Region:Global
Author(s):Geetanshi
Product Code:KRAC0089
Pages:95
Published On:August 2025
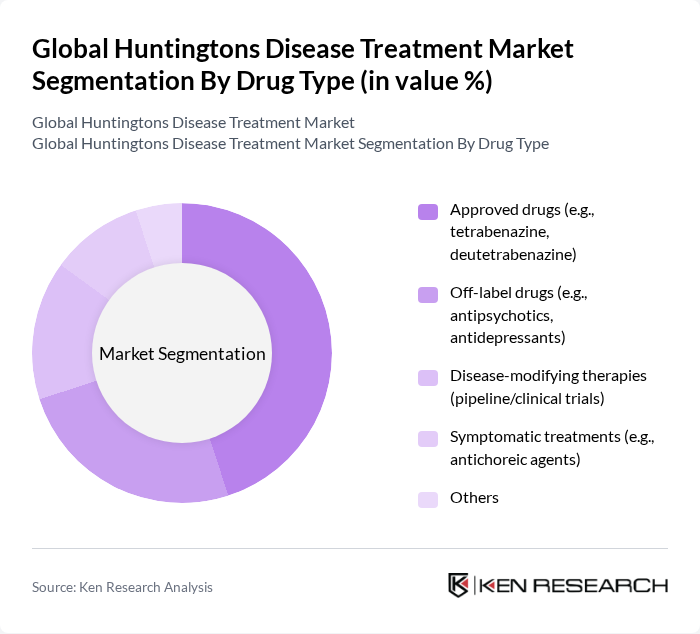
By Drug Type:The drug type segmentation includes various categories such as approved drugs, off-label drugs, disease-modifying therapies, symptomatic treatments, and others. Among these, approved drugs like tetrabenazine and deutetrabenazine are leading the market due to their established efficacy and safety profiles. Off-label drugs, including antipsychotics and antidepressants, are also widely used as they provide symptomatic relief. The pipeline for disease-modifying therapies is expanding, with increased focus on gene-targeting technologies and RNA-based treatments, indicating a promising future for innovative therapies .
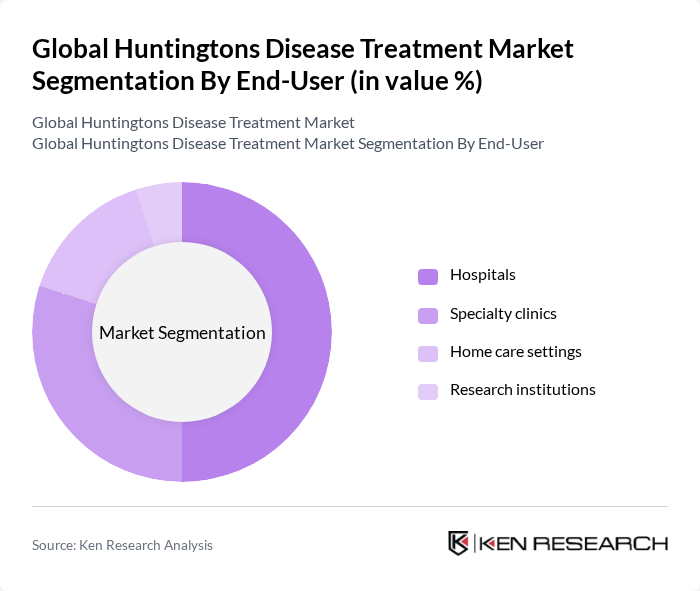
By End-User:The end-user segmentation encompasses hospitals, specialty clinics, home care settings, and research institutions. Hospitals are the dominant end-user segment, primarily due to their capacity to provide comprehensive care and access to advanced treatment options. Specialty clinics are significant as they focus on neurological disorders, offering tailored therapies. Home care settings are emerging as a viable option for patients seeking comfort and personalized care, while research institutions play a crucial role in developing new treatments .
The Global Huntingtons Disease Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Teva Pharmaceutical Industries Ltd., Roche Holding AG, Novartis AG, Pfizer Inc., Eli Lilly and Company, Sanofi S.A., AbbVie Inc., Amgen Inc., Biogen Inc., Merck & Co., Inc., H. Lundbeck A/S, Neurocrine Biosciences, Inc., Prilenia Therapeutics B.V., uniQure N.V., Ionis Pharmaceuticals, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of Huntington's Disease treatment is poised for transformation, driven by ongoing advancements in gene therapy and personalized medicine. As research progresses, innovative therapies tailored to individual genetic profiles are expected to emerge, enhancing treatment efficacy. Additionally, the expansion of digital health solutions will facilitate better patient monitoring and engagement, ultimately improving care delivery. These trends indicate a promising landscape for stakeholders in the Huntington's Disease treatment market, fostering collaboration and investment in research initiatives.
| Segment | Sub-Segments |
|---|---|
| By Drug Type | Approved drugs (e.g., tetrabenazine, deutetrabenazine) Off-label drugs (e.g., antipsychotics, antidepressants) Disease-modifying therapies (pipeline/clinical trials) Symptomatic treatments (e.g., antichoreic agents) Others |
| By End-User | Hospitals Specialty clinics Home care settings Research institutions |
| By Route of Administration | Oral Injectable Transdermal |
| By Distribution Channel | Hospital pharmacies Retail pharmacies Online pharmacies |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Patient Demographics | Age group (children, adults, elderly) Gender |
| By Treatment Stage | Early-stage treatment Mid-stage treatment Late-stage treatment |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurologists Specializing in Huntington's Disease | 100 | Neurologists, Clinical Researchers |
| Pharmaceutical Executives in Rare Disease Development | 70 | Product Managers, R&D Directors |
| Patient Advocacy Group Leaders | 50 | Advocacy Coordinators, Healthcare Policy Experts |
| Healthcare Payers and Insurers | 60 | Policy Analysts, Reimbursement Specialists |
| Caregivers of Huntington's Disease Patients | 40 | Family Caregivers, Support Group Leaders |
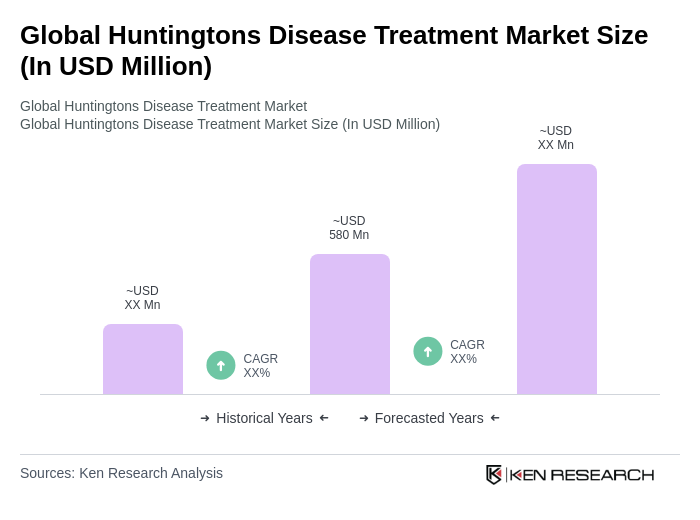
The Global Huntington's Disease Treatment Market is valued at approximately USD 580 million, driven by the increasing prevalence of the disease, advancements in drug development, and heightened awareness among healthcare professionals and patients.