Region:Global
Author(s):Dev
Product Code:KRAD0539
Pages:88
Published On:August 2025
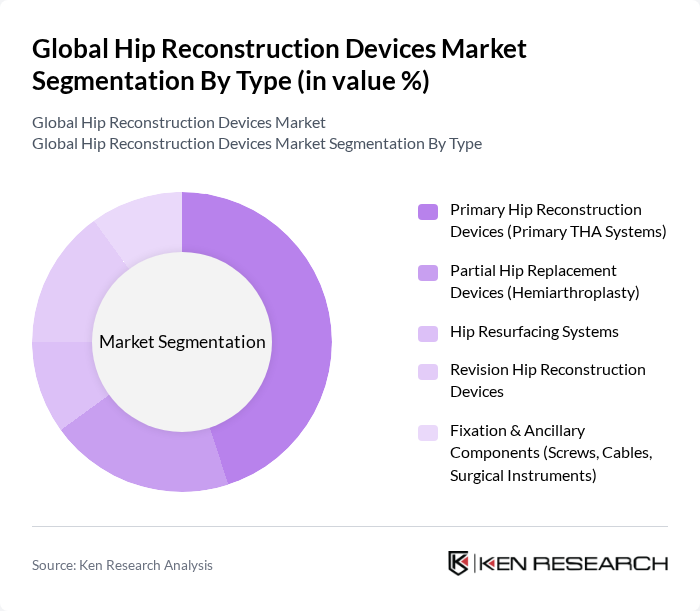
By Type:The market is segmented into various types of hip reconstruction devices, including Primary Hip Reconstruction Devices, Partial Hip Replacement Devices, Hip Resurfacing Systems, Revision Hip Reconstruction Devices, and Fixation & Ancillary Components. Among these, Primary Hip Reconstruction Devices are the most widely used due to their effectiveness in treating severe hip joint damage and their established track record in clinical outcomes.
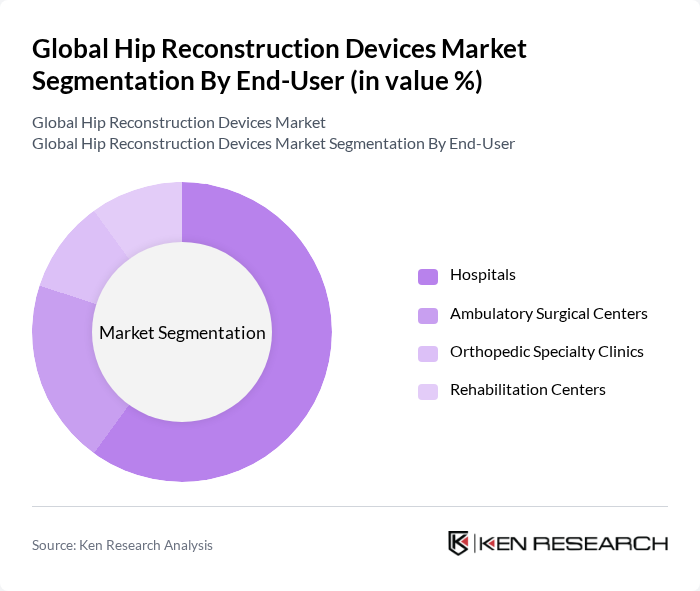
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Orthopedic Specialty Clinics, and Rehabilitation Centers. Hospitals dominate this segment due to their comprehensive facilities and the ability to perform complex surgeries, which require advanced hip reconstruction devices. The increasing number of orthopedic procedures performed in hospitals further solidifies their leading position in the market.
The Global Hip Reconstruction Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Johnson & Johnson MedTech (DePuy Synthes), Stryker Corporation, Zimmer Biomet Holdings, Inc., Smith & Nephew plc, B. Braun Melsungen AG (Aesculap), Exactech, Inc., Medacta Group SA, LimaCorporate S.p.A., DJO, LLC (Enovis Corporation), Corin Group, Waldemar Link GmbH & Co. KG (LINK), MicroPort Orthopedics Inc., ConMed Corporation, Orthofix Medical Inc., Heraeus Medical GmbH contribute to innovation, geographic expansion, and service delivery in this space.
The future of the hip reconstruction devices market appears promising, driven by ongoing technological advancements and an increasing focus on patient-centric care. As healthcare systems evolve, the integration of digital technologies and personalized medicine will likely enhance surgical outcomes. Furthermore, the rise in outpatient surgeries is expected to streamline procedures, making them more accessible. These trends indicate a dynamic market landscape, with opportunities for innovation and growth in the future.
| Segment | Sub-Segments |
|---|---|
| By Type | Primary Hip Reconstruction Devices (Primary THA Systems) Partial Hip Replacement Devices (Hemiarthroplasty) Hip Resurfacing Systems Revision Hip Reconstruction Devices Fixation & Ancillary Components (Screws, Cables, Surgical Instruments) |
| By End-User | Hospitals Ambulatory Surgical Centers Orthopedic Specialty Clinics Rehabilitation Centers |
| By Material/Bearing Surface | Metal-on-Polyethylene (MoP) Ceramic-on-Polyethylene (CoP) Ceramic-on-Ceramic (CoC) Advanced Polyethylene Inserts (Highly Cross-Linked/Antioxidant) |
| By Fixation Technique | Cementless Cemented Hybrid |
| By Distribution Channel | Direct Sales Distributors Group Purchasing Organizations (GPOs) and Integrated Delivery Networks (IDNs) |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Patient Demographics | Age Group (Elderly, Adults) Gender (Male, Female) Activity Level (Active, Sedentary) |
| By Surgical Approach | Anterior Approach Posterior Approach Lateral Approach Robotic-Assisted and Navigation-Enabled |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Orthopedic Surgeons | 120 | Surgeons specializing in hip procedures, Clinical Directors |
| Hospital Procurement Managers | 90 | Procurement Officers, Supply Chain Managers |
| Medical Device Sales Representatives | 80 | Sales Managers, Territory Representatives |
| Healthcare Policy Makers | 60 | Health Economists, Policy Analysts |
| Patients undergoing Hip Reconstruction | 70 | Patients, Caregivers, Rehabilitation Specialists |
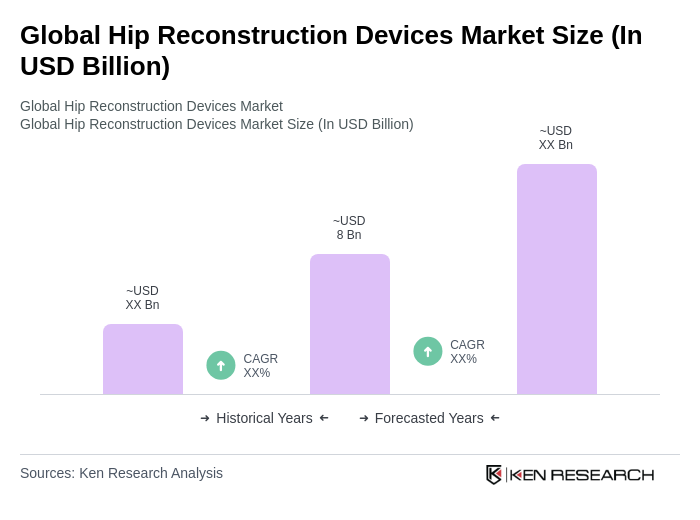
The Global Hip Reconstruction Devices Market is valued at approximately USD 8 billion, driven by factors such as the increasing prevalence of hip-related disorders, advancements in surgical techniques, and a growing geriatric population.