Region:Global
Author(s):Shubham
Product Code:KRAC0608
Pages:96
Published On:August 2025
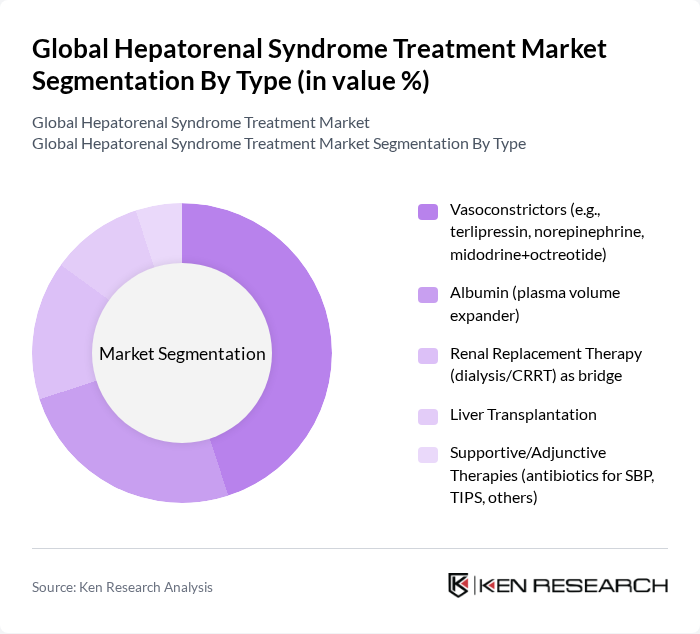
By Type:The treatment market is segmented into vasoconstrictors, albumin, renal replacement therapy, liver transplantation, and supportive therapies. Among these, vasoconstrictors—particularly terlipressin—with concomitant albumin are a mainstay for HRS-AKI and are widely recommended in guidelines and supported by randomized evidence for improving renal function; norepinephrine is commonly used in ICU settings, while midodrine plus octreotide is used where other options are limited . The increasing adoption in clinical practice reflects strengthened evidence, clearer diagnostic criteria for HRS-AKI, and broader availability of albumin and ICU support modalities .
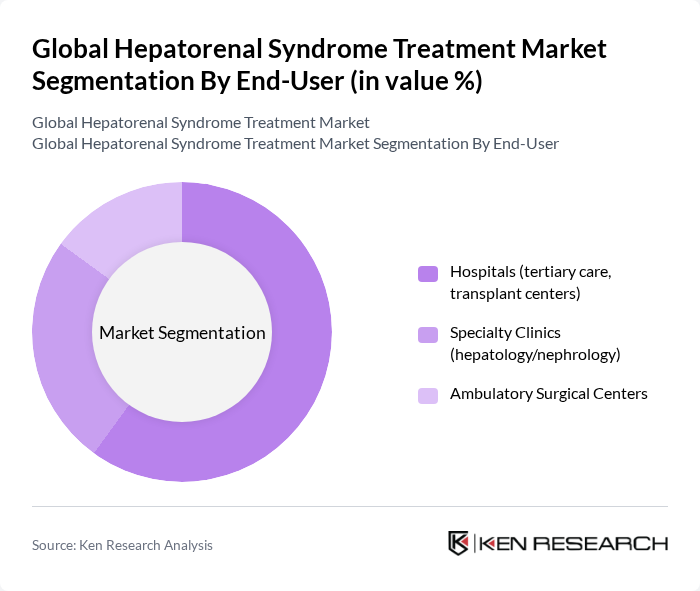
By End-User:The market is segmented by end-users into hospitals, specialty clinics, and ambulatory surgical centers. Hospitals—especially tertiary and transplant centers—dominate utilization because HRS management requires ICU-level monitoring, vasoactive infusions, albumin administration, and access to RRT and transplant evaluation pathways; specialty hepatology and nephrology clinics support diagnosis and post-acute optimization .
The Global Hepatorenal Syndrome Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Mallinckrodt Pharmaceuticals (terlipressin/Terlivaz for HRS-AKI in the United States), Grifols, S.A. (albumin/plasma products for HRS management), CSL Behring (CSL Limited) — albumin and plasma therapies, Baxter International Inc. (renal replacement therapy and CRRT systems), Fresenius Medical Care AG & Co. KGaA (dialysis/CRRT solutions), Terumo Corporation (blood management and apheresis/CRRT technologies), Nikkiso Co., Ltd. (dialysis/CRRT equipment), Ferring Pharmaceuticals (terlipressin across multiple ex-US markets), Pharmazz, Inc. (sovateltide PMZ-2010 program in ischemic conditions with AKI research relevance), BioVie Inc. (BIV201 terlipressin program history), Octapharma AG (human albumin solutions), Takeda Pharmaceutical Company Limited (liver disease and transplant support), AbbVie Inc. (advanced liver disease ecosystem), Gilead Sciences, Inc. (liver disease portfolio), and Medtronic plc (ICU infusion and monitoring) contribute to innovation, availability, and clinical practice alignment in this space .
The future of the Hepatorenal Syndrome treatment market in future appears promising, driven by ongoing advancements in medical research and technology. The integration of personalized medicine is expected to enhance treatment efficacy, while increased investment in R&D will likely yield innovative therapies. Additionally, the expansion of telemedicine services will improve patient access to specialists, facilitating timely interventions. These trends indicate a dynamic market landscape poised for growth, with a focus on improving patient outcomes and accessibility.
| Segment | Sub-Segments |
|---|---|
| By Type | Vasoconstrictors (e.g., terlipressin, norepinephrine, midodrine+octreotide) Albumin (plasma volume expander) Renal Replacement Therapy (dialysis/CRRT) as bridge Liver Transplantation Supportive/Adjunctive Therapies (antibiotics for SBP, TIPS, others) |
| By End-User | Hospitals (tertiary care, transplant centers) Specialty Clinics (hepatology/nephrology) Ambulatory Surgical Centers |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Patient Demographics | Age Group (Adults, Elderly) Gender (Male, Female) |
| By Treatment Setting | Inpatient (ICU/ward) Outpatient (clinic/infusion) |
| By Pricing Tier | Premium (innovator/biologics, ICU-based care) Mid-range (generics, standard of care) Economy (low-cost generics, LMIC procurement) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Nephrology Clinics | 120 | Nephrologists, Nurse Practitioners |
| Hepatology Departments | 90 | Hepatologists, Clinical Researchers |
| Pharmaceutical Sales Representatives | 70 | Sales Managers, Product Specialists |
| Patient Advocacy Groups | 60 | Patient Advocates, Community Health Workers |
| Healthcare Policy Makers | 50 | Health Economists, Policy Analysts |
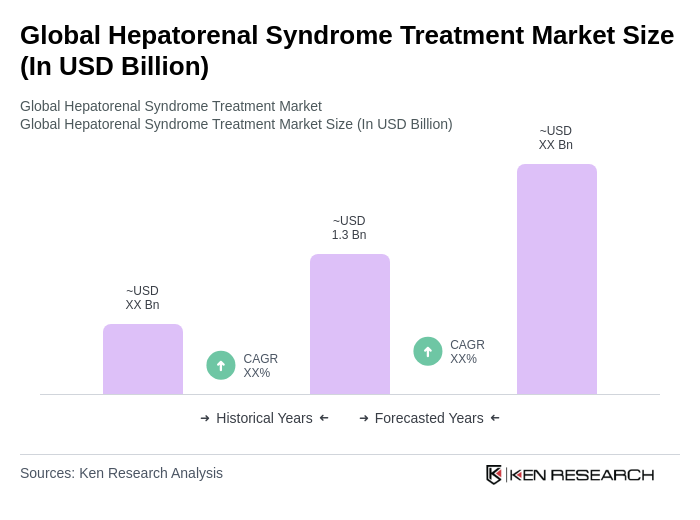
The Global Hepatorenal Syndrome Treatment Market is valued at approximately USD 1.3 billion, based on a five-year historical analysis, with recent reports indicating it is in the low-to-mid USD 1 billion range for the latest reporting year.