Region:Global
Author(s):Geetanshi
Product Code:KRAA2338
Pages:91
Published On:August 2025
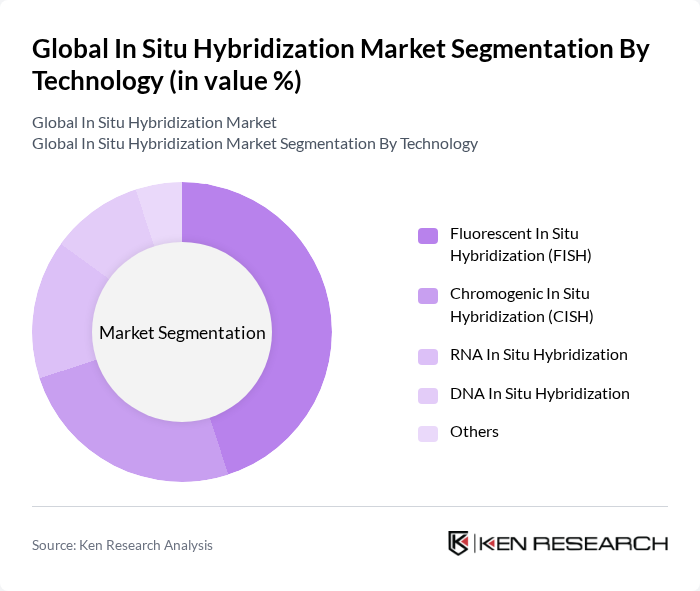
By Technology:The technology segment of the market includes various methods used for in situ hybridization, such as Fluorescent In Situ Hybridization (FISH), Chromogenic In Situ Hybridization (CISH), RNA In Situ Hybridization, DNA In Situ Hybridization, and others. Among these, FISH is the most widely adopted due to its high sensitivity and specificity, making it a preferred choice for cancer diagnostics and genetic research. The increasing focus on personalized medicine, the growing incidence of genetic disorders, and the need for precise cytogenetic analysis are further driving the demand for FISH technology .
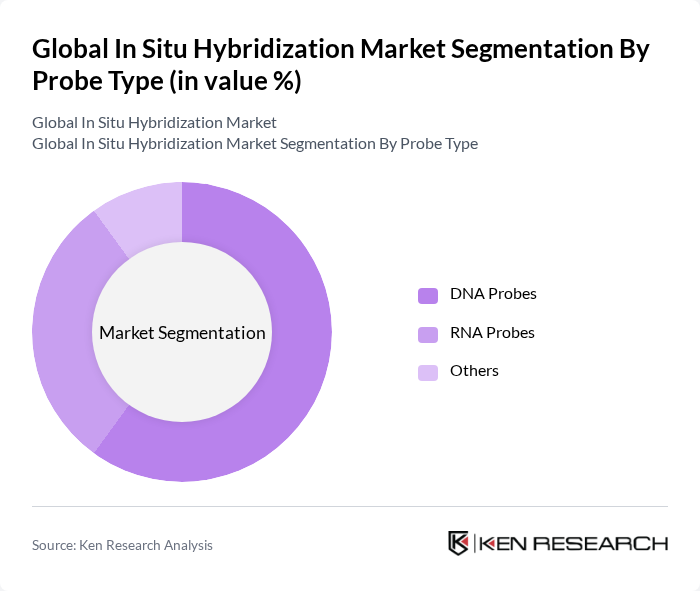
By Probe Type:The probe type segment encompasses DNA probes, RNA probes, and others. DNA probes dominate this segment due to their critical role in detecting specific genetic sequences, which is essential for cancer diagnostics and genetic research. The increasing prevalence of genetic disorders and the growing demand for targeted therapies are driving the adoption of DNA probes, making them a key component in the in situ hybridization market .
The Global In Situ Hybridization Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Agilent Technologies Inc., Roche Diagnostics, Bio-Techne Corporation, Merck KGaA, PerkinElmer Inc., Abcam plc, F. Hoffmann-La Roche Ltd., QIAGEN N.V., Bionano Genomics, Inc., Illumina, Inc., Bio-Rad Laboratories, Inc., Eppendorf AG, Sigma-Aldrich Corporation (now part of Merck KGaA), Advanced Cell Diagnostics, Inc. (a Bio-Techne brand), Leica Biosystems Nussloch GmbH, NeoGenomics Laboratories, Inc., BIOVIEW Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the in situ hybridization market appears promising, driven by technological advancements and increasing applications in cancer research and diagnostics. As healthcare systems prioritize precision medicine, the integration of automation and digital pathology is expected to enhance efficiency and accuracy in laboratory processes. Furthermore, collaborations between industry players and academic institutions will likely foster innovation, leading to the development of novel probes and reagents that can address unmet clinical needs.
| Segment | Sub-Segments |
|---|---|
| By Technology | Fluorescent In Situ Hybridization (FISH) Chromogenic In Situ Hybridization (CISH) RNA In Situ Hybridization DNA In Situ Hybridization Others |
| By Probe Type | DNA Probes RNA Probes Others |
| By Product Type | Instruments Kits & Probes Reagents Software Services Others |
| By Application | Cancer Diagnostics Cytogenetics Infectious Disease Diagnostics Developmental Biology Genetic Research Drug Development Others |
| By End-User | Hospitals Clinical Laboratories Research Laboratories Academic Institutions Pharmaceutical & Biotechnology Companies Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | North America Latin America Western Europe Eastern Europe Balkan & Baltic Countries Russia & Belarus Central Asia East Asia South Asia & Pacific Middle East & Africa |
| By Price Range | Low Medium High Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Diagnostics | 100 | Clinical Pathologists, Lab Directors |
| Research Laboratories | 70 | Research Scientists, Lab Managers |
| Biotechnology Firms | 50 | Product Managers, R&D Directors |
| Pharmaceutical Companies | 60 | Regulatory Affairs Specialists, Clinical Researchers |
| Academic Institutions | 40 | Professors, Graduate Researchers |
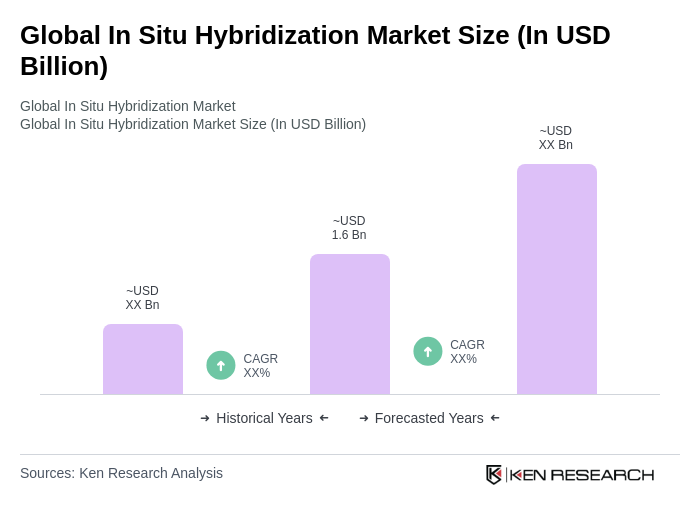
The Global In Situ Hybridization Market is valued at approximately USD 1.6 billion, driven by the increasing prevalence of cancer and genetic disorders, as well as advancements in diagnostic technologies and personalized medicine.