About the Report
Base Year 2024Global Contract Development Manufacturing Organization Market Overview
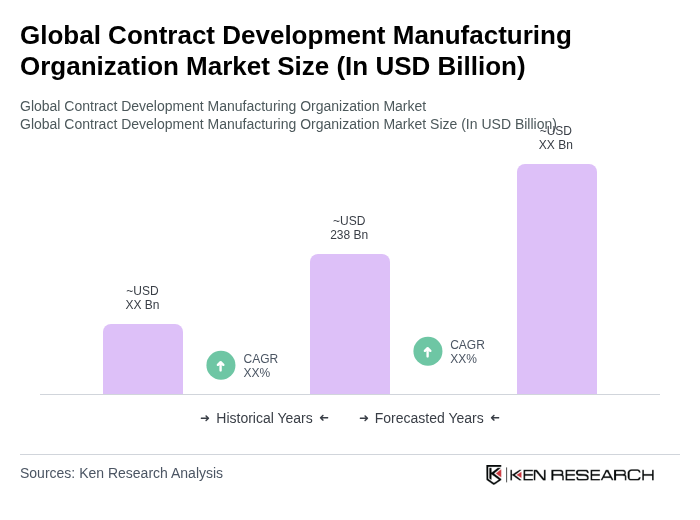
- The Global Contract Development Manufacturing Organization Market is valued at USD 238 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for outsourcing in the pharmaceutical and biotechnology sectors, as companies seek to reduce costs and enhance efficiency. The rise in drug development activities, particularly in biologics, advanced therapies, and complex generics, has further fueled the market's expansion. Additional growth drivers include the expansion of personalized medicine, increased investment in high-potency and injectable products, and the need for supply chain resilience in a post-pandemic environment .
- Key players in this market are predominantly located in North America and Europe, with the United States and Germany leading due to their advanced healthcare infrastructure, significant investment in R&D, and a high concentration of pharmaceutical companies. These regions benefit from a robust regulatory framework and a skilled workforce, making them attractive for contract development and manufacturing services. North America holds the largest market share, accounting for nearly 39% of global CDMO activity, while Asia-Pacific is rapidly growing due to investments in local manufacturing and capacity expansion .
- In 2024, the U.S. Food and Drug Administration (FDA) issued the “Contract Manufacturing Arrangements for Drugs: Quality Agreements Guidance for Industry,” which provides binding recommendations for ensuring compliance with Good Manufacturing Practices (GMP). This guidance emphasizes the importance of written quality agreements, clear delineation of responsibilities, and robust risk management in contract manufacturing, thereby strengthening product safety and efficacy standards .
Global Contract Development Manufacturing Organization Market Segmentation
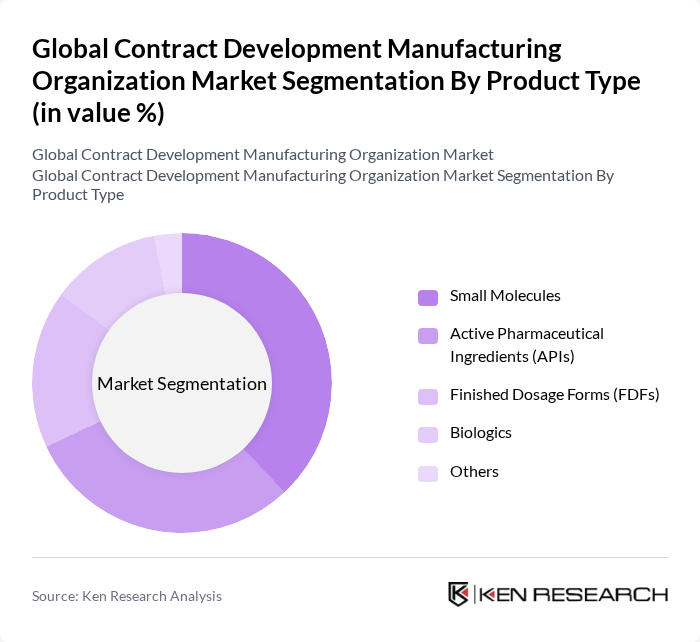
By Product Type:The product type segmentation includes Active Pharmaceutical Ingredients (APIs), Finished Dosage Forms (FDFs), Biologics, Small Molecules, and Others. Among these, Small Molecules currently dominate the market, driven by their widespread application in various therapeutic areas and the established nature of small molecule drug development. However, the biologics segment is experiencing the fastest growth due to increasing demand for advanced therapies and biosimilars. Outsourcing of API production remains significant, particularly for generics and high-potency compounds, while the complexity of biologics manufacturing continues to drive specialized service demand .
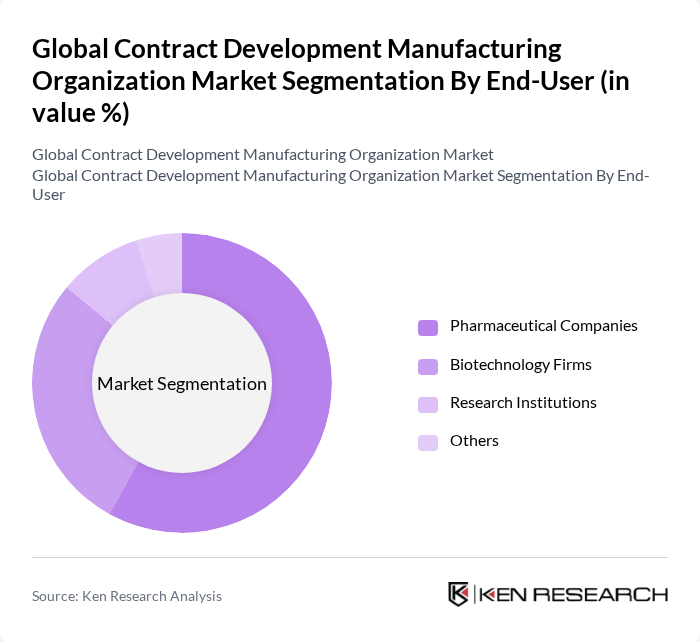
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, and Others. Pharmaceutical Companies remain the leading end-users, driven by the need for efficient production capabilities, cost containment, and the increasing complexity of drug formulations. Biotechnology Firms are rapidly increasing their share, particularly in the biologics and cell/gene therapy segments, as they leverage CDMO partnerships for specialized manufacturing and scale-up .
Global Contract Development Manufacturing Organization Market Competitive Landscape
The Global Contract Development Manufacturing Organization Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group, Catalent, Inc., WuXi AppTec, Thermo Fisher Scientific, Recipharm AB, Samsung Biologics, Patheon (Part of Thermo Fisher Scientific), Aenova Group, Siegfried Holding AG, Famar, Alcami Corporation, CordenPharma, Piramal Pharma Solutions, Jubilant HollisterStier, Bachem Holding AG, Boehringer Ingelheim BioXcellence, AbbVie Contract Manufacturing, Dr. Reddy’s Laboratories, Hovione, and Fareva contribute to innovation, geographic expansion, and service delivery in this space.
Global Contract Development Manufacturing Organization Market Industry Analysis
Growth Drivers
- Increasing Demand for Outsourcing in Pharmaceutical Development:The global pharmaceutical outsourcing market is projected to reach $400 billion by 2025, driven by the need for cost-effective solutions. Companies are increasingly outsourcing drug development to contract development manufacturing organizations (CDMOs) to focus on core competencies. This trend is supported by a 15% increase in outsourcing activities reported by major pharmaceutical firms in future, highlighting a shift towards leveraging external expertise for enhanced efficiency and innovation.
- Rising Investment in Biopharmaceuticals:The biopharmaceutical sector is experiencing unprecedented growth, with investments exceeding $200 billion in future. This surge is fueled by advancements in biologics and personalized medicine, which require specialized manufacturing capabilities. The World Health Organization reported a 25% increase in biopharmaceutical approvals, indicating a robust pipeline that necessitates the services of CDMOs to meet production demands and regulatory requirements effectively.
- Technological Advancements in Drug Development:The integration of cutting-edge technologies, such as AI and automation, is revolutionizing drug development processes. In future, over 60% of CDMOs adopted AI-driven solutions to enhance efficiency and reduce time-to-market. This technological shift is supported by a $10 billion investment in digital transformation initiatives across the pharmaceutical industry, enabling CDMOs to streamline operations and improve product quality, thereby attracting more clients.
Market Challenges
- Stringent Regulatory Requirements:Compliance with regulatory standards remains a significant challenge for CDMOs. In future, the FDA issued over 1,000 warning letters related to manufacturing violations, emphasizing the need for strict adherence to Good Manufacturing Practices (GMP). The complexity of navigating these regulations can lead to increased operational costs, with estimates suggesting that compliance-related expenses can account for up to 20% of total production costs for CDMOs.
- High Competition Among Service Providers:The CDMO market is highly competitive, with over 1,500 players globally. This saturation leads to price wars and reduced profit margins, as companies strive to differentiate themselves. In future, the average profit margin for CDMOs fell to 10%, down from 15%, as firms invest heavily in marketing and service diversification to maintain market share amidst fierce competition.
Global Contract Development Manufacturing Organization Market Future Outlook
The future of the CDMO market appears promising, driven by ongoing advancements in technology and increasing demand for biopharmaceuticals. As companies continue to seek cost-effective solutions, the trend towards outsourcing is expected to strengthen. Additionally, the rise of personalized medicine will create new opportunities for CDMOs to innovate and expand their service offerings, particularly in emerging markets where healthcare investments are surging. The focus on sustainability will also shape operational strategies in the coming years.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets, particularly in Asia and Africa, are witnessing rapid growth in healthcare spending, projected to reach $1 trillion by 2026. CDMOs can capitalize on this trend by establishing local partnerships and expanding their footprint, thereby tapping into new customer bases and enhancing service accessibility in these regions.
- Adoption of Digital Technologies in Manufacturing:The shift towards Industry 4.0 is creating significant opportunities for CDMOs to enhance operational efficiency. Investments in digital technologies, such as IoT and blockchain, are expected to increase by 30% in future, allowing CDMOs to improve traceability, reduce waste, and optimize supply chain management, ultimately leading to better service delivery.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Active Pharmaceutical Ingredients (APIs) Finished Dosage Forms (FDFs) Biologics Small Molecules Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Service Type | Drug Development Services Manufacturing Services Packaging Services Analytical Testing Services Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Delivery Method | Oral Injectable Transdermal Others |
| By Contract Type | Fixed-Price Contracts Cost-Plus Contracts Time and Materials Contracts Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical and Biotechnology Companies
Medical Device Manufacturers
Contract Research Organizations
Supply Chain and Logistics Providers
Quality Assurance and Compliance Professionals
Healthcare Providers and Institutions
Players Mentioned in the Report:
Lonza Group
Catalent, Inc.
WuXi AppTec
Thermo Fisher Scientific
Recipharm AB
Samsung Biologics
Patheon (Part of Thermo Fisher Scientific)
Aenova Group
Siegfried Holding AG
Famar
Alcami Corporation
CordenPharma
Piramal Pharma Solutions
Jubilant HollisterStier
Bachem Holding AG
Boehringer Ingelheim BioXcellence
AbbVie Contract Manufacturing
Dr. Reddys Laboratories
Hovione
Fareva
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Contract Development Manufacturing Organization Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Contract Development Manufacturing Organization Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Contract Development Manufacturing Organization Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for outsourcing in pharmaceutical development
3.1.2 Rising investment in biopharmaceuticals
3.1.3 Technological advancements in drug development
3.1.4 Growing focus on cost efficiency and time-to-market
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High competition among service providers
3.2.3 Quality assurance and compliance issues
3.2.4 Fluctuating demand from end-users
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Strategic partnerships and collaborations
3.3.3 Development of personalized medicine
3.3.4 Adoption of digital technologies in manufacturing
3.4 Market Trends
3.4.1 Increasing use of AI and machine learning in drug development
3.4.2 Shift towards integrated service offerings
3.4.3 Growing emphasis on sustainability in manufacturing
3.4.4 Rise of contract manufacturing for biologics
3.5 Government Regulation
3.5.1 FDA guidelines for contract manufacturers
3.5.2 EMA regulations for drug approval
3.5.3 Compliance with GMP standards
3.5.4 Intellectual property protection laws
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Contract Development Manufacturing Organization Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Contract Development Manufacturing Organization Market Segmentation
8.1 By Product Type
8.1.1 Active Pharmaceutical Ingredients (APIs)
8.1.2 Finished Dosage Forms (FDFs)
8.1.3 Biologics
8.1.4 Small Molecules
8.1.5 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Research Institutions
8.2.4 Others
8.3 By Region
8.3.1 North America
8.3.2 Europe
8.3.3 Asia-Pacific
8.3.4 Latin America
8.3.5 Middle East & Africa
8.4 By Service Type
8.4.1 Drug Development Services
8.4.2 Manufacturing Services
8.4.3 Packaging Services
8.4.4 Analytical Testing Services
8.4.5 Others
8.5 By Therapeutic Area
8.5.1 Oncology
8.5.2 Cardiovascular
8.5.3 Neurology
8.5.4 Infectious Diseases
8.5.5 Others
8.6 By Delivery Method
8.6.1 Oral
8.6.2 Injectable
8.6.3 Transdermal
8.6.4 Others
8.7 By Contract Type
8.7.1 Fixed-Price Contracts
8.7.2 Cost-Plus Contracts
8.7.3 Time and Materials Contracts
8.7.4 Others
9. Global Contract Development Manufacturing Organization Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Revenue (USD Billion, latest available)
9.2.3 Global Manufacturing Capacity (in metric tons or batches/year)
9.2.4 Number of Facilities (by region)
9.2.5 Regulatory Approvals (FDA, EMA, etc.)
9.2.6 Client Retention Rate (%)
9.2.7 Average Contract Value (USD Million)
9.2.8 Time-to-Market Efficiency (average days from development to launch)
9.2.9 Client Satisfaction Score (based on industry surveys)
9.2.10 R&D Investment as % of Revenue
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Lonza Group
9.5.2 Catalent, Inc.
9.5.3 WuXi AppTec
9.5.4 Thermo Fisher Scientific
9.5.5 Recipharm AB
9.5.6 Samsung Biologics
9.5.7 Patheon (Part of Thermo Fisher Scientific)
9.5.8 Aenova Group
9.5.9 Siegfried Holding AG
9.5.10 Famar
9.5.11 Alcami Corporation
9.5.12 CordenPharma
9.5.13 Piramal Pharma Solutions
9.5.14 Jubilant HollisterStier
9.5.15 Bachem Holding AG
9.5.16 Boehringer Ingelheim BioXcellence
9.5.17 AbbVie Contract Manufacturing
9.5.18 Dr. Reddy’s Laboratories
9.5.19 Hovione
9.5.20 Fareva
10. Global Contract Development Manufacturing Organization Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Contracting Preferences
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in R&D
10.2.2 Spending on Manufacturing Facilities
10.2.3 Budget for Outsourcing Services
10.2.4 Cost Management Strategies
10.3 Pain Point Analysis by End-User Category
10.3.1 Quality Control Issues
10.3.2 Supply Chain Disruptions
10.3.3 Regulatory Compliance Challenges
10.3.4 Cost Overruns
10.4 User Readiness for Adoption
10.4.1 Technology Adoption Rates
10.4.2 Training and Support Needs
10.4.3 Change Management Strategies
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Scalability of Solutions
10.5.4 Future Investment Plans
11. Global Contract Development Manufacturing Organization Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Identification of Market Gaps
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Identification
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
2.6 Customer Engagement Tactics
2.7 Performance Metrics
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
3.5 Distribution Partnerships
3.6 Inventory Management
3.7 Performance Tracking
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Discount Strategies
4.7 Pricing Optimization Techniques
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends Identification
5.4 Customer Feedback Analysis
5.5 Market Research Insights
5.6 Product Development Opportunities
5.7 Future Demand Projections
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Support Strategies
6.4 Feedback Mechanisms
6.5 Relationship Management Tools
6.6 Customer Retention Strategies
6.7 Performance Metrics
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Unique Selling Points
7.4 Customer-Centric Approaches
7.5 Competitive Advantages
7.6 Market Differentiation Strategies
7.7 Performance Metrics
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Marketing Campaigns
8.5 Training and Development
8.6 Performance Monitoring
8.7 Continuous Improvement Strategies
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
10.5 Risk Assessment
10.6 Strategic Fit Analysis
10.7 Performance Metrics
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Financial Projections
11.4 Funding Sources
11.5 Risk Management Strategies
11.6 Performance Metrics
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnership Opportunities
12.3 Risk Mitigation Strategies
12.4 Performance Metrics
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
13.3 Financial Health Assessment
13.4 Performance Metrics
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
14.4 Strategic Alliances
14.5 Performance Metrics
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for Key Activities
15.2.2 Milestone Tracking
15.2.3 Performance Metrics
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from global market research firms focusing on contract development and manufacturing organizations (CDMOs)
- Market analysis from pharmaceutical and biotechnology journals detailing trends in outsourcing
- Regulatory frameworks and guidelines from health authorities such as the FDA and EMA
Primary Research
- Interviews with executives from leading CDMOs to understand service offerings and market dynamics
- Surveys targeting R&D managers in pharmaceutical companies regarding outsourcing preferences
- Focus groups with industry experts to discuss emerging trends and challenges in the CDMO landscape
Validation & Triangulation
- Cross-validation of data through multiple industry reports and expert opinions
- Triangulation of findings from primary interviews with secondary data sources
- Sanity checks conducted through expert panel reviews to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of global pharmaceutical spending trends to estimate the CDMO market size
- Segmentation of the market by therapeutic area and service type (e.g., API manufacturing, formulation development)
- Incorporation of growth rates from emerging markets and biopharmaceutical sectors
Bottom-up Modeling
- Data collection on revenue figures from key CDMO players to establish a baseline
- Estimation of service demand based on historical outsourcing rates in the pharmaceutical industry
- Volume and pricing analysis for various CDMO services to derive market potential
Forecasting & Scenario Analysis
- Multi-variable forecasting models incorporating factors such as drug development timelines and regulatory changes
- Scenario analysis based on potential shifts in outsourcing trends and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical API Manufacturing | 120 | Production Managers, Quality Assurance Leads |
| Biologics Development Services | 100 | R&D Directors, Project Managers |
| Formulation Development | 80 | Formulation Scientists, Regulatory Affairs Specialists |
| Clinical Trial Material Supply | 70 | Clinical Operations Managers, Supply Chain Coordinators |
| Packaging and Labeling Services | 60 | Packaging Engineers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Global Contract Development Manufacturing Organization Market?
The Global Contract Development Manufacturing Organization Market is valued at approximately USD 238 billion, reflecting significant growth driven by increased outsourcing in the pharmaceutical and biotechnology sectors, particularly in drug development activities and personalized medicine.