About the Report
Base Year 2024Global Syndromic Multiplex Diagnostics Market Overview
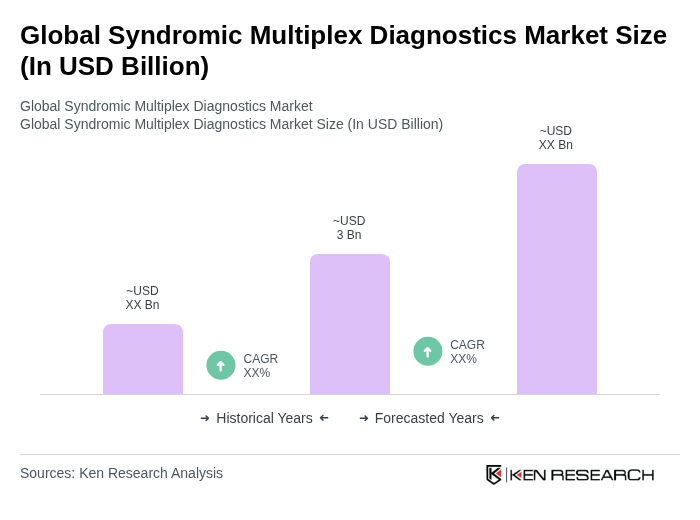
- The Global Syndromic Multiplex Diagnostics Market is valued at USD 3 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of infectious diseases, advancements in diagnostic technologies such as PCR and microfluidics, and the rising demand for rapid and accurate testing solutions. The market has seen a surge in adoption due to the need for efficient disease management and outbreak control, particularly highlighted during global health crises.
- Key regions in this market include North America, Europe, and Asia Pacific, which dominate due to their robust healthcare infrastructure, significant investments in research and development, and a high prevalence of infectious diseases. The presence of leading diagnostic companies and a strong focus on innovation further contribute to their market leadership, making these regions pivotal in shaping the future of multiplex diagnostics.
- The Clinical Laboratory Improvement Amendments of 1988 (CLIA), issued by the U.S. Department of Health and Human Services, govern laboratory testing including multiplex diagnostics for infectious diseases. This regulation establishes quality standards for laboratory practices, requiring certification for high-complexity tests, proficiency testing, personnel qualifications, and quality control measures to ensure accurate and reliable results.
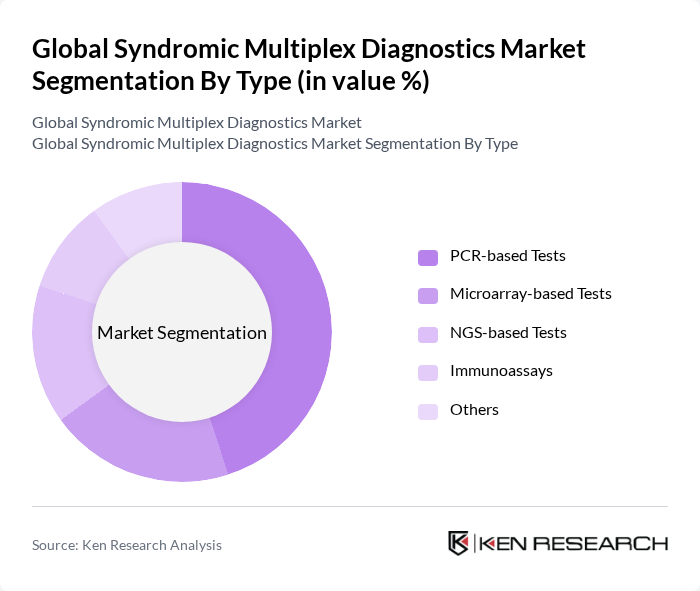
Global Syndromic Multiplex Diagnostics Market Segmentation
By Type:The market is segmented into various types, including PCR-based tests, NGS-based tests, microarray-based tests, immunoassays, and others. Among these, PCR-based tests are leading due to their high sensitivity and specificity, making them the preferred choice for diagnosing infectious diseases. The demand for NGS-based tests is also growing, driven by their ability to provide comprehensive genomic information.
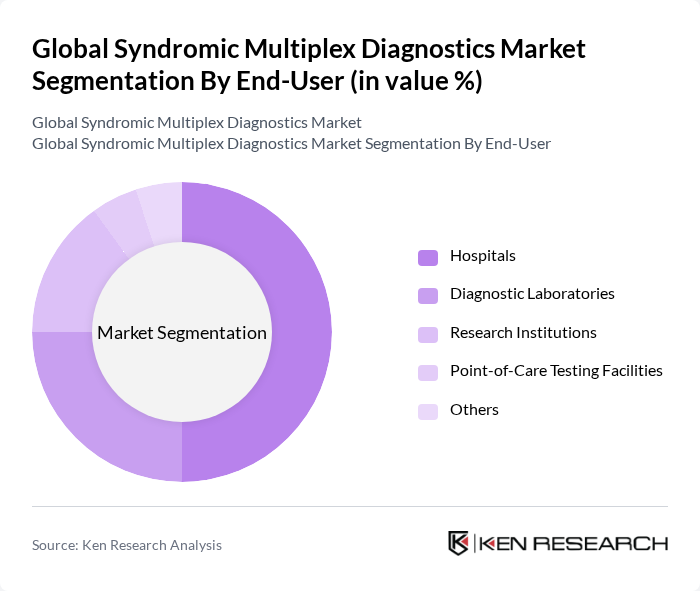
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, research institutions, point-of-care settings, and others. Hospitals are the dominant end-user, driven by the increasing number of patients requiring rapid diagnostics and the integration of advanced diagnostic technologies in clinical settings. Diagnostic laboratories are also significant, as they provide specialized testing services and contribute to the overall efficiency of disease diagnosis.
Global Syndromic Multiplex Diagnostics Market Competitive Landscape
The Global Syndromic Multiplex Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Thermo Fisher Scientific, BioMérieux, Cepheid, Hologic, Qiagen, Luminex Corporation, Becton, Dickinson and Company, Agilent Technologies, PerkinElmer, Siemens Healthineers, GenMark Diagnostics, Illumina, Eiken Chemical Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
Global Syndromic Multiplex Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Infectious Diseases:The World Health Organization reported that infectious diseases account for approximately 15 million deaths annually, with a significant rise in cases of diseases like COVID-19, tuberculosis, and HIV. This alarming trend has led to a heightened demand for effective diagnostic solutions. In future, the global burden of infectious diseases is projected to increase, necessitating advanced multiplex diagnostics to facilitate timely and accurate disease detection, thereby driving market growth.
- Advancements in Diagnostic Technologies:The global investment in healthcare technology is expected to reach $10 trillion in future, with a substantial portion directed towards diagnostic innovations. Technologies such as next-generation sequencing and microfluidics are revolutionizing multiplex diagnostics, enabling simultaneous detection of multiple pathogens. These advancements not only enhance accuracy but also reduce turnaround times, making diagnostics more efficient and accessible, thus propelling market expansion in the None region.
- Rising Demand for Rapid Diagnostic Tests:The global market for rapid diagnostic tests is projected to exceed $30 billion in future, driven by the need for quick and reliable results in clinical settings. The COVID-19 pandemic has underscored the importance of rapid testing, leading to increased investments in multiplex diagnostic systems. This trend is expected to continue, as healthcare providers seek to improve patient outcomes through timely diagnosis, further stimulating market growth in the None region.
Market Challenges
- High Costs of Multiplex Diagnostic Systems:The initial investment for multiplex diagnostic systems can exceed $100,000, posing a significant barrier for many healthcare facilities, particularly in resource-limited settings. This high cost can limit access to advanced diagnostic technologies, hindering widespread adoption. As healthcare budgets tighten, the financial burden associated with these systems may slow market growth, especially in the None region where funding is often constrained.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape for multiplex diagnostics can be complex and time-consuming. In the U.S., the FDA requires extensive clinical validation, which can delay product launches by several months. Similarly, in Europe, compliance with CE marking requirements adds to the challenges. These regulatory hurdles can deter innovation and slow the introduction of new multiplex diagnostic solutions, impacting market dynamics in the None region.
Global Syndromic Multiplex Diagnostics Market Future Outlook
The future of the syndromic multiplex diagnostics market appears promising, driven by technological advancements and an increasing focus on personalized medicine. As healthcare systems evolve, the integration of artificial intelligence and machine learning into diagnostic processes is expected to enhance accuracy and efficiency. Additionally, the shift towards decentralized testing and point-of-care solutions will likely expand access to diagnostics, particularly in underserved areas, fostering growth and innovation in the None region.
Market Opportunities
- Expansion in Emerging Markets:Emerging markets, particularly in Asia and Africa, present significant growth opportunities for multiplex diagnostics. With rising healthcare expenditures projected to reach $1.5 trillion in future in these regions, there is an increasing demand for advanced diagnostic solutions. This growth is driven by improving healthcare infrastructure and a growing awareness of the importance of early disease detection.
- Development of Point-of-Care Testing Solutions:The global point-of-care testing market is expected to surpass $50 billion in future, driven by the need for rapid and accessible diagnostics. Innovations in portable diagnostic devices and mobile health technologies are creating opportunities for multiplex diagnostics at the point of care. This trend is particularly relevant in rural and underserved areas, where access to traditional laboratory services is limited.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | PCR-based tests NGS-based tests Microarray-based tests Immunoassays Others |
| By End-User | Hospitals Diagnostic laboratories Research institutions Point-of-care settings Others |
| By Application | Infectious disease diagnostics Cancer diagnostics Genetic testing Blood screening Others |
| By Sample Type | Blood samples Urine samples Saliva samples Tissue samples Others |
| By Technology | Real-time PCR Digital PCR Next-generation sequencing Microfluidics Others |
| By Distribution Channel | Direct sales Distributors Online sales Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers of Diagnostic Devices
Distributors and Retailers of Medical Equipment
Healthcare Providers and Hospitals
Technology Providers for Diagnostic Solutions
Industry Associations and Advocacy Groups
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Roche Diagnostics
Abbott Laboratories
Thermo Fisher Scientific
BioMerieux
Cepheid
Hologic
Qiagen
Luminex Corporation
Becton, Dickinson and Company
Agilent Technologies
PerkinElmer
Siemens Healthineers
GenMark Diagnostics
Illumina
Eiken Chemical Co., Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Syndromic Multiplex Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Syndromic Multiplex Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Syndromic Multiplex Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of infectious diseases
3.1.2 Advancements in diagnostic technologies
3.1.3 Rising demand for rapid diagnostic tests
3.1.4 Growing focus on personalized medicine
3.2 Market Challenges
3.2.1 High costs of multiplex diagnostic systems
3.2.2 Regulatory hurdles and compliance issues
3.2.3 Limited awareness among healthcare providers
3.2.4 Competition from traditional diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Development of point-of-care testing solutions
3.3.3 Collaborations with research institutions
3.3.4 Integration of AI in diagnostic processes
3.4 Market Trends
3.4.1 Shift towards decentralized testing
3.4.2 Increasing adoption of telemedicine
3.4.3 Focus on molecular diagnostics
3.4.4 Rise in consumer-driven healthcare
3.5 Government Regulation
3.5.1 FDA guidelines for multiplex diagnostics
3.5.2 CE marking requirements in Europe
3.5.3 Reimbursement policies for diagnostic tests
3.5.4 Compliance with health and safety standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Syndromic Multiplex Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Syndromic Multiplex Diagnostics Market Segmentation
8.1 By Type
8.1.1 PCR-based tests
8.1.2 NGS-based tests
8.1.3 Microarray-based tests
8.1.4 Immunoassays
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic laboratories
8.2.3 Research institutions
8.2.4 Point-of-care settings
8.2.5 Others
8.3 By Application
8.3.1 Infectious disease diagnostics
8.3.2 Cancer diagnostics
8.3.3 Genetic testing
8.3.4 Blood screening
8.3.5 Others
8.4 By Sample Type
8.4.1 Blood samples
8.4.2 Urine samples
8.4.3 Saliva samples
8.4.4 Tissue samples
8.4.5 Others
8.5 By Technology
8.5.1 Real-time PCR
8.5.2 Digital PCR
8.5.3 Next-generation sequencing
8.5.4 Microfluidics
8.5.5 Others
8.6 By Distribution Channel
8.6.1 Direct sales
8.6.2 Distributors
8.6.3 Online sales
8.6.4 Others
8.7 By Region
8.7.1 North America
8.7.2 Europe
8.7.3 Asia-Pacific
8.7.4 Latin America
8.7.5 Middle East & Africa
9. Global Syndromic Multiplex Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Innovation Rate
9.2.8 Distribution Efficiency
9.2.9 Brand Recognition Score
9.2.10 Customer Satisfaction Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Abbott Laboratories
9.5.3 Thermo Fisher Scientific
9.5.4 BioMérieux
9.5.5 Cepheid
9.5.6 Hologic
9.5.7 Qiagen
9.5.8 Luminex Corporation
9.5.9 Becton, Dickinson and Company
9.5.10 Agilent Technologies
9.5.11 PerkinElmer
9.5.12 Siemens Healthineers
9.5.13 GenMark Diagnostics
9.5.14 Illumina
9.5.15 Eiken Chemical Co., Ltd.
10. Global Syndromic Multiplex Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for diagnostics
10.1.2 Decision-making processes
10.1.3 Preferred suppliers and vendors
10.1.4 Evaluation criteria for procurement
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in diagnostic infrastructure
10.2.2 Spending on technology upgrades
10.2.3 Budget for training and development
10.2.4 Allocation for research and development
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges faced by hospitals
10.3.2 Issues in diagnostic laboratories
10.3.3 Concerns of research institutions
10.3.4 Needs of point-of-care settings
10.4 User Readiness for Adoption
10.4.1 Awareness of multiplex diagnostics
10.4.2 Training requirements for staff
10.4.3 Infrastructure readiness
10.4.4 Financial readiness for investment
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of diagnostic accuracy
10.5.2 Cost savings analysis
10.5.3 Expansion into new applications
10.5.4 User feedback and improvement cycles
11. Global Syndromic Multiplex Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships and alliances
1.7 Risk assessment and mitigation
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategies
2.5 Digital marketing initiatives
2.6 Trade show participation
2.7 Customer engagement tactics
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales force deployment
3.5 Partnerships with healthcare providers
3.6 Logistics and supply chain management
3.7 Inventory management practices
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Discounting strategies
4.6 Bundling offers
4.7 Value-based pricing models
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends in diagnostics
5.4 Feedback from healthcare professionals
5.5 Future needs assessment
5.6 Innovation opportunities
5.7 Market entry barriers
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer feedback mechanisms
6.4 Relationship management strategies
6.5 Community engagement initiatives
6.6 Customer education programs
6.7 Retention strategies
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Cost-effectiveness of solutions
7.4 Quality assurance measures
7.5 Customer-centric approach
7.6 Innovation in product offerings
7.7 Long-term partnerships
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Training and development
8.5 Market research and analysis
8.6 Stakeholder engagement
8.7 Performance monitoring
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries selection
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for key activities
15.2.2 Milestone tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare organizations and market research firms
- Review of scientific literature and clinical studies on multiplex diagnostics
- Examination of regulatory guidelines and approvals from health authorities
Primary Research
- Interviews with key opinion leaders in the diagnostics and healthcare sectors
- Surveys with laboratory managers and technicians using multiplex diagnostic tools
- Field interviews with healthcare providers and specialists in infectious diseases
Validation & Triangulation
- Cross-validation of findings through multiple data sources including sales and usage statistics
- Triangulation of insights from primary interviews and secondary data analysis
- Sanity checks through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on global healthcare expenditure trends
- Segmentation by application areas such as infectious diseases, oncology, and genetic testing
- Incorporation of growth rates from emerging markets and technological advancements
Bottom-up Modeling
- Collection of sales data from leading multiplex diagnostic manufacturers
- Estimation of market share based on product types and geographic distribution
- Volume and pricing analysis based on laboratory usage and test frequency
Forecasting & Scenario Analysis
- Multi-variable forecasting using trends in disease prevalence and healthcare policies
- Scenario analysis based on potential market disruptions and technological innovations
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Infectious Disease Diagnostics | 120 | Clinical Pathologists, Laboratory Directors |
| Oncology Multiplex Testing | 100 | Oncologists, Genetic Counselors |
| Point-of-Care Testing | 80 | Healthcare Practitioners, Medical Technologists |
| Genetic Testing Services | 70 | Geneticists, Research Scientists |
| Regulatory Compliance in Diagnostics | 60 | Regulatory Affairs Specialists, Quality Assurance Managers |
Frequently Asked Questions
What is the current value of the Global Syndromic Multiplex Diagnostics Market?
The Global Syndromic Multiplex Diagnostics Market is valued at approximately USD 3 billion, reflecting a significant growth trend driven by the increasing prevalence of infectious diseases and advancements in diagnostic technologies.