About the Report
Base Year 2024Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Overview
- The Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market is valued at USD 27 billion, based on a five-year historical analysis. This growth is primarily driven by the rising demand for targeted and personalized therapies, particularly in oncology and antibody-drug conjugates (ADCs). Additionally, significant investments in research and development within the biopharmaceutical sector are propelling market expansion.
- Key regions dominating the market include North America, which benefits from a robust manufacturing infrastructure and pharmaceutical leadership, and the rapidly growing Asia-Pacific region, particularly India and China, where cost-effective contract development and manufacturing organizations (CDMOs) are expanding. These regions are pivotal in meeting the increasing global demand for HPAPIs.
- In 2024, the U.S. government initiated the Strategic Active Pharmaceutical Ingredients Reserve (SAPIR) program, supported by an executive order aimed at enhancing domestic HPAPI availability. This initiative seeks to bolster supply chain resilience and reduce reliance on foreign sources, thereby ensuring a stable supply of critical pharmaceutical ingredients.
 Market.png)
Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Segmentation
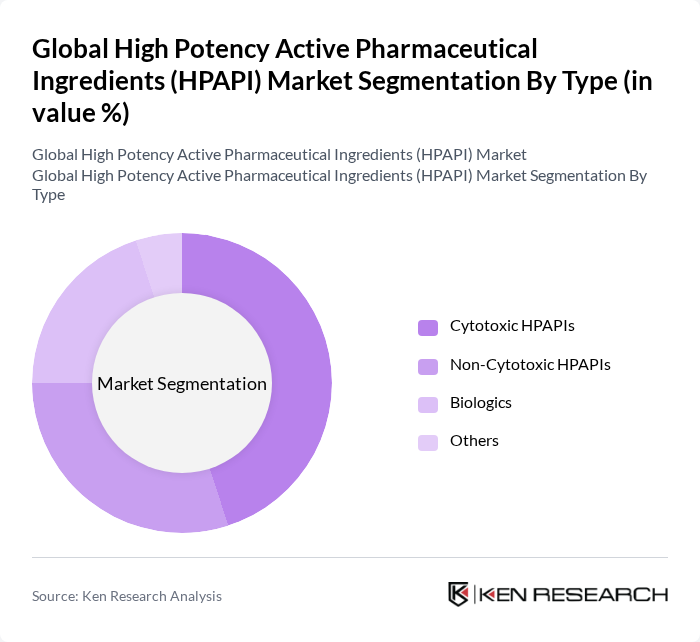
By Type:The market is segmented into various types of HPAPIs, including Cytotoxic HPAPIs, Non-Cytotoxic HPAPIs, Biologics, and Others. Cytotoxic HPAPIs are particularly dominant due to their essential role in cancer therapies, which are increasingly in demand as the prevalence of cancer rises globally. Non-Cytotoxic HPAPIs are also gaining traction, especially in the development of targeted therapies that minimize side effects. Biologics are becoming increasingly significant as they offer innovative treatment options for various diseases.
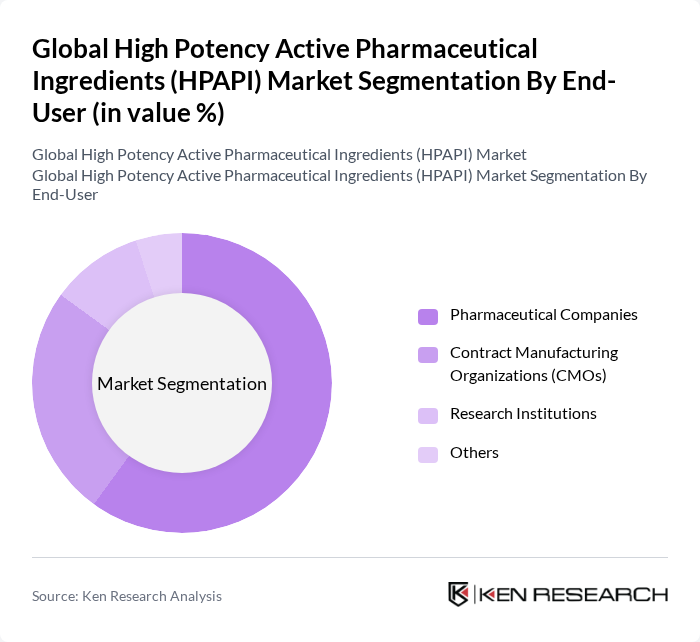
By End-User:The HPAPI market is also categorized by end-users, which include Pharmaceutical Companies, Contract Manufacturing Organizations (CMOs), Research Institutions, and Others. Pharmaceutical companies are the leading end-users, driven by their need for high-quality APIs to develop innovative therapies. CMOs are increasingly important as they provide specialized manufacturing services, allowing pharmaceutical companies to focus on research and development while outsourcing production. Research institutions are also significant as they contribute to the development of new drugs and therapies.
Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Competitive Landscape
The Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group, Pfizer Inc., Teva Pharmaceutical Industries Ltd., AbbVie Inc., Merck KGaA, Boehringer Ingelheim, Aenova Group, Cambrex Corporation, Hovione, WuXi AppTec, Sandoz (Novartis), Evonik Industries AG, Famar Health, Recipharm AB, Siegfried AG contribute to innovation, geographic expansion, and service delivery in this space.
Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Industry Analysis
Growth Drivers
- Rising Oncology Demand and Cancer Prevalence:The HPAPI market value is projected to reach USD 27.1 billion in future, with oncology applications contributing USD 15.2 billion. The increasing global incidence of cancer, which is expected to affect 19.3 million people in future, drives the demand for targeted HPAPI therapies, particularly antibody-drug conjugates that require high-potency compounds. This trend underscores the critical role of HPAPIs in modern oncology treatments.
- Dominance of Synthetic HPAPIs:In future, synthetic HPAPIs are anticipated to generate USD 18 billion, reflecting their significant market share. The preference for synthetic routes stems from their scalability and efficiency, allowing manufacturers to meet the growing therapeutic demand effectively. This trend is supported by advancements in synthetic chemistry, which enhance the production capabilities of high-potency compounds, ensuring timely availability for critical therapies.
- Adoption of Outsourced Manufacturing (CMOs/CDMOs):Outsourced HPAPI production is expected to account for USD 18.5 billion in future. The high costs associated with establishing containment facilities compel pharmaceutical companies to outsource production to specialized contract manufacturing organizations (CMOs). This strategy not only reduces capital expenditure but also ensures compliance with stringent safety regulations, thereby enhancing operational efficiency and focus on core competencies.
Market Challenges
- High Capital and Operational Costs:Establishing containment facilities for HPAPI production can require investments ranging from USD 50 million to over USD 100 million. These substantial capital requirements pose significant barriers to entry, particularly for smaller firms lacking the financial resources. The ongoing operational costs associated with maintaining compliance and safety standards further strain budgets, limiting market participation.
- Stringent Regulatory and Safety Compliance:The HPAPI sector faces complex regulatory demands from agencies like the FDA and EMA, which require rigorous safety and quality standards. Compliance involves extensive documentation, regular inspections, and continuous validation processes, contributing to increased operational burdens. These regulatory challenges can delay product development timelines and increase costs, impacting overall market agility and competitiveness.
Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Future Outlook
The future of the HPAPI market is poised for significant transformation, driven by advancements in manufacturing technologies and increasing demand for personalized medicine. The integration of continuous manufacturing processes and advanced containment systems will enhance production efficiency and safety. Additionally, the expansion of contract development and manufacturing organizations (CDMOs) will facilitate greater access to HPAPI capabilities, enabling pharmaceutical companies to innovate and respond to market needs more effectively.
Market Opportunities
- Growth through CDMO Partnerships:The outsourced HPAPI revenues are projected to reach USD 18.5 billion in future, with the CDMO sector expected to grow to EUR 293 billion in future. This growth presents pharmaceutical firms with opportunities to leverage CDMOs for advanced manufacturing capabilities, overcoming entry barriers and enhancing production efficiency.
- Regional Manufacturing Expansion (Asia-Pacific, India):India's pharmaceutical sector is projected to reach USD 65 billion in future, with the country holding 57% of the WHO prequalified API list. This cost-effective manufacturing environment, coupled with rising regional capacity, creates significant export and production opportunities, allowing companies to diversify their supply chains and reduce dependency on traditional markets.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Cytotoxic HPAPIs Non-Cytotoxic HPAPIs Biologics Others |
| By End-User | Pharmaceutical Companies Contract Manufacturing Organizations (CMOs) Research Institutions Others |
| By Application | Oncology Cardiovascular Neurology Others |
| By Delivery Method | Injectable Oral Transdermal Others |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Manufacturing Process | Chemical Synthesis Fermentation Others |
| By Regulatory Compliance | cGMP Compliance FDA Approval EMA Approval Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers
Pharmaceutical Companies
Contract Research Organizations (CROs)
Contract Manufacturing Organizations (CMOs)
Industry Associations (e.g., International Pharmaceutical Excipients Council)
Financial Institutions
Players Mentioned in the Report:
Lonza Group
Pfizer Inc.
Teva Pharmaceutical Industries Ltd.
AbbVie Inc.
Merck KGaA
Boehringer Ingelheim
Aenova Group
Cambrex Corporation
Hovione
WuXi AppTec
Sandoz (Novartis)
Evonik Industries AG
Famar Health
Recipharm AB
Siegfried AG
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Targeted Therapies
3.1.2 Advancements in Drug Development Technologies
3.1.3 Rising Prevalence of Chronic Diseases
3.1.4 Growing Investment in Biopharmaceuticals
3.2 Market Challenges
3.2.1 Stringent Regulatory Requirements
3.2.2 High Production Costs
3.2.3 Limited Availability of Skilled Workforce
3.2.4 Risk of Contamination and Safety Issues
3.3 Market Opportunities
3.3.1 Expansion in Emerging Markets
3.3.2 Development of Novel Drug Formulations
3.3.3 Collaborations and Partnerships
3.3.4 Increasing Focus on Personalized Medicine
3.4 Market Trends
3.4.1 Shift Towards Outsourcing HPAPI Production
3.4.2 Integration of AI in Drug Development
3.4.3 Growing Popularity of Contract Manufacturing Organizations (CMOs)
3.4.4 Emphasis on Sustainable Manufacturing Practices
3.5 Government Regulation
3.5.1 FDA Guidelines for HPAPI Production
3.5.2 EMA Regulations on Drug Safety
3.5.3 WHO Standards for Pharmaceutical Manufacturing
3.5.4 Environmental Regulations Impacting Production
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Segmentation
8.1 By Type
8.1.1 Cytotoxic HPAPIs
8.1.2 Non-Cytotoxic HPAPIs
8.1.3 Biologics
8.1.4 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Contract Manufacturing Organizations (CMOs)
8.2.3 Research Institutions
8.2.4 Others
8.3 By Application
8.3.1 Oncology
8.3.2 Cardiovascular
8.3.3 Neurology
8.3.4 Others
8.4 By Delivery Method
8.4.1 Injectable
8.4.2 Oral
8.4.3 Transdermal
8.4.4 Others
8.5 By Geography
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.6 By Manufacturing Process
8.6.1 Chemical Synthesis
8.6.2 Fermentation
8.6.3 Others
8.7 By Regulatory Compliance
8.7.1 cGMP Compliance
8.7.2 FDA Approval
8.7.3 EMA Approval
8.7.4 Others
9. Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Competitive Analysis
9.1 Market Share of Key Players(Micro, Small, Medium, Large Enterprises)
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Diversification
9.2.8 Supply Chain Efficiency
9.2.9 R&D Investment
9.2.10 Regulatory Compliance Rate
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis(By Class and Payload)
9.5 Detailed Profile of Major Companies
9.5.1 Lonza Group
9.5.2 Pfizer Inc.
9.5.3 Teva Pharmaceutical Industries Ltd.
9.5.4 AbbVie Inc.
9.5.5 Merck KGaA
9.5.6 Boehringer Ingelheim
9.5.7 Aenova Group
9.5.8 Cambrex Corporation
9.5.9 Hovione
9.5.10 WuXi AppTec
9.5.11 Sandoz (Novartis)
9.5.12 Evonik Industries AG
9.5.13 Famar Health
9.5.14 Recipharm AB
9.5.15 Siegfried AG
10. Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Procurement Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contract Management Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in HPAPI
10.2.2 Infrastructure Development Needs
10.2.3 Energy Consumption Patterns
10.2.4 Sustainability Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Quality Assurance Challenges
10.3.2 Supply Chain Disruptions
10.3.3 Cost Management Issues
10.3.4 Regulatory Compliance Difficulties
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Adoption Barriers
10.4.3 User Feedback Mechanisms
10.4.4 Change Management Strategies
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Satisfaction Levels
10.5.3 Scalability of Solutions
10.5.4 Future Investment Plans
11. Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
2.6 Customer Engagement Tactics
2.7 Performance Measurement
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 E-commerce Integration
3.4 Logistics and Supply Chain Management
3.5 Distribution Partnerships
3.6 Inventory Management
3.7 Customer Service Framework
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitive Pricing Strategies
4.4 Value-Based Pricing Models
4.5 Discount and Promotion Strategies
4.6 Price Sensitivity Assessment
4.7 Pricing Optimization Techniques
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Product Development Opportunities
5.4 Market Entry Strategies
5.5 Customer Feedback Utilization
5.6 Innovation in Product Offerings
5.7 Future Trends Forecasting
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Relationship Management Strategies
6.5 Customer Retention Techniques
6.6 Engagement through Social Media
6.7 Personalization of Services
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Cost-Effectiveness
7.4 Quality Assurance
7.5 Innovation in Offerings
7.6 Customer-Centric Approach
7.7 Competitive Advantage
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Market Research Activities
8.5 Training and Development
8.6 Performance Monitoring
8.7 Stakeholder Engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Identification
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from pharmaceutical associations and market research firms
- Review of scientific publications and patents related to HPAPIs
- Examination of regulatory guidelines from global health authorities (FDA, EMA, etc.)
Primary Research
- Interviews with R&D heads at leading pharmaceutical companies
- Surveys targeting quality assurance managers in HPAPI manufacturing
- Field interviews with regulatory affairs specialists in the pharmaceutical sector
Validation & Triangulation
- Cross-validation of data from multiple industry sources and expert opinions
- Triangulation of market size estimates using production, consumption, and import/export data
- Sanity checks through feedback from industry panels and focus groups
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of global HPAPI market size based on pharmaceutical sales data
- Segmentation by therapeutic area and geographical region
- Incorporation of trends in biopharmaceuticals and personalized medicine
Bottom-up Modeling
- Volume estimates based on production capacities of key HPAPI manufacturers
- Cost analysis derived from pricing models of active pharmaceutical ingredients
- Market share calculations based on sales data from major players
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical growth rates and market drivers
- Scenario analysis based on potential regulatory changes and market dynamics
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing | 150 | Production Managers, Quality Control Analysts |
| Regulatory Affairs | 100 | Regulatory Affairs Managers, Compliance Officers |
| Research & Development | 120 | R&D Directors, Formulation Scientists |
| Market Access & Pricing | 80 | Market Access Managers, Pricing Analysts |
| Supply Chain Management | 90 | Supply Chain Directors, Procurement Managers |
Frequently Asked Questions
What is the current value of the Global High Potency Active Pharmaceutical Ingredients (HPAPI) Market?
The Global HPAPI Market is valued at approximately USD 27 billion, driven by the increasing demand for targeted therapies, particularly in oncology and antibody-drug conjugates (ADCs), alongside significant investments in biopharmaceutical research and development.