Region:Global
Author(s):Shubham
Product Code:KRAC8906
Pages:84
Published On:November 2025
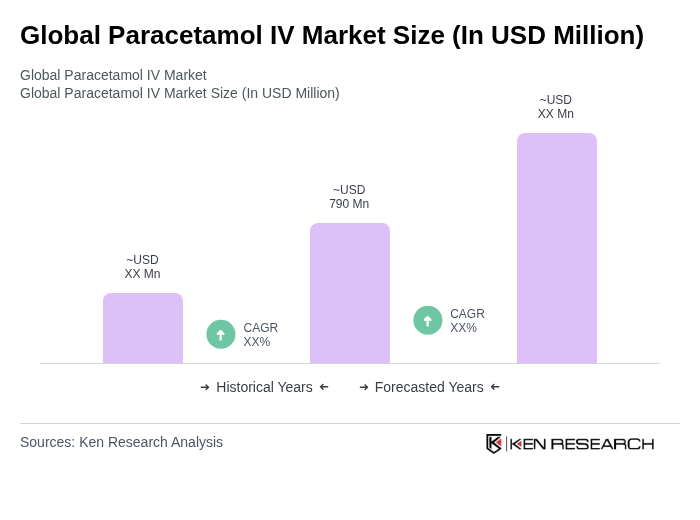
By Type:The market is segmented into Single-Dose Vials, Multi-Dose Vials, Pre-filled Syringes, and IV Infusion Bags. Among these, Single-Dose Vials are currently leading the market due to their convenience and safety in administration, particularly in hospital settings. Multi-Dose Vials are also widely used, especially in outpatient care, while Pre-filled Syringes are gaining traction for their ease of use and infection control benefits. IV Infusion Bags are utilized in more specialized applications, such as intensive care and emergency settings, contributing to the overall market growth .
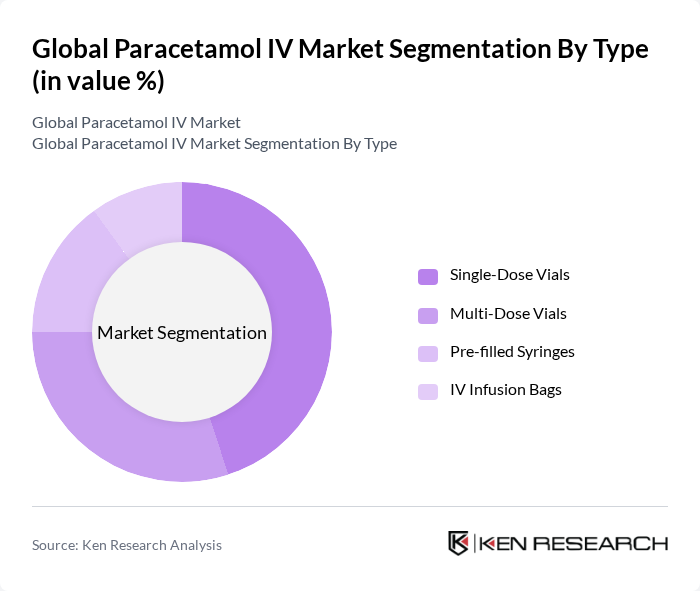
By End-User:The end-user segmentation includes Hospitals and Clinics, Ambulatory Surgical Centers (ASCs), Home Healthcare Settings, and Others. Hospitals and Clinics dominate the market due to the high volume of patients requiring intravenous medications for pain management and fever control. ASCs are also experiencing growth as outpatient procedures increase, while Home Healthcare Settings are emerging as a significant segment due to the rising trend of at-home care and the growing geriatric population .
The Global Paracetamol IV Market is characterized by a dynamic mix of regional and international players. Leading participants such as Fresenius Kabi AG, Pfizer Inc., B. Braun Melsungen AG, Hikma Pharmaceuticals PLC, Baxter International Inc., Sandoz International GmbH, Teva Pharmaceutical Industries Ltd., Aurobindo Pharma Ltd., Sun Pharmaceutical Industries Ltd., Novartis AG, Sanofi S.A., Johnson & Johnson, Eton Pharmaceuticals, Inc., Mylan N.V., Amgen Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Paracetamol IV market appears promising, driven by the increasing integration of digital health technologies and a shift towards personalized medicine. As healthcare systems evolve, the demand for patient-centric care will likely lead to greater adoption of IV medications in outpatient settings. Additionally, the expansion of healthcare infrastructure in emerging economies presents significant growth opportunities, enabling wider access to effective pain management solutions and enhancing overall patient care.
| Segment | Sub-Segments |
|---|---|
| By Type | Single-Dose Vials Multi-Dose Vials Pre-filled Syringes IV Infusion Bags |
| By End-User | Hospitals and Clinics Ambulatory Surgical Centers (ASCs) Home Healthcare Settings Others |
| By Indication | Pain Management Pyrexia (Fever) Management Post-operative Care Others |
| By Application | Surgical Applications Non-surgical Applications |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Distribution Channel | Direct Hospital Supply Pharmaceutical Wholesalers Retail Pharmacies Online Channels |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Pharmacy Management | 120 | Pharmacy Directors, Clinical Pharmacists |
| Pain Management Specialists | 100 | Anesthesiologists, Pain Management Physicians |
| Healthcare Procurement Officers | 80 | Procurement Managers, Supply Chain Directors |
| Clinical Trial Coordinators | 60 | Clinical Researchers, Trial Managers |
| Regulatory Affairs Experts | 50 | Regulatory Managers, Compliance Officers |
The Global Paracetamol IV Market is valued at approximately USD 790 million, reflecting a significant growth driven by increasing pain management needs and the rising demand for intravenous medications in healthcare settings.