Region:Global
Author(s):Rebecca
Product Code:KRAB0228
Pages:83
Published On:August 2025
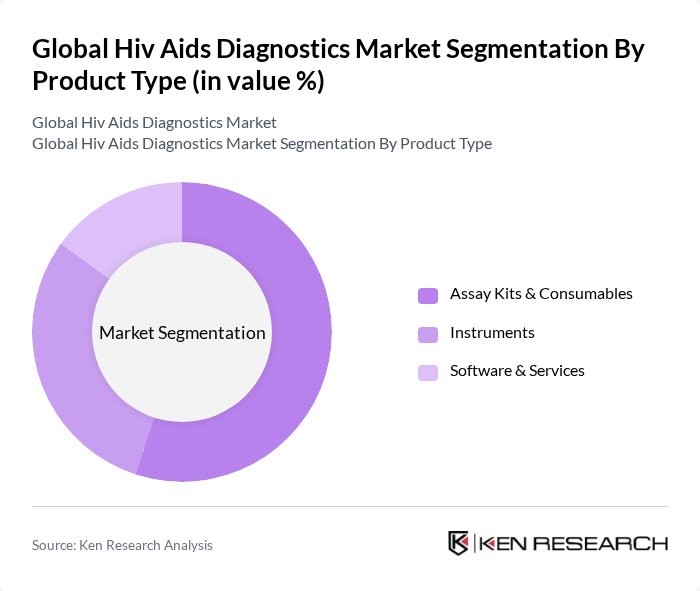
By Product Type:The product type segmentation includes Assay Kits & Consumables, Instruments, and Software & Services. Among these, Assay Kits & Consumables dominate the market due to their essential role in the testing process, with a growing demand for rapid and accurate testing solutions. The increasing number of testing facilities and the need for regular monitoring of HIV patients further bolster the growth of this segment.
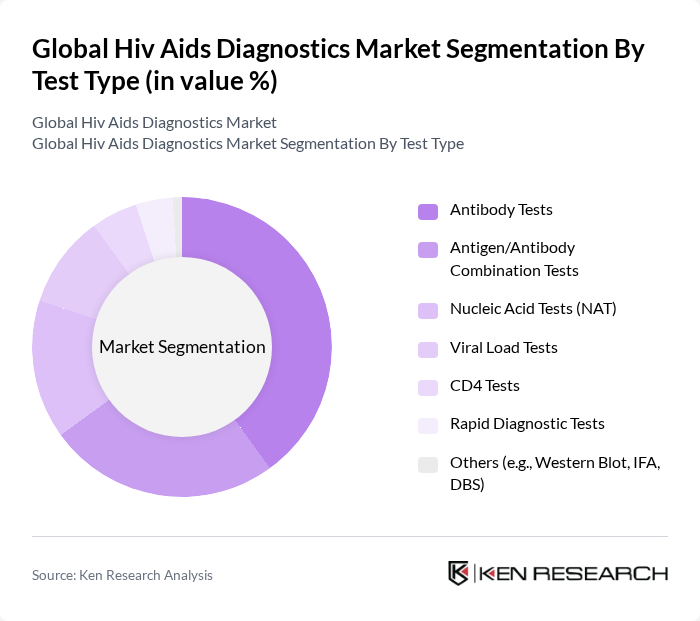
By Test Type:The test type segmentation encompasses Antibody Tests, Antigen/Antibody Combination Tests, Nucleic Acid Tests (NAT), Viral Load Tests, CD4 Tests, Rapid Diagnostic Tests, and Others. Antibody Tests are the leading sub-segment, primarily due to their widespread use in initial screening processes. The simplicity and cost-effectiveness of these tests make them a preferred choice in various healthcare settings, contributing significantly to their market dominance.
The Global Hiv Aids Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics (F. Hoffmann-La Roche Ltd.), Siemens Healthineers AG, Becton, Dickinson and Company, Bio-Rad Laboratories, Inc., Cepheid (Danaher Corporation), Hologic, Inc., OraSure Technologies, Inc., Grifols, S.A., QuidelOrtho Corporation, bioMérieux SA, Thermo Fisher Scientific Inc., Merck & Co., Inc., Sysmex Corporation, Danaher Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of HIV/AIDS diagnostics is poised for transformative growth, driven by technological advancements and increased public awareness. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the shift towards home-based testing will empower individuals to take charge of their health. Additionally, ongoing government funding and international collaborations will further support innovative solutions, ensuring that diagnostics remain accessible and effective in combating the HIV/AIDS epidemic.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Assay Kits & Consumables Instruments Software & Services |
| By Test Type | Antibody Tests Antigen/Antibody Combination Tests Nucleic Acid Tests (NAT) Viral Load Tests CD4 Tests Rapid Diagnostic Tests Others (e.g., Western Blot, IFA, DBS) |
| By End-User | Diagnostic Laboratories Hospitals & Clinics Blood Banks Home Care Settings NGOs and Community Health Organizations |
| By Distribution Channel | Direct Sales Online Sales Retail Pharmacies Hospitals and Clinics |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa GCC Countries |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Managers, Diagnostic Technicians |
| Healthcare Providers | 80 | Infectious Disease Specialists, General Practitioners |
| Public Health Organizations | 60 | Public Health Officials, Epidemiologists |
| Diagnostic Equipment Manufacturers | 50 | Product Development Managers, Sales Executives |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Community Health Workers |
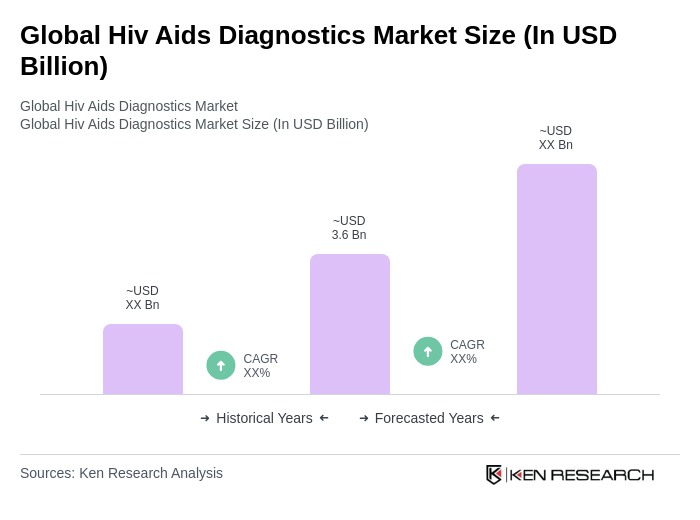
The Global HIV AIDS Diagnostics Market is valued at approximately USD 3.6 billion, reflecting a significant growth driven by the increasing prevalence of HIV infections and advancements in diagnostic technologies.