Global Clinical Trials Site Management Organizations (SMO) Market Overview
- The Global Clinical Trials Site Management Organizations (SMO) Market is valued at USD 6.6 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing number of clinical trials, the rising demand for efficient site management, and the growing complexity of clinical studies. The need for specialized services to streamline trial processes and enhance patient recruitment has further propelled market expansion. Additional growth drivers include the surge in drug discovery activities, the prevalence of chronic diseases, and the trend toward outsourcing clinical trial operations to SMOs for operational efficiency and regulatory compliance .
- Key regions dominating the market include North America and Europe, primarily due to their advanced healthcare infrastructure, high investment in research and development, and a large number of pharmaceutical and biotechnology companies. The presence of established regulatory frameworks and a skilled workforce also contribute to their market leadership. North America, in particular, leads the market owing to its extensive clinical trial activity and robust network of clinical research sites .
- In 2023, the U.S. Food and Drug Administration (FDA) implemented the “Modernization of Clinical Trials Conduct and Reporting” guidance under the authority of the Food and Drug Administration (FDA), United States Department of Health and Human Services. This guidance focuses on streamlining the approval process for clinical trial protocols, enhancing patient safety measures, and promoting the use of innovative trial designs, thereby encouraging more organizations to engage in clinical research and expand their operational capabilities .
 Market.png)
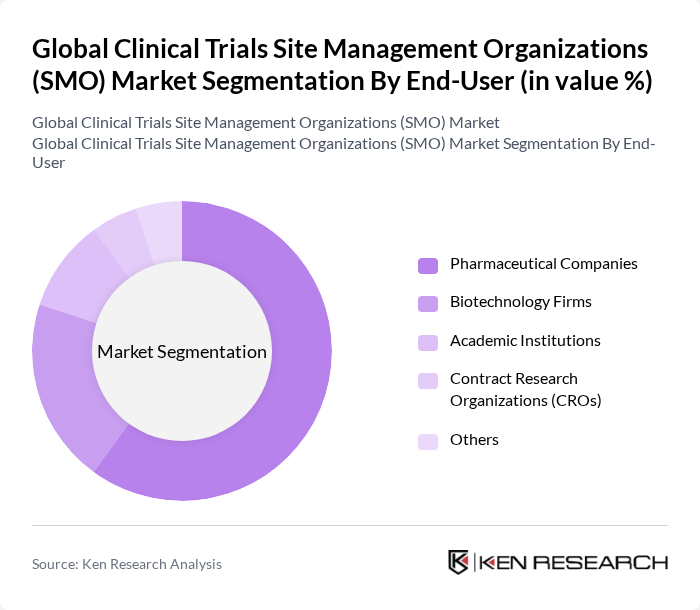
Global Clinical Trials Site Management Organizations (SMO) Market Segmentation
By Type:The market is segmented into Full-Service SMOs, Specialty SMOs, Virtual/Decentralized SMOs, and Regional/Niche SMOs. Full-Service SMOs dominate the market due to their comprehensive offerings that cover all aspects of clinical trial management, making them a preferred choice for pharmaceutical companies seeking efficiency and expertise. Specialty SMOs are also gaining traction as they focus on specific therapeutic areas, catering to the unique needs of clients. The growing complexity of clinical trials, particularly in oncology, neurology, and rare diseases, is driving demand for both full-service and specialty SMOs .
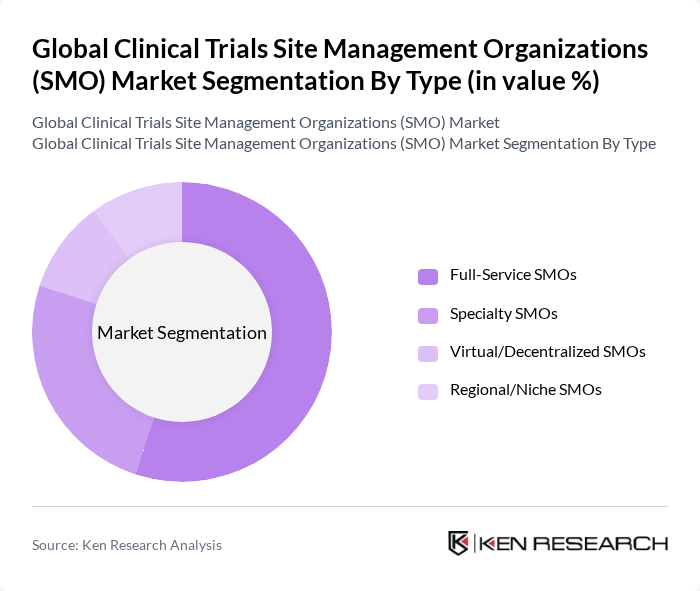
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic Institutions, Contract Research Organizations (CROs), and Others. Pharmaceutical Companies are the leading end-users, driven by their need for extensive clinical trials to bring new drugs to market. The increasing collaboration between pharmaceutical companies and SMOs for efficient trial management is a significant trend in this segment. Biotechnology firms and CROs are also notable end-users, leveraging SMO expertise for specialized and outsourced trial support .
Global Clinical Trials Site Management Organizations (SMO) Market Competitive Landscape
The Global Clinical Trials Site Management Organizations (SMO) Market is characterized by a dynamic mix of regional and international players. Leading participants such as ICON plc, Covance Inc., PPD, Inc., Syneos Health, Medpace, Inc., PRA Health Sciences, Charles River Laboratories, Parexel International Corporation, Clinedge, WCG (WIRB-Copernicus Group), ClinChoice, Access Clinical Research, FOMAT Medical Research Inc., SGS, KV Clinical, SMO-Pharmina, Xylem Clinical Research, Aurum Clinical Research, Sarah Cannon, EthosExcel, Grand Pacific CRO, PANACRO, Worldwide Clinical Trials, Clinipace, Celerion, Veristat, BioClinica, ERT contribute to innovation, geographic expansion, and service delivery in this space .
Global Clinical Trials Site Management Organizations (SMO) Market Industry Analysis
Growth Drivers
- Increasing Demand for Clinical Trials:The global clinical trials market is projected to reach $65 billion, driven by a surge in demand for innovative therapies. The World Health Organization reported that over 1.5 million clinical trials were registered globally, reflecting a growing need for effective treatments. This demand is further fueled by the increasing prevalence of diseases, necessitating more extensive research and development efforts, particularly in oncology and rare diseases, which are expected to dominate the clinical trial landscape.
- Rise in Chronic Diseases:Chronic diseases, such as diabetes and cardiovascular conditions, are on the rise, with the World Health Organization estimating that 60% of the global population will be living with at least one chronic condition. This alarming trend is driving the need for clinical trials focused on long-term treatment solutions. In future, the global expenditure on chronic disease management reached $8 trillion, highlighting the urgent need for innovative clinical research to address these health challenges effectively.
- Technological Advancements in Trial Management:The integration of advanced technologies in clinical trial management is revolutionizing the industry. In future, investments in clinical trial technology reached $15 billion, with significant advancements in data analytics, patient monitoring, and trial management systems. These technologies enhance efficiency, reduce costs, and improve patient engagement, making clinical trials more attractive to sponsors. As a result, the adoption of these technologies is expected to accelerate, further driving the growth of SMOs in the coming years.
Market Challenges
- Regulatory Complexities:Navigating the regulatory landscape remains a significant challenge for SMOs. In future, the average time for regulatory approval of clinical trials was approximately 12 months, with variations across regions. The increasing complexity of regulations, particularly in emerging markets, adds to the burden on SMOs. Compliance with Good Clinical Practice (GCP) guidelines and local regulations requires substantial resources, which can hinder the timely execution of clinical trials and increase operational costs.
- Recruitment and Retention of Trial Participants:The recruitment and retention of participants in clinical trials pose ongoing challenges. In future, it was reported that nearly 30% of clinical trials failed to meet their enrollment targets, leading to delays and increased costs. Factors such as stringent eligibility criteria, participant awareness, and logistical issues contribute to these challenges. As the competition for participants intensifies, SMOs must develop innovative strategies to enhance recruitment and retention rates to ensure successful trial outcomes.
Global Clinical Trials Site Management Organizations (SMO) Market Future Outlook
The future of the clinical trials site management organizations market appears promising, driven by the increasing adoption of decentralized clinical trials and the integration of artificial intelligence in trial processes. As regulatory bodies adapt to new methodologies, SMOs are expected to leverage technology to enhance patient engagement and streamline operations. Furthermore, the growing emphasis on real-world evidence will likely shape trial designs, making them more relevant to patient needs and improving overall trial efficiency in the coming years.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets present significant opportunities for SMOs, with a projected growth rate of 15% in clinical trials. Countries like India and Brazil are becoming attractive destinations due to their large patient populations and lower operational costs. This expansion can enhance trial diversity and accelerate the development of new therapies tailored to diverse populations.
- Adoption of Decentralized Clinical Trials:The shift towards decentralized clinical trials is creating new avenues for SMOs. In future, it is estimated that 40% of clinical trials will adopt decentralized methodologies, improving patient access and engagement. This trend not only reduces geographical barriers but also enhances data collection efficiency, making it a lucrative opportunity for SMOs to innovate and expand their service offerings.
 Market.png)