About the Report
Base Year 2024Global Sustained Release Excipients Market Overview
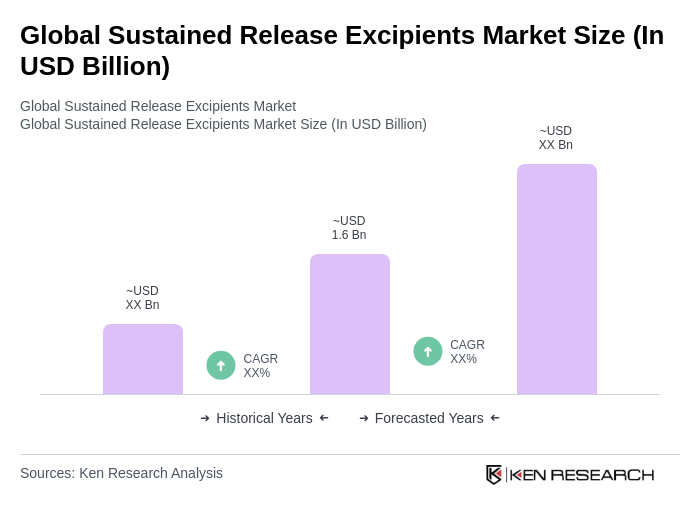
- The Global Sustained Release Excipients Market is valued at USD 1.6 billion, based on a five-year historical analysis. This value aligns with multiple industry estimates indicating the market in the low-to-mid billion range driven by uptake in controlled-release formulations across oral and parenteral dosage forms .
- Key players in this market are concentrated in regions such as North America and Europe, where robust pharmaceutical industries and significant R&D investments are prevalent. Countries like the United States and Germany dominate due to advanced healthcare infrastructure, strong regulatory frameworks, and dense clusters of pharmaceutical manufacturers and excipient suppliers, fostering innovation and market growth .
- In 2023, the U.S. FDA implemented new guidelines for the use of excipients in drug formulations, emphasizing the need for comprehensive safety and efficacy data. This is consistent with the FDA’s modern regulatory approach to excipients via Quality by Design, DMF review, and adoption of novel excipients frameworks, where safety, functionality, and performance data are central to acceptance in sustained-release applications .
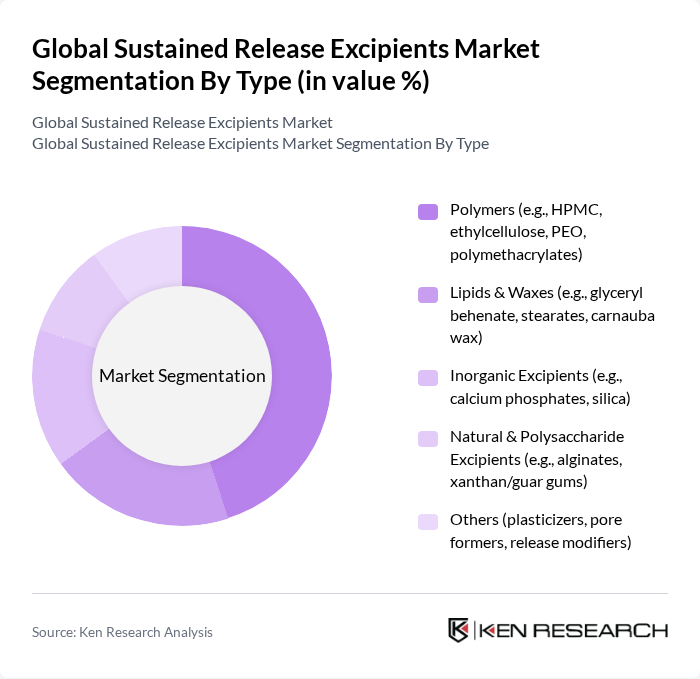
Global Sustained Release Excipients Market Segmentation
By Type:The market is segmented into various types of excipients, including polymers, lipids & waxes, inorganic excipients, natural & polysaccharide excipients, and others. Among these, polymers are the most dominant due to their versatility and effectiveness in controlling drug release rates. The increasing preference for polymer-based formulations in the pharmaceutical industry is driven by their ability to enhance bioavailability and stability of active pharmaceutical ingredients .
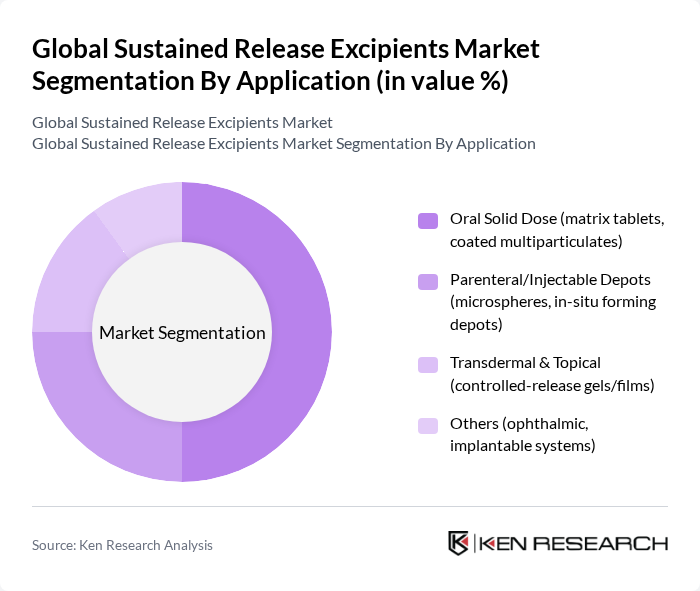
By Application:The applications of sustained release excipients are diverse, including oral solid doses, parenteral/injectable depots, transdermal & topical applications, and others. Oral solid doses dominate the market due to their widespread use and the growing trend towards patient-friendly formulations. The demand for matrix tablets and coated multiparticulates is particularly high, as they offer controlled release profiles that improve therapeutic outcomes .
Global Sustained Release Excipients Market Competitive Landscape
The Global Sustained Release Excipients Market is characterized by a dynamic mix of regional and international players. Leading participants such as BASF SE, Evonik Industries AG, Ashland Inc., Dow (The Dow Chemical Company), Croda International Plc, Merck KGaA (EMD Millipore Life Science), JRS Pharma GmbH & Co. KG, Colorcon, Inc., FMC Corporation (note: pharma excipients now via IFF/Nouryon affiliates), Ingredion Incorporated, Roquette Frères, DuPont de Nemours, Inc., Sensient Technologies Corporation, Gattefossé S.A., TSI Group Ltd. contribute to innovation, geographic expansion, and service delivery in this space .
Global Sustained Release Excipients Market Industry Analysis
Growth Drivers
- Increasing Demand for Controlled Release Formulations:The global market for controlled release formulations is projected to reach $50 billion in future, driven by the need for improved therapeutic outcomes. This demand is fueled by the rising incidence of chronic diseases, which accounted for approximately 74% of all global deaths, according to the World Health Organization. As healthcare systems prioritize patient-centric approaches, the adoption of sustained release excipients is expected to rise significantly, enhancing drug efficacy and patient compliance.
- Rising Prevalence of Chronic Diseases:Chronic diseases such as diabetes and cardiovascular disorders are on the rise, with the World Health Organization estimating that over 1.5 billion people will be living with diabetes globally in future. This surge necessitates innovative drug delivery systems, including sustained release excipients, to manage long-term treatment regimens effectively. The increasing healthcare expenditure, projected to reach over $9 trillion in future, further supports the growth of this market segment.
- Advancements in Drug Delivery Technologies:The sustained release excipients market is benefiting from rapid advancements in drug delivery technologies, with investments in R&D expected to exceed $200 billion in future. Innovations such as nanotechnology and 3D printing are enhancing the formulation of sustained release systems, allowing for more precise dosing and improved bioavailability. These technological advancements are crucial in meeting the evolving demands of the pharmaceutical industry, particularly in personalized medicine.
Market Challenges
- High Development Costs:The development of sustained release excipients involves significant financial investment, often exceeding $1 million per formulation. This high cost is a barrier for many small to medium-sized enterprises, limiting their ability to innovate and compete in the market. Additionally, the lengthy development timelines, which can span several years, further exacerbate the financial strain on companies, hindering market entry and expansion.
- Stringent Regulatory Requirements:The pharmaceutical industry faces rigorous regulatory scrutiny, with the FDA and EMA imposing strict guidelines on excipient safety and efficacy. Compliance with these regulations can delay product launches by up to 18 months, significantly impacting market dynamics. The complexity of navigating these regulatory landscapes poses a challenge for manufacturers, particularly those introducing novel excipients, as they must ensure adherence to evolving standards.
Global Sustained Release Excipients Market Future Outlook
The future of the sustained release excipients market appears promising, driven by the increasing focus on personalized medicine and the integration of advanced technologies. As healthcare systems evolve, the demand for tailored drug delivery solutions will likely rise, fostering innovation in excipient development. Furthermore, the growing emphasis on sustainability will push manufacturers to explore eco-friendly excipients, aligning with global environmental goals and enhancing market competitiveness.
Market Opportunities
- Expansion in Emerging Markets:Emerging markets, particularly in Asia-Pacific, are witnessing rapid growth in healthcare infrastructure, with spending projected to reach $1.5 trillion in future. This expansion presents significant opportunities for sustained release excipients, as local pharmaceutical companies seek to enhance their product offerings and meet the increasing demand for advanced drug delivery systems.
- Increasing Adoption of Biologics:The biologics market is expected to surpass $500 billion in future, driven by the rising prevalence of chronic diseases and advancements in biotechnology. This trend creates opportunities for sustained release excipients, as they play a crucial role in the formulation of biologics, ensuring optimal delivery and therapeutic effectiveness, thereby enhancing patient outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Polymers (e.g., HPMC, ethylcellulose, PEO, polymethacrylates) Lipids & Waxes (e.g., glyceryl behenate, stearates, carnauba wax) Inorganic Excipients (e.g., calcium phosphates, silica) Natural & Polysaccharide Excipients (e.g., alginates, xanthan/guar gums) Others (plasticizers, pore formers, release modifiers) |
| By Application | Oral Solid Dose (matrix tablets, coated multiparticulates) Parenteral/Injectable Depots (microspheres, in-situ forming depots) Transdermal & Topical (controlled-release gels/films) Others (ophthalmic, implantable systems) |
| By End-User | Pharmaceutical & Biopharma Manufacturers Contract Development & Manufacturing Organizations (CDMOs/CMOs) Academic & Research Institutes Others (compounding pharmacies, specialty formulators) |
| By Distribution Channel | Direct Sales to Manufacturers Distributors & Channel Partners Online/Procurement Platforms Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Price Range | Low Price Medium Price High Price |
| By Regulatory Compliance | Pharmacopoeial Grade (USP/NF, Ph. Eur., JP) GMP/EXCiPACT/NSF/IPEC Certified Others (DMF-supported, QbD-enabled) |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical Manufacturers
Biotechnology Companies
Contract Research Organizations
Raw Material Suppliers
Pharmaceutical Packaging Companies
Quality Control and Testing Laboratories
Players Mentioned in the Report:
BASF SE
Evonik Industries AG
Ashland Inc.
Dow (The Dow Chemical Company)
Croda International Plc
Merck KGaA (EMD Millipore Life Science)
JRS Pharma GmbH & Co. KG
Colorcon, Inc.
FMC Corporation (note: pharma excipients now via IFF/Nouryon affiliates)
Ingredion Incorporated
Roquette Freres
DuPont de Nemours, Inc.
Sensient Technologies Corporation
Gattefosse S.A.
TSI Group Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Sustained Release Excipients Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Sustained Release Excipients Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Sustained Release Excipients Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Controlled Release Formulations
3.1.2 Rising Prevalence of Chronic Diseases
3.1.3 Advancements in Drug Delivery Technologies
3.1.4 Growing Investment in Pharmaceutical R&D
3.2 Market Challenges
3.2.1 High Development Costs
3.2.2 Stringent Regulatory Requirements
3.2.3 Competition from Alternative Drug Delivery Systems
3.2.4 Limited Awareness Among End-Users
3.3 Market Opportunities
3.3.1 Expansion in Emerging Markets
3.3.2 Increasing Adoption of Biologics
3.3.3 Development of Novel Excipients
3.3.4 Collaborations and Partnerships in R&D
3.4 Market Trends
3.4.1 Shift Towards Personalized Medicine
3.4.2 Integration of AI in Drug Formulation
3.4.3 Focus on Sustainable and Green Excipients
3.4.4 Growth of Online Pharmaceutical Sales Channels
3.5 Government Regulation
3.5.1 FDA Guidelines on Excipients
3.5.2 EMA Regulations for Drug Formulations
3.5.3 WHO Standards for Pharmaceutical Excipients
3.5.4 Environmental Regulations Impacting Excipients
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Sustained Release Excipients Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Sustained Release Excipients Market Segmentation
8.1 By Type
8.1.1 Polymers (e.g., HPMC, ethylcellulose, PEO, polymethacrylates)
8.1.2 Lipids & Waxes (e.g., glyceryl behenate, stearates, carnauba wax)
8.1.3 Inorganic Excipients (e.g., calcium phosphates, silica)
8.1.4 Natural & Polysaccharide Excipients (e.g., alginates, xanthan/guar gums)
8.1.5 Others (plasticizers, pore formers, release modifiers)
8.2 By Application
8.2.1 Oral Solid Dose (matrix tablets, coated multiparticulates)
8.2.2 Parenteral/Injectable Depots (microspheres, in-situ forming depots)
8.2.3 Transdermal & Topical (controlled-release gels/films)
8.2.4 Others (ophthalmic, implantable systems)
8.3 By End-User
8.3.1 Pharmaceutical & Biopharma Manufacturers
8.3.2 Contract Development & Manufacturing Organizations (CDMOs/CMOs)
8.3.3 Academic & Research Institutes
8.3.4 Others (compounding pharmacies, specialty formulators)
8.4 By Distribution Channel
8.4.1 Direct Sales to Manufacturers
8.4.2 Distributors & Channel Partners
8.4.3 Online/Procurement Platforms
8.4.4 Others
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.6 By Price Range
8.6.1 Low Price
8.6.2 Medium Price
8.6.3 High Price
8.7 By Regulatory Compliance
8.7.1 Pharmacopoeial Grade (USP/NF, Ph. Eur., JP)
8.7.2 GMP/EXCiPACT/NSF/IPEC Certified
8.7.3 Others (DMF-supported, QbD-enabled)
9. Global Sustained Release Excipients Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue from Sustained/Controlled-Release Excipient Portfolio (%)
9.2.4 Global Supply Footprint (number of GMP sites/regions served)
9.2.5 Regulatory Strength (DMFs filed; EXCiPACT/NSF/IPEC certifications)
9.2.6 R&D Intensity (R&D spend %, patents/publications in SR excipients)
9.2.7 Portfolio Breadth (polymers, lipids, polysaccharides, inorganic)
9.2.8 Key Partnerships with Pharma/CDMOs (number and notables)
9.2.9 On-time Delivery/Lead Time Performance
9.2.10 Quality Metrics (batch failure rate, audit observations, recall history)
9.2.11 Sustainability Metrics (biobased content, scope 1–3 targets, audits)
9.2.12 Pricing Positioning (premium/standard/value for SR grades)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 BASF SE
9.5.2 Evonik Industries AG
9.5.3 Ashland Inc.
9.5.4 Dow (The Dow Chemical Company)
9.5.5 Croda International Plc
9.5.6 Merck KGaA (EMD Millipore Life Science)
9.5.7 JRS Pharma GmbH & Co. KG
9.5.8 Colorcon, Inc.
9.5.9 FMC Corporation (note: pharma excipients now via IFF/Nouryon affiliates)
9.5.10 Ingredion Incorporated
9.5.11 Roquette Frères
9.5.12 DuPont de Nemours, Inc.
9.5.13 Sensient Technologies Corporation
9.5.14 Gattefossé S.A.
9.5.15 TSI Group Ltd.
10. Global Sustained Release Excipients Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government Procurement Policies
10.1.2 Budget Allocations for Pharmaceuticals
10.1.3 Supplier Selection Criteria
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Pharmaceutical Infrastructure
10.2.2 Energy Consumption Trends
10.2.3 Budgeting for Sustainable Practices
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Sourcing Excipients
10.3.2 Quality Assurance Issues
10.3.3 Regulatory Compliance Difficulties
10.4 User Readiness for Adoption
10.4.1 Awareness of Sustained Release Technologies
10.4.2 Training and Support Needs
10.4.3 Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Case Opportunities
11. Global Sustained Release Excipients Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from pharmaceutical and biotechnology associations
- Market analysis from trade publications focusing on excipients and drug delivery systems
- Regulatory documents and guidelines from the FDA and EMA regarding excipient usage
Primary Research
- Interviews with formulation scientists at pharmaceutical companies
- Surveys with procurement managers in the excipients supply chain
- Field interviews with quality assurance professionals in drug manufacturing
Validation & Triangulation
- Cross-validation of data through multiple industry sources and expert opinions
- Triangulation of market size estimates from sales data and production statistics
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of global pharmaceutical market size to estimate excipient demand
- Segmentation by therapeutic area and excipient type for detailed insights
- Incorporation of trends in drug formulation and delivery methods
Bottom-up Modeling
- Volume estimates based on production capacities of leading excipient manufacturers
- Cost analysis derived from pricing models of various excipient categories
- Estimation of market share based on sales data from key players
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors like R&D spending and regulatory changes
- Scenario modeling based on potential shifts in drug development trends and market dynamics
- Baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Formulation Development | 120 | Formulation Scientists, R&D Managers |
| Excipients Procurement | 90 | Procurement Managers, Supply Chain Analysts |
| Quality Control in Manufacturing | 70 | Quality Assurance Managers, Compliance Officers |
| Regulatory Affairs in Pharmaceuticals | 60 | Regulatory Affairs Specialists, Compliance Managers |
| Market Trends in Drug Delivery Systems | 80 | Market Analysts, Business Development Managers |
Frequently Asked Questions
What is the current value of the Global Sustained Release Excipients Market?
The Global Sustained Release Excipients Market is valued at approximately USD 1.6 billion, based on a five-year historical analysis. This valuation reflects the growing demand for controlled-release formulations in both oral and parenteral dosage forms.