Region:Global
Author(s):Shubham
Product Code:KRAC0727
Pages:81
Published On:August 2025
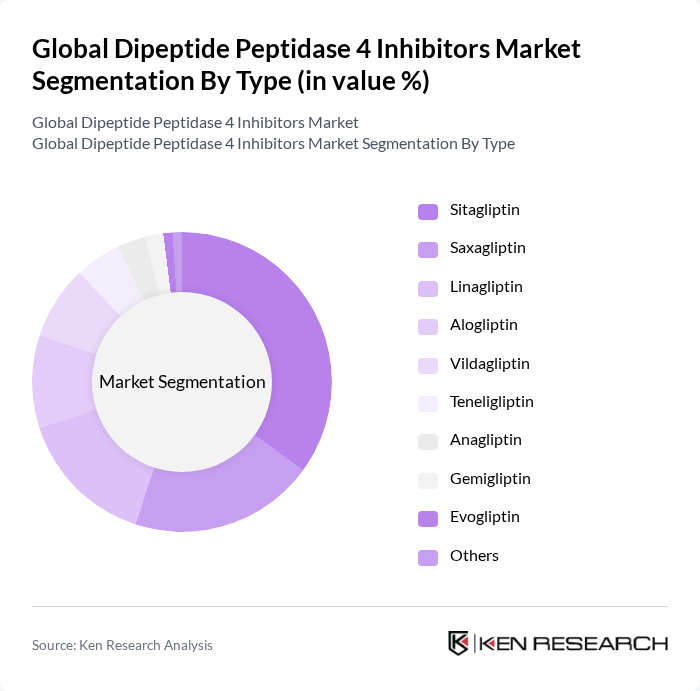
By Type:The market is segmented into various types of Dipeptide Peptidase 4 inhibitors, including Sitagliptin, Saxagliptin, Linagliptin, Alogliptin, Vildagliptin, Teneligliptin, Anagliptin, Gemigliptin, Evogliptin, and Others. Among these, Sitagliptin has emerged as the leading sub-segment due to its established efficacy and widespread acceptance among healthcare providers. The increasing number of prescriptions and favorable clinical outcomes associated with Sitagliptin contribute to its dominance in the market.
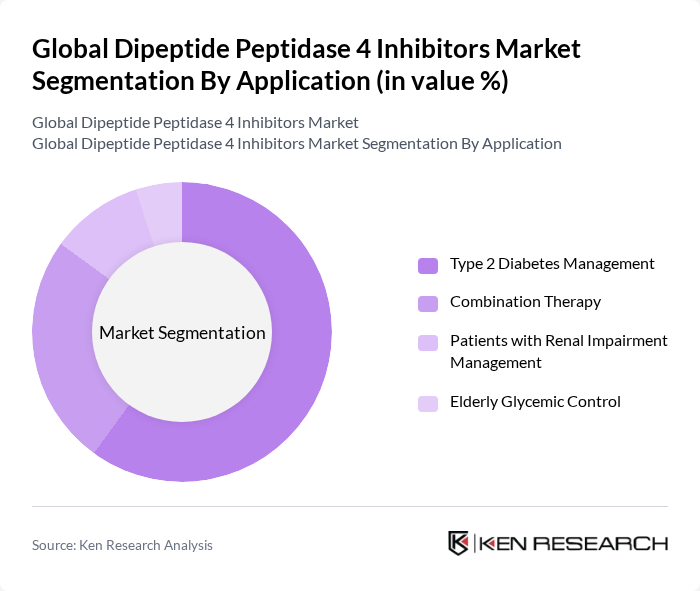
By Application:The applications of Dipeptide Peptidase 4 inhibitors include Type 2 Diabetes Management, Combination Therapy (e.g., with Metformin, SGLT2 inhibitors), Patients with Renal Impairment Management, and Elderly Glycemic Control. The Type 2 Diabetes Management segment is the most significant, driven by the increasing incidence of diabetes globally. The growing focus on personalized medicine and combination therapies further enhances the demand for these inhibitors in managing diabetes effectively.
The Global Dipeptide Peptidase 4 Inhibitors Market is characterized by a dynamic mix of regional and international players. Leading participants such as Merck & Co., Inc. (Januvia/Sitagliptin), AstraZeneca PLC (Onglyza/Saxagliptin; Kombiglyze XR), Boehringer Ingelheim GmbH (Tradjenta/Linagliptin; Jentadueto), Takeda Pharmaceutical Company Limited (Nesina/Alogliptin; Kazano; Oseni), Novartis AG (Galvus/Vildagliptin; Eucreas), Eli Lilly and Company (co-developer/co-marketer with Boehringer Ingelheim), Bristol Myers Squibb (historic co-developer of Saxagliptin), Sun Pharmaceutical Industries Ltd. (generics incl. Teneligliptin), Lupin Limited (generic DPP-4 portfolio), Glenmark Pharmaceuticals Ltd. (Teneligliptin; combinations), Teva Pharmaceutical Industries Ltd. (global generics), Sandoz Group AG (generics and biosimilars), Dr. Reddy’s Laboratories Ltd. (generic DPP-4s), Daiichi Sankyo Company, Limited (select APAC markets), Astellas Pharma Inc. (regional partnerships/licensing) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the dipeptide peptidase 4 inhibitors market appears promising, driven by increasing healthcare investments and a growing focus on personalized medicine. As the prevalence of diabetes continues to rise, pharmaceutical companies are likely to prioritize the development of innovative therapies tailored to individual patient needs. Additionally, the integration of digital health solutions and telemedicine is expected to enhance patient engagement and adherence, further supporting market growth in the None region.
| Segment | Sub-Segments |
|---|---|
| By Type | Sitagliptin Saxagliptin Linagliptin Alogliptin Vildagliptin Teneligliptin Anagliptin Gemigliptin Evogliptin Others |
| By Application | Type 2 Diabetes Management Combination Therapy (e.g., with Metformin, SGLT2 inhibitors) Patients with Renal Impairment Management Elderly Glycemic Control |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
| By End-User | Hospitals Clinics Homecare Settings Ambulatory Surgical Centers |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Patient Demographics | Adults Elderly Pediatric (off-label/rare use) |
| By Pricing Tier | Branded Generic |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Endocrinologists | 100 | Diabetes Specialists, Clinical Researchers |
| Pharmaceutical Sales Representatives | 80 | Sales Managers, Product Specialists |
| Healthcare Providers | 120 | General Practitioners, Nurse Practitioners |
| Patients on Dipeptide Peptidase 4 Inhibitors | 140 | Diabetes Patients, Caregivers |
| Pharmacy Managers | 70 | Pharmacists, Inventory Managers |
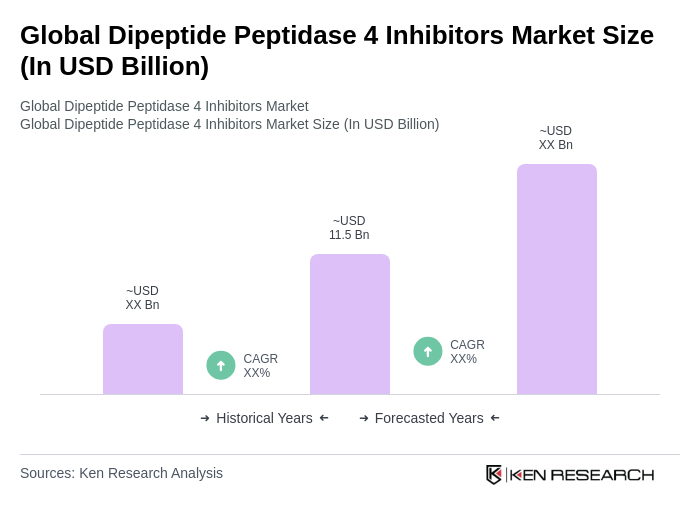
The Global Dipeptide Peptidase 4 Inhibitors Market is valued at approximately USD 11.5 billion, driven by the increasing prevalence of type 2 diabetes and the demand for effective glycemic control therapies.