About the Report
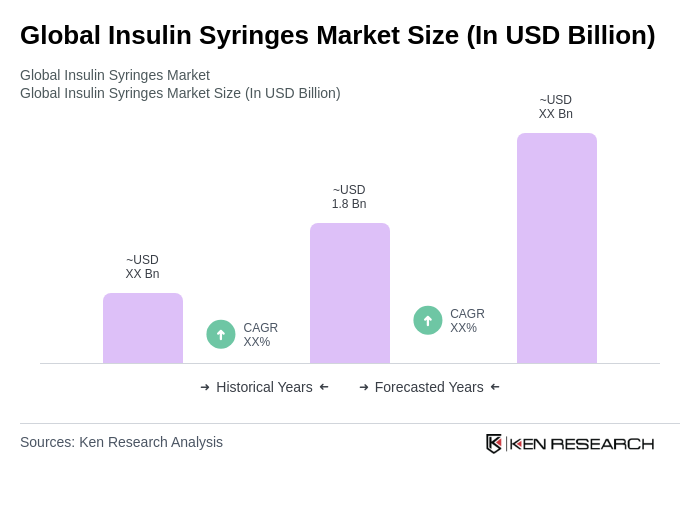
Base Year 2024Global Insulin Syringes Market Overview
- The Global Insulin Syringes Market is valued at USD 1.8 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of diabetes, rising awareness about diabetes management, and continued reliance on low-cost insulin delivery in low- and middle-income settings, where syringes remain the most affordable and widely accessible option. The market has seen steady demand due to the growing number of people with diabetes globally and the role of public procurement programs that prioritize cost-effective insulin administration methods.
- Key demand centers include the United States, Germany, and Japan, supported by advanced healthcare infrastructure and sustained investments in diabetes care; however, Asia-Pacific has become a prominent growth engine given the expanding diabetes population and supportive policies, especially in India. The presence of major medical device companies and strong R&D focus in these markets underpins their influence across the global insulin syringes landscape.
- In response to occupational safety risks, the U.S. Needlestick Safety and Prevention Act requires employers to use safety-engineered sharps where feasible and to update exposure control plans, which has increased adoption of injury-prevention syringe designs in healthcare settings. These measures aim to reduce needlestick injuries and enhance safety, reinforcing demand for safety-engineered insulin delivery options in clinical use.
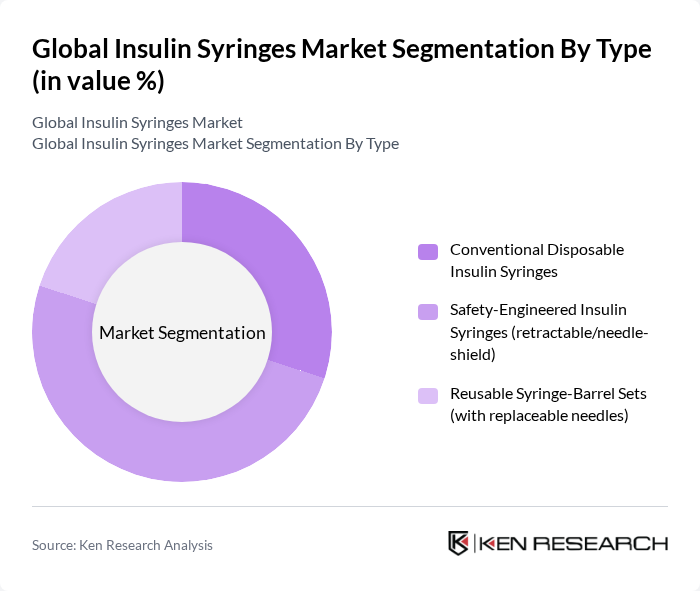
Global Insulin Syringes Market Segmentation
By Type:The market is segmented into three main types: Conventional Disposable Insulin Syringes, Safety-Engineered Insulin Syringes (retractable/needle-shield), and Reusable Syringe-Barrel Sets (with replaceable needles). Safety-Engineered Insulin Syringes are gaining traction because injury-prevention features lower needlestick risk and align with occupational safety requirements in hospitals and clinics. The growing emphasis on patient and worker safety and procurement preferences for safety-engineered devices are supporting this segment’s momentum.
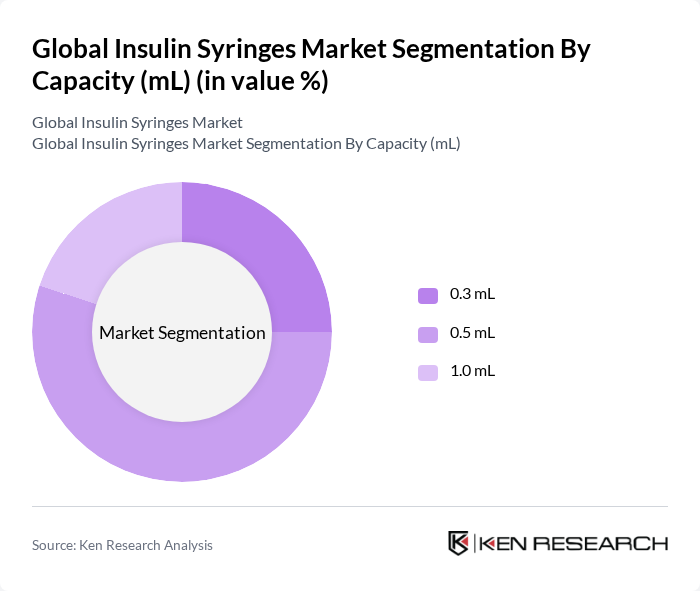
By Capacity (mL):The capacity segment includes 0.3 mL, 0.5 mL, and 1.0 mL syringes. The 0.5 mL capacity holds the leading share in recent industry assessments, reflecting its versatility for common insulin dosing ranges and broad use by patients and providers. Recent data indicate the 0.5 mL segment leads the market by share in the latest year assessed.
Global Insulin Syringes Market Competitive Landscape
The Global Insulin Syringes Market is characterized by a dynamic mix of regional and international players. Leading participants such as Becton, Dickinson and Company (BD), Terumo Corporation, Nipro Corporation, Hindustan Syringes & Medical Devices Ltd. (HMD), B. Braun Melsungen AG, Cardinal Health, Inc., Smiths Medical (ICU Medical, Inc.), Gerresheimer AG, ARKRAY, Inc., Ypsomed Holding AG, Owen Mumford Ltd., Poly Medicure Ltd., Nipro Medical Europe/North America (Nipro group), HTL?Strefa S.A. (Mylan/BD partnership history), UltiMed, Inc. (UltiCare) contribute to innovation, geographic expansion, and service delivery in this space.
Global Insulin Syringes Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Diabetes:The global diabetes prevalence is projected to reach643 million adultsin future, according to the International Diabetes Federation. This alarming rise, driven by lifestyle changes and aging populations, significantly boosts the demand for insulin syringes. In regions like None, where diabetes rates are climbing, healthcare systems are increasingly focused on providing effective management solutions, thereby enhancing the market for insulin delivery devices.
- Technological Advancements in Insulin Delivery:Innovations in insulin delivery systems, such as pre-filled syringes and smart insulin pens, are transforming diabetes management. The global market for smart insulin delivery devices is expected to reach$7.5 billionin future, driven by enhanced patient compliance and improved outcomes. In None, these advancements are crucial as they cater to the growing demand for user-friendly and efficient diabetes management solutions, thereby propelling market growth.
- Rising Awareness about Diabetes Management:Increased awareness campaigns and educational programs about diabetes management are leading to higher adoption rates of insulin syringes. In None, healthcare initiatives have resulted in a40%increase in diabetes screening and management programs since 2020. This heightened awareness is crucial for early diagnosis and treatment, driving the demand for insulin syringes as patients seek effective management tools to control their condition.
Market Challenges
- High Cost of Insulin Syringes:The cost of insulin syringes can be a significant barrier to access, particularly in regions with limited healthcare funding. In None, the average cost of a box of insulin syringes is approximately$60, which can be prohibitive for low-income patients. This financial burden can lead to non-compliance with diabetes management, ultimately affecting health outcomes and limiting market growth in the region.
- Regulatory Hurdles:The insulin syringe market faces stringent regulatory requirements that can delay product approvals and increase costs. In None, the regulatory framework mandates extensive clinical trials and safety assessments, which can take years to complete. These hurdles not only slow down the introduction of innovative products but also deter new entrants, creating a challenging environment for market expansion and innovation.
Global Insulin Syringes Market Future Outlook
The future of the insulin syringes market in None appears promising, driven by ongoing technological advancements and a growing emphasis on personalized healthcare solutions. As the prevalence of diabetes continues to rise, the demand for innovative insulin delivery methods will likely increase. Additionally, partnerships between manufacturers and healthcare providers are expected to enhance distribution channels, ensuring better access to insulin syringes. This collaborative approach will be vital in addressing the challenges posed by high costs and regulatory barriers, ultimately improving patient outcomes.
Market Opportunities
- Expansion in Emerging Markets:Emerging markets present significant growth opportunities for insulin syringes, with an estimated70%of the global diabetes population residing in these regions. In future, investments in healthcare infrastructure and increased access to diabetes management tools are expected to drive demand, making it a lucrative area for market players to explore.
- Development of Smart Insulin Syringes:The rise of digital health technologies offers a unique opportunity for the development of smart insulin syringes. These devices, equipped with features like dose tracking and connectivity to mobile apps, can enhance patient adherence and management. The anticipated growth in this segment could significantly impact the market, particularly in tech-savvy regions like None.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Conventional Disposable Insulin Syringes Safety-Engineered Insulin Syringes (retractable/needle-shield) Reusable Syringe-Barrel Sets (with replaceable needles) |
| By Capacity (mL) | mL mL mL |
| By Needle Gauge | G–30G G–32G |
| By Needle Length | –6 mm mm mm |
| By End-User | Hospitals & Clinics Homecare Settings Diabetes Centers & Pharmacies |
| By Distribution Channel | Retail Pharmacies Online Pharmacies & E-commerce Hospital/Institutional Procurement |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Diabetes Type | Type 1 Diabetes Type 2 Diabetes |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Pharmaceutical Companies
Health Insurance Companies
Medical Device Regulatory Authorities
Players Mentioned in the Report:
Becton, Dickinson and Company (BD)
Terumo Corporation
Nipro Corporation
Hindustan Syringes & Medical Devices Ltd. (HMD)
B. Braun Melsungen AG
Cardinal Health, Inc.
Smiths Medical (ICU Medical, Inc.)
Gerresheimer AG
ARKRAY, Inc.
Ypsomed Holding AG
Owen Mumford Ltd.
Poly Medicure Ltd.
Nipro Medical Europe/North America (Nipro group)
HTLStrefa S.A. (Mylan/BD partnership history)
UltiMed, Inc. (UltiCare)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Insulin Syringes Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Insulin Syringes Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Insulin Syringes Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of diabetes
3.1.2 Technological advancements in insulin delivery
3.1.3 Rising awareness about diabetes management
3.1.4 Growth in geriatric population
3.2 Market Challenges
3.2.1 High cost of insulin syringes
3.2.2 Regulatory hurdles
3.2.3 Competition from alternative delivery methods
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Development of smart insulin syringes
3.3.3 Partnerships with healthcare providers
3.3.4 Increasing focus on preventive healthcare
3.4 Market Trends
3.4.1 Shift towards home healthcare solutions
3.4.2 Integration of digital health technologies
3.4.3 Customization of insulin delivery devices
3.4.4 Sustainability in product manufacturing
3.5 Government Regulation
3.5.1 Compliance with FDA regulations
3.5.2 Guidelines for medical device safety
3.5.3 Policies promoting diabetes care
3.5.4 Reimbursement policies for insulin syringes
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Insulin Syringes Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Insulin Syringes Market Segmentation
8.1 By Type
8.1.1 Conventional Disposable Insulin Syringes
8.1.2 Safety-Engineered Insulin Syringes (retractable/needle-shield)
8.1.3 Reusable Syringe-Barrel Sets (with replaceable needles)
8.2 By Capacity (mL)
8.2.1 0.3 mL
8.2.2 0.5 mL
8.2.3 1.0 mL
8.3 By Needle Gauge
8.3.1 28G–30G
8.3.2 31G–32G
8.4 By Needle Length
8.4.1 4–6 mm
8.4.2 8 mm
8.4.3 12.7 mm
8.5 By End-User
8.5.1 Hospitals & Clinics
8.5.2 Homecare Settings
8.5.3 Diabetes Centers & Pharmacies
8.6 By Distribution Channel
8.6.1 Retail Pharmacies
8.6.2 Online Pharmacies & E-commerce
8.6.3 Hospital/Institutional Procurement
8.7 By Region
8.7.1 North America
8.7.2 Europe
8.7.3 Asia-Pacific
8.7.4 Latin America
8.7.5 Middle East & Africa
8.8 By Diabetes Type
8.8.1 Type 1 Diabetes
8.8.2 Type 2 Diabetes
9. Global Insulin Syringes Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name (Insulin Syringes Portfolio)
9.2.2 Group Size (Global, Regional, Niche)
9.2.3 Insulin Syringes Revenue (USD, latest FY or estimate)
9.2.4 Volume Throughput (annual syringe units)
9.2.5 Geographic Footprint (No. of countries/regions served)
9.2.6 Portfolio Breadth (gauges, lengths, capacities offered)
9.2.7 Safety-Engineered Mix (% of sales from safety syringes)
9.2.8 Institutional Tender Wins (No./value in last 12–24 months)
9.2.9 Average Selling Price (ASP) band
9.2.10 Regulatory & Quality Credentials (FDA 510(k), CE, ISO)
9.2.11 Manufacturing Capacity & Localization (plants, regions)
9.2.12 On-time Delivery/Fill Rate (%)
9.2.13 Complaint Rate/100k units and Sharps Injury Incidents
9.2.14 New Product Launches (past 24 months)
9.2.15 Sustainability & Waste Management Programs
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Becton, Dickinson and Company (BD)
9.5.2 Terumo Corporation
9.5.3 Nipro Corporation
9.5.4 Hindustan Syringes & Medical Devices Ltd. (HMD)
9.5.5 B. Braun Melsungen AG
9.5.6 Cardinal Health, Inc.
9.5.7 Smiths Medical (ICU Medical, Inc.)
9.5.8 Gerresheimer AG
9.5.9 ARKRAY, Inc.
9.5.10 Ypsomed Holding AG
9.5.11 Owen Mumford Ltd.
9.5.12 Poly Medicure Ltd.
9.5.13 Nipro Medical Europe/North America (Nipro group)
9.5.14 HTL?Strefa S.A. (Mylan/BD partnership history)
9.5.15 UltiMed, Inc. (UltiCare)
10. Global Insulin Syringes Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government procurement policies
10.1.2 Budget allocation for diabetes care
10.1.3 Collaboration with healthcare providers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare infrastructure
10.2.2 Funding for diabetes management programs
10.3 Pain Point Analysis by End-User Category
10.3.1 Accessibility of insulin syringes
10.3.2 Affordability issues
10.3.3 Quality concerns
10.4 User Readiness for Adoption
10.4.1 Awareness of insulin syringe options
10.4.2 Training and education needs
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost savings from effective diabetes management
10.5.2 Expansion into new markets
11. Global Insulin Syringes Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategy
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing comparison
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band strategy
9.1.3 Packaging solutions
9.2 Export Entry Strategy
9.2.1 Target countries analysis
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone tracking
15.2.2 Activity scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of global insulin syringe market reports from industry associations and health organizations
- Review of published articles and white papers on diabetes management and insulin delivery systems
- Examination of regulatory frameworks and guidelines from health authorities regarding insulin syringe usage
Primary Research
- Interviews with endocrinologists and diabetes care specialists to understand market needs
- Surveys with healthcare providers and pharmacists regarding insulin syringe preferences
- Field interviews with patients using insulin syringes to gather user experience insights
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert opinions
- Triangulation of data from healthcare professionals, manufacturers, and patient feedback
- Sanity checks through expert panel reviews to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total addressable market based on global diabetes prevalence statistics
- Segmentation of market size by geographic regions and insulin delivery methods
- Incorporation of trends in diabetes management and technological advancements in insulin delivery
Bottom-up Modeling
- Volume estimates based on sales data from leading insulin syringe manufacturers
- Cost analysis of insulin syringes including production, distribution, and retail pricing
- Calculation of market size based on projected growth rates in diabetes cases and treatment adoption
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as population growth and healthcare access
- Scenario modeling based on potential changes in healthcare policies and insurance coverage
- Baseline, optimistic, and pessimistic forecasts through 2030 based on market dynamics
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 150 | Endocrinologists, Diabetes Educators |
| Pharmaceutical Distributors | 100 | Supply Chain Managers, Product Managers |
| Patients Using Insulin Syringes | 140 | Diabetes Patients, Caregivers |
| Pharmacy Retailers | 80 | Pharmacy Owners, Pharmacists |
| Health Insurance Providers | 70 | Policy Analysts, Claims Managers |
Frequently Asked Questions
What is the current value of the Global Insulin Syringes Market?
The Global Insulin Syringes Market is valued at approximately USD 1.8 billion, driven by the increasing prevalence of diabetes and the demand for affordable insulin delivery methods, particularly in low- and middle-income regions.