About the Report
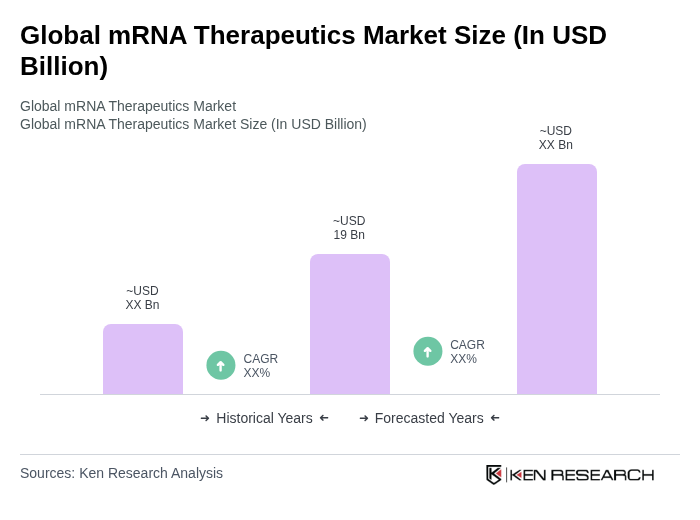
Base Year 2024Global mRNA Therapeutics Market Overview
- The Global mRNA Therapeutics Market is valued at USD 19 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for innovative therapies, particularly in the wake of the COVID-19 pandemic, which has accelerated the development and acceptance of mRNA-based vaccines and treatments. The market is also supported by advancements in lipid nanoparticle delivery systems, improved mRNA design and manufacturing technologies, expanding indications in oncology and rare genetic disorders, and increased public and private investment in research and development.
- Key players in this market are predominantly located in North America and Europe, with the United States and Germany leading the charge. The dominance of these regions can be attributed to their robust healthcare infrastructure, significant investment in biotechnology, supportive regulatory frameworks under agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), and a strong presence of leading pharmaceutical and biotechnology companies such as Moderna, BioNTech, Pfizer, and CureVac that are pioneering mRNA technology.
- In the United States, the regulatory environment for mRNA vaccines and therapeutics is guided by existing biologics and vaccine frameworks, including the Public Health Service Act and the Federal Food, Drug, and Cosmetic Act, administered by the U.S. Food and Drug Administration (FDA). During the COVID?19 public health emergency, the FDA relied on Emergency Use Authorizations (EUAs) under section 564 of the Federal Food, Drug, and Cosmetic Act to expedite the review and availability of mRNA vaccines, and has since provided guidance on the development and licensure of mRNA vaccines and variant-updated formulations, including requirements for clinical data, manufacturing quality, and post?marketing safety surveillance. These instruments collectively facilitate accelerated pathways, clear evidentiary standards, and ongoing oversight for mRNA-based products, thereby enhancing the overall landscape for mRNA therapeutics and supporting broad access to these innovative treatments.
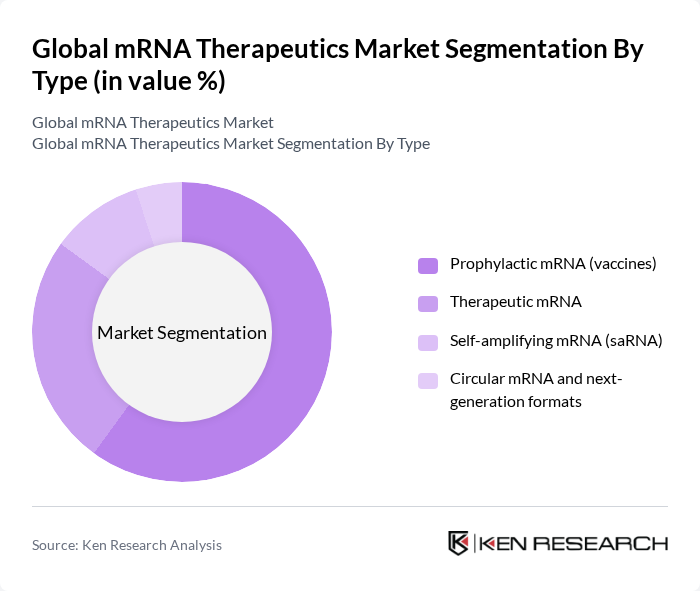
Global mRNA Therapeutics Market Segmentation
By Type:The mRNA therapeutics market can be segmented into four main types: Prophylactic mRNA (vaccines), Therapeutic mRNA, Self-amplifying mRNA (saRNA), and Circular mRNA and next-generation formats. Each of these sub-segments plays a crucial role in addressing various health challenges, with prophylactic mRNA vaccines currently leading the market due to their pivotal role in combating infectious diseases, particularly COVID-19.
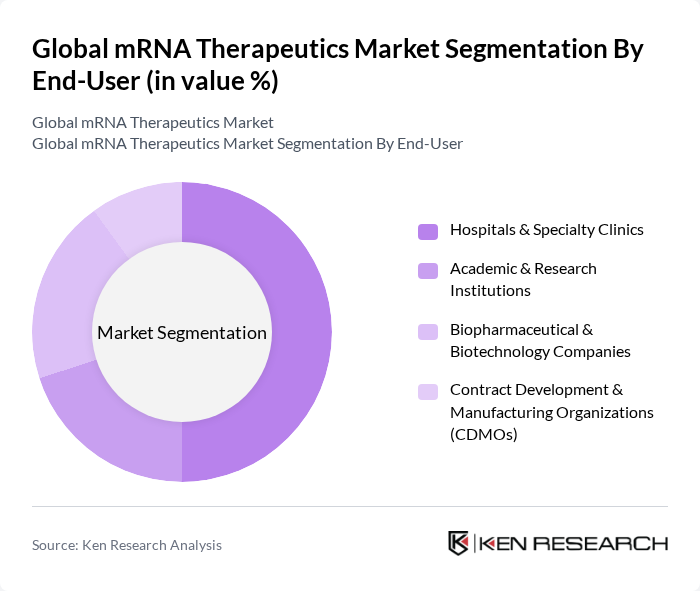
By End-User:The end-user segmentation includes Hospitals & Specialty Clinics, Academic & Research Institutions, Biopharmaceutical & Biotechnology Companies, and Contract Development & Manufacturing Organizations (CDMOs). Hospitals and specialty clinics are currently the leading end-users, driven by the increasing adoption of mRNA therapies in clinical settings and the growing need for advanced treatment options.
Global mRNA Therapeutics Market Competitive Landscape
The Global mRNA Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Moderna, Inc., BioNTech SE, CureVac SE, Arcturus Therapeutics Holdings Inc., Gennova Biopharmaceuticals Ltd., eTheRNA immunotherapies NV, Zydus Lifesciences Ltd. (formerly Zydus Cadila), Sanofi S.A., Pfizer Inc., AstraZeneca plc, Merck KGaA (MilliporeSigma), Eli Lilly and Company, BioNTech Genmab A/S (mRNA oncology collaboration), Translate Bio (a Sanofi company), Roche Holding AG contribute to innovation, geographic expansion, and service delivery in this space.
Global mRNA Therapeutics Market Industry Analysis
Growth Drivers
- Increasing Investment in mRNA Research:In future, global investment in mRNA research is projected to exceed $12 billion, driven by the success of COVID-19 vaccines. The U.S. National Institutes of Health (NIH) allocated approximately $1.8 billion for mRNA-related projects, reflecting a commitment to advancing this technology. Additionally, private sector funding has surged, with companies like Moderna and BioNTech raising over $4 billion collectively, indicating robust confidence in mRNA's potential for various therapeutic applications.
- Rising Prevalence of Infectious Diseases:The World Health Organization (WHO) reported that infectious diseases account for approximately 17 million deaths annually, highlighting a critical need for effective vaccines and treatments. The COVID-19 pandemic has underscored the urgency for rapid vaccine development, with mRNA technology proving effective. In future, the global market for infectious disease vaccines is expected to reach $50 billion, further driving demand for innovative mRNA solutions to combat emerging pathogens and enhance public health responses.
- Advancements in Vaccine Technology:The global vaccine market is projected to reach $110 billion by future, with mRNA vaccines leading the charge due to their rapid development capabilities. The FDA has streamlined approval processes for mRNA vaccines, reducing timeframes from years to months. Furthermore, clinical trials for mRNA-based vaccines have shown efficacy rates exceeding 92% against various diseases, reinforcing the technology's potential. This momentum is expected to attract further investments and research initiatives, propelling market growth.
Market Challenges
- High Production Costs:The production of mRNA therapeutics remains costly, with estimates indicating that manufacturing expenses can exceed $900 per dose. This high cost is primarily due to the complexity of the synthesis process and the need for specialized facilities. As the market expands, companies are under pressure to reduce costs while maintaining quality, which poses a significant challenge to widespread adoption and accessibility of mRNA therapies in various healthcare settings.
- Regulatory Hurdles:Navigating the regulatory landscape for mRNA therapeutics can be daunting, with agencies like the FDA imposing stringent guidelines. In future, the average time for regulatory approval for new mRNA therapies is projected to be around 16 months, which can delay market entry. Additionally, the lack of established frameworks for novel mRNA applications complicates the approval process, potentially stifling innovation and limiting the availability of new treatments to patients in need.
Global mRNA Therapeutics Market Future Outlook
The future of the mRNA therapeutics market appears promising, driven by ongoing advancements in technology and increasing applications across various therapeutic areas. As the industry continues to innovate, particularly in oncology and infectious diseases, the integration of artificial intelligence in drug development is expected to enhance efficiency and precision. Furthermore, the focus on sustainable manufacturing practices will likely attract more investments, fostering a more resilient and adaptive market landscape in the coming years.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets, particularly in Asia and Africa, present significant growth opportunities for mRNA therapeutics. With a combined population exceeding 4.5 billion, these regions are increasingly investing in healthcare infrastructure. In future, the demand for vaccines and therapeutics is expected to rise, driven by government initiatives and international partnerships aimed at improving health outcomes and combating infectious diseases.
- Collaborations with Biotech Firms:Strategic collaborations between pharmaceutical companies and biotech firms are set to enhance mRNA therapeutic development. In future, partnerships are expected to increase by 35%, facilitating knowledge sharing and resource pooling. These collaborations can accelerate the development of innovative therapies, reduce time-to-market, and expand the therapeutic applications of mRNA technology, ultimately benefiting patients and healthcare systems.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Prophylactic mRNA (vaccines) Therapeutic mRNA Self-amplifying mRNA (saRNA) Circular mRNA and next?generation formats |
| By End-User | Hospitals & Specialty Clinics Academic & Research Institutions Biopharmaceutical & Biotechnology Companies Contract Development & Manufacturing Organizations (CDMOs) |
| By Delivery Platform | Lipid Nanoparticles (LNPs) Polymer- and lipid–polymer hybrid nanoparticles Viral and virus?like vectors Physical delivery methods (e.g., electroporation) |
| By Application | Infectious Diseases Oncology (cancer immunotherapy and vaccines) Rare Genetic & Metabolic Disorders Cardiovascular, Respiratory & Other Chronic Diseases |
| By Region | North America Europe Asia-Pacific Latin America |
| By Development Stage | Preclinical Clinical Trials (Phase I–III) Commercialized Platform / Discovery Programs |
| By Funding Source | Government & Multilateral Grants Venture Capital & Private Equity Public Markets (IPOs & Follow?on Offerings) Strategic Partnerships & Licensing Deals |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical and Biotechnology Companies
Contract Research Organizations (CROs)
Healthcare Providers and Hospitals
Biomanufacturers
Insurance Companies and Payers
Public Health Organizations (e.g., World Health Organization)
Players Mentioned in the Report:
Moderna, Inc.
BioNTech SE
CureVac SE
Arcturus Therapeutics Holdings Inc.
Gennova Biopharmaceuticals Ltd.
eTheRNA immunotherapies NV
Zydus Lifesciences Ltd. (formerly Zydus Cadila)
Sanofi S.A.
Pfizer Inc.
AstraZeneca plc
Merck KGaA (MilliporeSigma)
Eli Lilly and Company
BioNTech Genmab A/S (mRNA oncology collaboration)
Translate Bio (a Sanofi company)
Roche Holding AG
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global mRNA Therapeutics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global mRNA Therapeutics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global mRNA Therapeutics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing investment in mRNA research
3.1.2 Rising prevalence of infectious diseases
3.1.3 Advancements in vaccine technology
3.1.4 Growing demand for personalized medicine
3.2 Market Challenges
3.2.1 High production costs
3.2.2 Regulatory hurdles
3.2.3 Limited public awareness
3.2.4 Competition from traditional therapies
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Collaborations with biotech firms
3.3.3 Development of novel delivery systems
3.3.4 Increasing applications in oncology
3.4 Market Trends
3.4.1 Rise of mRNA-based therapeutics
3.4.2 Integration of AI in drug development
3.4.3 Focus on sustainable manufacturing practices
3.4.4 Growth of decentralized clinical trials
3.5 Government Regulation
3.5.1 Streamlined approval processes
3.5.2 Funding for mRNA research initiatives
3.5.3 Guidelines for clinical trials
3.5.4 Intellectual property protections
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global mRNA Therapeutics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global mRNA Therapeutics Market Segmentation
8.1 By Type
8.1.1 Prophylactic mRNA (vaccines)
8.1.2 Therapeutic mRNA
8.1.3 Self-amplifying mRNA (saRNA)
8.1.4 Circular mRNA and next?generation formats
8.2 By End-User
8.2.1 Hospitals & Specialty Clinics
8.2.2 Academic & Research Institutions
8.2.3 Biopharmaceutical & Biotechnology Companies
8.2.4 Contract Development & Manufacturing Organizations (CDMOs)
8.3 By Delivery Platform
8.3.1 Lipid Nanoparticles (LNPs)
8.3.2 Polymer- and lipid–polymer hybrid nanoparticles
8.3.3 Viral and virus?like vectors
8.3.4 Physical delivery methods (e.g., electroporation)
8.4 By Application
8.4.1 Infectious Diseases
8.4.2 Oncology (cancer immunotherapy and vaccines)
8.4.3 Rare Genetic & Metabolic Disorders
8.4.4 Cardiovascular, Respiratory & Other Chronic Diseases
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.6 By Development Stage
8.6.1 Preclinical
8.6.2 Clinical Trials (Phase I–III)
8.6.3 Commercialized
8.6.4 Platform / Discovery Programs
8.7 By Funding Source
8.7.1 Government & Multilateral Grants
8.7.2 Venture Capital & Private Equity
8.7.3 Public Markets (IPOs & Follow?on Offerings)
8.7.4 Strategic Partnerships & Licensing Deals
9. Global mRNA Therapeutics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Core Focus (prophylactic vs therapeutic, key indications)
9.2.3 Total Revenue and mRNA?related Revenue
9.2.4 R&D Intensity (mRNA R&D spend as % of total revenue)
9.2.5 Size of mRNA Pipeline (number of candidates by phase)
9.2.6 Time?to?Market for Lead mRNA Assets
9.2.7 Manufacturing Capacity (doses / grams of mRNA per year)
9.2.8 Geographic Reach and Regulatory Approvals (regions/countries)
9.2.9 Strategic Collaborations & Licensing Agreements
9.2.10 Market Share in Global mRNA Therapeutics
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Moderna, Inc.
9.5.2 BioNTech SE
9.5.3 CureVac SE
9.5.4 Arcturus Therapeutics Holdings Inc.
9.5.5 Gennova Biopharmaceuticals Ltd.
9.5.6 eTheRNA immunotherapies NV
9.5.7 Zydus Lifesciences Ltd. (formerly Zydus Cadila)
9.5.8 Sanofi S.A.
9.5.9 Pfizer Inc.
9.5.10 AstraZeneca plc
9.5.11 Merck KGaA (MilliporeSigma)
9.5.12 Eli Lilly and Company
9.5.13 BioNTech Genmab A/S (mRNA oncology collaboration)
9.5.14 Translate Bio (a Sanofi company)
9.5.15 Roche Holding AG
10. Global mRNA Therapeutics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Spending Trends
10.2.3 Budgeting Cycles
10.2.4 Cost Management Strategies
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Innovative Therapies
10.3.2 Cost of Treatment
10.3.3 Supply Chain Reliability
10.3.4 Regulatory Compliance Challenges
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.4.4 Support Systems
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Feedback Mechanisms
10.5.3 Scalability Potential
10.5.4 Future Investment Opportunities
11. Global mRNA Therapeutics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports from industry associations and research firms
- Review of scientific literature and clinical trial data related to mRNA therapeutics
- Examination of regulatory frameworks and guidelines from health authorities globally
Primary Research
- Interviews with key opinion leaders in the biotechnology and pharmaceutical sectors
- Surveys targeting clinical researchers and healthcare professionals involved in mRNA studies
- Focus groups with patients and advocacy groups to understand perceptions and needs
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert insights
- Triangulation of quantitative data with qualitative insights from interviews
- Sanity checks through peer reviews and expert panel discussions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of global mRNA market size based on pharmaceutical sales data and growth rates
- Segmentation by therapeutic area, including oncology, infectious diseases, and rare diseases
- Incorporation of trends in vaccine development and public health initiatives
Bottom-up Modeling
- Collection of data on production capacities and sales volumes from leading mRNA manufacturers
- Cost analysis of mRNA production processes and pricing strategies
- Volume x price calculations for various therapeutic applications
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating technological advancements and market dynamics
- Scenario modeling based on potential regulatory changes and market entry of new players
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology mRNA Therapeutics | 120 | Oncologists, Clinical Researchers |
| Infectious Disease Vaccines | 110 | Immunologists, Vaccine Developers |
| Rare Disease Treatments | 70 | Geneticists, Rare Disease Specialists |
| Regulatory Perspectives | 80 | Regulatory Affairs Managers, Compliance Officers |
| Market Access Strategies | 75 | Market Access Managers, Health Economists |
Frequently Asked Questions
What is the current value of the Global mRNA Therapeutics Market?
The Global mRNA Therapeutics Market is valued at approximately USD 19 billion, reflecting significant growth driven by the demand for innovative therapies, particularly following the COVID-19 pandemic and advancements in mRNA technology.