic expansion, and service delivery in this space.
Global Clinical Trial Support Services Market Industry Analysis
Growth Drivers
- Increasing R&D Expenditure:Global spending on research and development (R&D) reached approximately USD 2.5 trillion, reflecting a 5% increase from the previous year. This surge is driven by pharmaceutical companies aiming to innovate and expedite drug development processes. The U.S. alone accounted for over USD 700 billion in R&D investments, highlighting a robust commitment to clinical trials. This financial commitment directly supports the demand for clinical trial support services, facilitating more efficient and effective trial execution.
- Rising Demand for Outsourcing:The global outsourcing market for clinical trials is projected to exceed USD 60 billion, driven by pharmaceutical companies seeking cost-effective solutions. With over 50% of clinical trials now outsourced, organizations are increasingly relying on specialized service providers to manage complex trial logistics. This trend is particularly pronounced in North America and Europe, where regulatory complexities necessitate expert support, thereby fueling the growth of clinical trial support services.
- Technological Advancements in Clinical Trials:The integration of advanced technologies, such as electronic data capture (EDC) and remote monitoring, is transforming clinical trials. In future, the market for clinical trial technology solutions is expected to reach USD 15 billion, driven by the need for efficiency and accuracy. These innovations enhance data collection and patient monitoring, reducing trial timelines and costs. As a result, clinical trial support services are increasingly adopting these technologies to improve trial outcomes and streamline processes.
Market Challenges
- Regulatory Compliance Issues:Navigating the complex regulatory landscape remains a significant challenge for clinical trial support services. In future, the average time for regulatory approval in the U.S. is projected to be around 12 months, with delays often caused by stringent compliance requirements. This complexity can lead to increased costs and extended timelines for clinical trials, impacting the overall efficiency of the drug development process and posing a challenge for service providers.
- High Costs of Clinical Trials:The average cost of conducting a clinical trial in future is estimated to be around USD 2.6 million, a figure that has steadily increased over the past decade. These high costs are attributed to factors such as patient recruitment, site management, and regulatory compliance. As a result, many organizations face budget constraints, which can limit their ability to conduct extensive trials and ultimately hinder the growth of clinical trial support services.
Global Clinical Trial Support Services Market Future Outlook
The future of clinical trial support services is poised for significant transformation, driven by technological advancements and evolving patient needs. As the industry increasingly embraces decentralized trials, the focus will shift towards enhancing patient engagement and utilizing real-world evidence to inform trial designs. Furthermore, collaborations with biotech firms are expected to foster innovation, enabling more efficient trial processes. These trends will likely reshape the landscape, making clinical trials more accessible and efficient, ultimately benefiting both patients and researchers.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets, particularly in Asia and Latin America, present significant growth opportunities for clinical trial support services. With a combined population exceeding 4 billion, these regions offer diverse patient populations and lower operational costs. In future, investments in clinical trials in these markets are expected to increase by 20%, driven by the need for localized research and faster patient recruitment.
- Adoption of Decentralized Trials:The shift towards decentralized clinical trials is gaining momentum, with an estimated 30% of trials adopting this model in future. This approach enhances patient accessibility and engagement, allowing for remote monitoring and data collection. As a result, clinical trial support services that specialize in decentralized methodologies are likely to see increased demand, positioning themselves as key players in the evolving landscape of clinical research.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Observational Studies Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic Institutions Contract Research Organizations (CROs) |
| By Study Design | Randomized Controlled Trials Non-Randomized Trials Cross-Sectional Studies |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Service Type | Site Management Patient Recruitment & Retention Data Management & Biostatistics Regulatory Affairs & Start-Up (IRB/IEC, submissions) Pharmacovigilance & Safety Monitoring Clinical Trial Supply, Logistics & Ancillary Services eClinical Solutions (CTMS, EDC, eCOA/ePRO, RTSM/IWRS) Central Laboratory & Bioanalytical Services Medical Writing & Scientific Communication Feasibility, Site Selection & Study Start-Up Decentralized/Hybrid Trial Enablement (home health, wearables, tele-visits) |
| By Funding Source | Public Funding Private Funding Non-Profit Organizations Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical and Biotechnology Companies
Clinical Research Organizations (CROs)
Healthcare Providers and Hospitals
Medical Device Manufacturers
Insurance Companies and Payers
Pharmaceutical Supply Chain Stakeholders
Players Mentioned in the Report:
IQVIA Inc.
Labcorp (Laboratory Corporation of America Holdings)
Parexel International Corporation
ICON plc
Charles River Laboratories International, Inc.
Medpace Holdings, Inc.
Syneos Health, Inc.
PPD, Inc. (Thermo Fisher Scientific)
WuXi AppTec Co., Ltd.
KCR S.A.
Celerion, Inc.
Worldwide Clinical Trials
Clario (formerly ERT and Bioclinica)
Medidata Solutions, Inc. (a Dassault Systemes company)
Advarra, Inc.
Veristat, LLC
Caidya
Eurofins Scientific SE
CTI Clinical Trial and Consulting Services
Advanced Clinical
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Clinical Trial Support Services Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Clinical Trial Support Services Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Clinical Trial Support Services Market Analysis
3.1 Growth Drivers
3.1.1 Increasing R&D Expenditure
3.1.2 Rising Demand for Outsourcing
3.1.3 Technological Advancements in Clinical Trials
3.1.4 Growing Focus on Patient-Centric Trials
3.2 Market Challenges
3.2.1 Regulatory Compliance Issues
3.2.2 High Costs of Clinical Trials
3.2.3 Data Privacy Concerns
3.2.4 Recruitment and Retention of Participants
3.3 Market Opportunities
3.3.1 Expansion into Emerging Markets
3.3.2 Adoption of Decentralized Trials
3.3.3 Integration of AI and Machine Learning
3.3.4 Collaborations with Biotech Firms
3.4 Market Trends
3.4.1 Increased Use of Real-World Evidence
3.4.2 Shift Towards Virtual Trials
3.4.3 Emphasis on Patient Engagement
3.4.4 Growth of Adaptive Trial Designs
3.5 Government Regulation
3.5.1 FDA Guidelines on Clinical Trials
3.5.2 EMA Regulations for Drug Development
3.5.3 ICH GCP Guidelines
3.5.4 Data Protection Regulations (GDPR)
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Clinical Trial Support Services Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Clinical Trial Support Services Market Segmentation
8.1 By Type
8.1.1 Phase I Trials
8.1.2 Phase II Trials
8.1.3 Phase III Trials
8.1.4 Phase IV Trials
8.1.5 Observational Studies
8.1.6 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Academic Institutions
8.2.4 Contract Research Organizations (CROs)
8.3 By Study Design
8.3.1 Randomized Controlled Trials
8.3.2 Non-Randomized Trials
8.3.3 Cross-Sectional Studies
8.4 By Therapeutic Area
8.4.1 Oncology
8.4.2 Cardiovascular
8.4.3 Neurology
8.4.4 Infectious Diseases
8.4.5 Others
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.6 By Service Type
8.6.1 Site Management
8.6.2 Patient Recruitment & Retention
8.6.3 Data Management & Biostatistics
8.6.4 Regulatory Affairs & Start-Up (IRB/IEC, submissions)
8.6.5 Pharmacovigilance & Safety Monitoring
8.6.6 Clinical Trial Supply, Logistics & Ancillary Services
8.6.7 eClinical Solutions (CTMS, EDC, eCOA/ePRO, RTSM/IWRS)
8.6.8 Central Laboratory & Bioanalytical Services
8.6.9 Medical Writing & Scientific Communication
8.6.10 Feasibility, Site Selection & Study Start-Up
8.6.11 Decentralized/Hybrid Trial Enablement (home health, wearables, tele-visits)
8.7 By Funding Source
8.7.1 Public Funding
8.7.2 Private Funding
8.7.3 Non-Profit Organizations
8.7.4 Others
9. Global Clinical Trial Support Services Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Global, Regional, Niche)
9.2.3 Revenue from Clinical Trial Support Services (USD, latest FY)
9.2.4 CAGR of Services Revenue (3–5 years)
9.2.5 Geographic Footprint (number of countries/sites)
9.2.6 Therapeutic Coverage Breadth (number of TAs supported)
9.2.7 Patient Enrollment Velocity (patients/month/site)
9.2.8 Study Start-Up Cycle Time (site activation days)
9.2.9 Protocol Deviation Rate (%)
9.2.10 On-Time/On-Budget Delivery Rate (%)
9.2.11 Decentralized Trial Capability Index (DCT-enabled studies %)
9.2.12 Technology Stack Maturity (CTMS/EDC/eCOA integration score)
9.2.13 Quality & Compliance KPIs (audit findings per study, CAPA closure time)
9.2.14 Repeat Business/Client Retention Rate (%)
9.2.15 Average Pricing vs. Peer Median (index)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 IQVIA Inc.
9.5.2 Labcorp (Laboratory Corporation of America Holdings)
9.5.3 Parexel International Corporation
9.5.4 ICON plc
9.5.5 Charles River Laboratories International, Inc.
9.5.6 Medpace Holdings, Inc.
9.5.7 Syneos Health, Inc.
9.5.8 PPD, Inc. (Thermo Fisher Scientific)
9.5.9 WuXi AppTec Co., Ltd.
9.5.10 KCR S.A.
9.5.11 Celerion, Inc.
9.5.12 Worldwide Clinical Trials
9.5.13 Clario (formerly ERT and Bioclinica)
9.5.14 Medidata Solutions, Inc. (a Dassault Systèmes company)
9.5.15 Advarra, Inc.
9.5.16 Veristat, LLC
9.5.17 Caidya
9.5.18 Eurofins Scientific SE
9.5.19 CTI Clinical Trial and Consulting Services
9.5.20 Advanced Clinical
10. Global Clinical Trial Support Services Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Service Providers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Clinical Research Facilities
10.2.2 Spending on Technology Integration
10.2.3 Budget for Patient Engagement Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Delays in Trial Approvals
10.3.2 High Operational Costs
10.3.3 Difficulty in Patient Recruitment
10.4 User Readiness for Adoption
10.4.1 Awareness of Clinical Trial Benefits
10.4.2 Training and Support Needs
10.4.3 Technology Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Trial Success
10.5.2 Opportunities for Future Trials
10.5.3 Feedback Mechanisms for Improvement
11. Global Clinical Trial Support Services Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Key Partnerships
1.5 Cost Structure Analysis
1.6 Customer Segmentation
1.7 Competitive Advantage
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Options
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from global health organizations and clinical trial registries
- Review of published articles in peer-reviewed journals focusing on clinical trial methodologies
- Examination of market research reports detailing trends in clinical trial support services
Primary Research
- Interviews with clinical trial managers and coordinators from leading pharmaceutical companies
- Surveys targeting clinical research organizations (CROs) to gather insights on service offerings
- Field interviews with regulatory affairs specialists to understand compliance challenges
Validation & Triangulation
- Cross-validation of findings through multiple data sources including industry publications and expert opinions
- Triangulation of qualitative insights from interviews with quantitative data from market reports
- Sanity checks conducted through expert panel reviews to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the global clinical trial market size based on pharmaceutical R&D expenditure
- Segmentation of market data by therapeutic area and geographical region
- Incorporation of trends in outsourcing clinical trial services by biopharmaceutical companies
Bottom-up Modeling
- Collection of data on service pricing from leading clinical trial support service providers
- Estimation of service volumes based on historical data from completed clinical trials
- Analysis of growth rates in clinical trial phases (Phase I, II, III) to project future demand
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating variables such as drug approval rates and technological advancements
- Scenario modeling based on potential changes in regulatory frameworks and funding availability
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 120 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Biotechnology Clinical Trials | 90 | Research Scientists, Project Managers |
| Medical Device Trials | 80 | Clinical Research Associates, Quality Assurance Managers |
| Oncology Trials | 70 | Oncologists, Clinical Operations Directors |
| Cardiovascular Trials | 80 | Cardiologists, Clinical Data Managers |
Frequently Asked Questions
What is the current value of the Global Clinical Trial Support Services Market?
The Global Clinical Trial Support Services Market is valued at approximately USD 25 billion, reflecting steady demand for outsourced support services across various areas such as patient recruitment, site management, and data management.