Region:Global
Author(s):Dev
Product Code:KRAC1941
Pages:85
Published On:October 2025
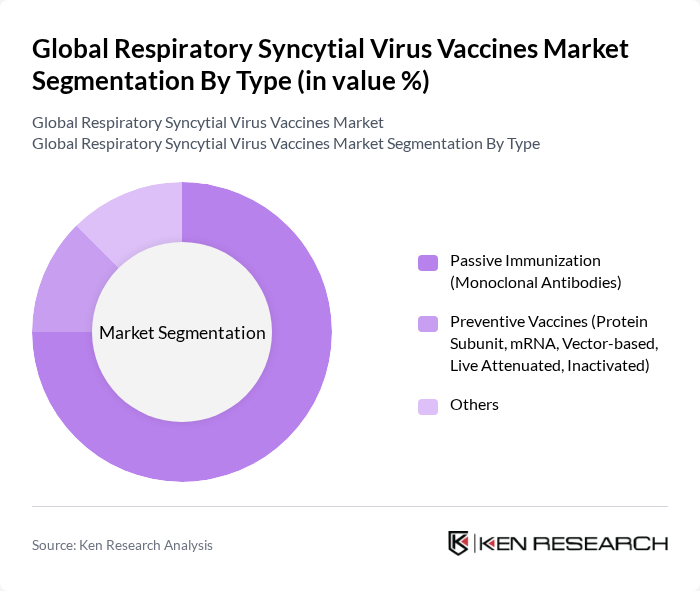
By Type:The market is segmented into Passive Immunization (Monoclonal Antibodies), Preventive Vaccines (Protein Subunit, mRNA, Vector-based, Live Attenuated, Inactivated), and Others. Preventive Vaccines dominate the market due to their effectiveness in preventing RSV infections in vulnerable populations, particularly infants and the elderly. The increasing focus on preventive healthcare and the rising incidence of RSV infections are driving the demand for these vaccines. Passive Immunization, while important, is primarily used in specific high-risk cases, thus holding a smaller market share.
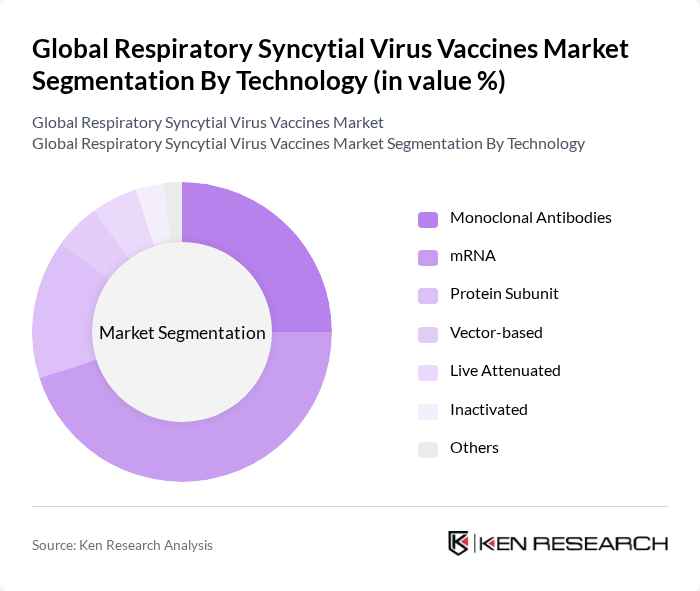
By Technology:The market is categorized into Monoclonal Antibodies, mRNA, Protein Subunit, Vector-based, Live Attenuated, Inactivated, and Others. The mRNA technology is currently leading the market due to its rapid development and effectiveness demonstrated in recent vaccine candidates. The success of mRNA vaccines during the COVID-19 pandemic has accelerated interest and investment in this technology for RSV, making it a preferred choice among healthcare providers and patients. Monoclonal Antibodies also play a significant role, particularly in high-risk populations, but their market presence is comparatively smaller.
The Global Respiratory Syncytial Virus Vaccines Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., GlaxoSmithKline plc (GSK), Sanofi S.A., AstraZeneca plc, Moderna, Inc., Merck & Co., Inc., Johnson & Johnson, Bavarian Nordic A/S, Novavax, Inc., Icosavax, Inc., Meissa Vaccines, Inc., Codagenix, Inc., Blue Lake Biotechnology, Inc., Vaxart, Inc., Clover Biopharmaceuticals, Advaccine Biopharmaceuticals, Nuance Pharma, Emergent BioSolutions Inc., Seqirus (CSL Limited), Immunovant, Inc., Inovio Pharmaceuticals, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the RSV vaccine market appears promising, driven by ongoing technological advancements and increased public health initiatives. As healthcare systems continue to prioritize preventive measures, the integration of digital health technologies will enhance vaccine distribution and monitoring. Furthermore, collaborations between pharmaceutical companies and healthcare organizations are expected to foster innovation, leading to the development of more effective vaccines. This collaborative approach will likely address public concerns and improve vaccination rates, ultimately benefiting the market landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Passive Immunization (Monoclonal Antibodies) Preventive Vaccines (Protein Subunit, mRNA, Vector-based, Live Attenuated, Inactivated) Others |
| By Technology | Monoclonal Antibodies mRNA Protein Subunit Vector-based Live Attenuated Inactivated Others |
| By Targeted Population | Infants Children Adults Elderly Immunocompromised Individuals |
| By Distribution Channel | Hospitals Retail Pharmacies Government Suppliers Online Sales Others |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Clinical Stage | Preclinical Phase I Phase II Phase III |
| By Pricing Strategy | Premium Pricing Competitive Pricing Value-based Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatric Healthcare Providers | 95 | Pediatricians, Family Physicians |
| Vaccine Manufacturers | 65 | Product Managers, R&D Directors |
| Public Health Officials | 55 | Health Policy Advisors, Epidemiologists |
| Parents of Young Children | 80 | Caregivers, Health Advocates |
| Pharmacists and Immunization Specialists | 70 | Pharmacy Managers, Immunization Coordinators |
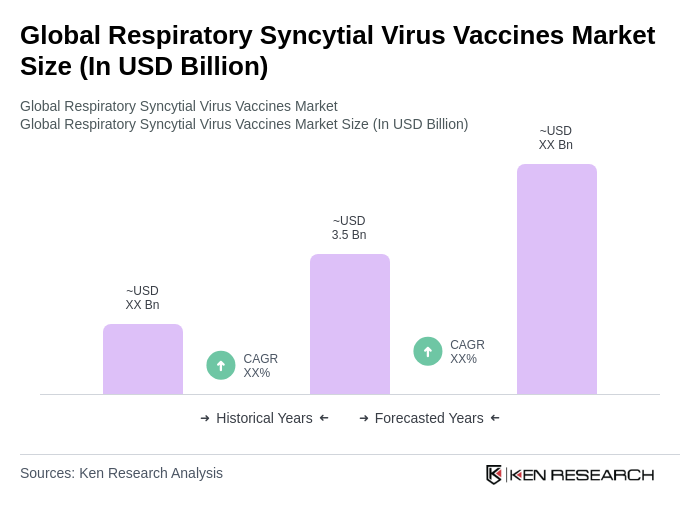
The Global Respiratory Syncytial Virus Vaccines Market is valued at approximately USD 3.5 billion, driven by the rising prevalence of RSV infections, particularly among infants and the elderly, along with advancements in vaccine technology and increased healthcare expenditures.