About the Report
Base Year 2024Global Total Iron Binding Capacity Reagents Market Overview
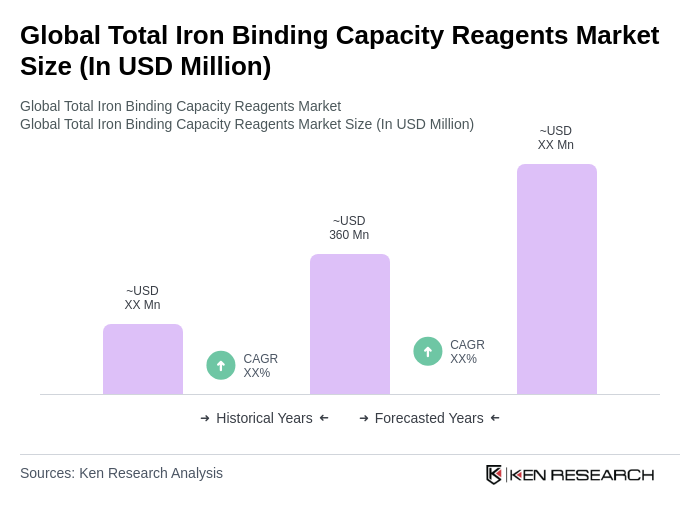
- The Global Total Iron Binding Capacity Reagents Market is valued at USD 360 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of iron deficiency anemia and the rising demand for diagnostic testing in clinical laboratories; global estimates consistently identify iron deficiency as the most common nutritional deficiency and a leading cause of anemia in women and children, underpinning sustained test volumes for iron studies including TIBC .
- Key regions dominating the market include North America and Europe, driven by advanced healthcare infrastructure, higher diagnostic testing penetration, and strong installed bases of automated clinical chemistry analyzers from major IVD vendors; Asia-Pacific is emerging due to increasing healthcare expenditure and greater awareness and screening for iron deficiency disorders in expanding hospital and independent lab networks .
- The U.S. Food and Drug Administration regulates TIBC reagents as in vitro diagnostic devices under existing 510(k) and labeling requirements for clinical chemistry assays; no specific new regulation mandating enhanced labeling and performance standards for iron binding capacity reagents was implemented in the cited period. Compliance continues to follow established IVD quality system regulation, 510(k) substantial equivalence pathways, and labeling standards applicable to chemistry assays .
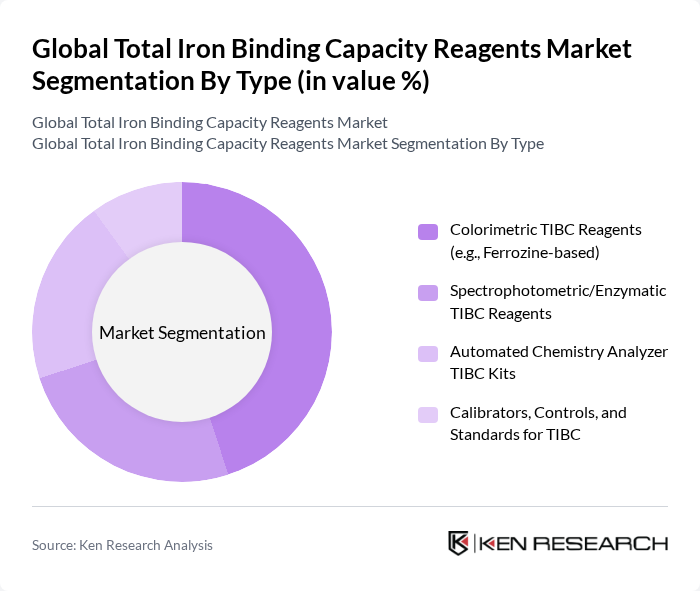
Global Total Iron Binding Capacity Reagents Market Segmentation
By Type:The market is segmented into various types of reagents used for measuring total iron binding capacity. The primary subsegments include Colorimetric TIBC Reagents (e.g., Ferrozine-based), Spectrophotometric/Enzymatic TIBC Reagents, Automated Chemistry Analyzer TIBC Kits, and Calibrators, Controls, and Standards for TIBC. Among these, Colorimetric TIBC Reagents are leading the market due to their widespread use in clinical laboratories for routine diagnostics, driven by their cost-effectiveness and ease of use .
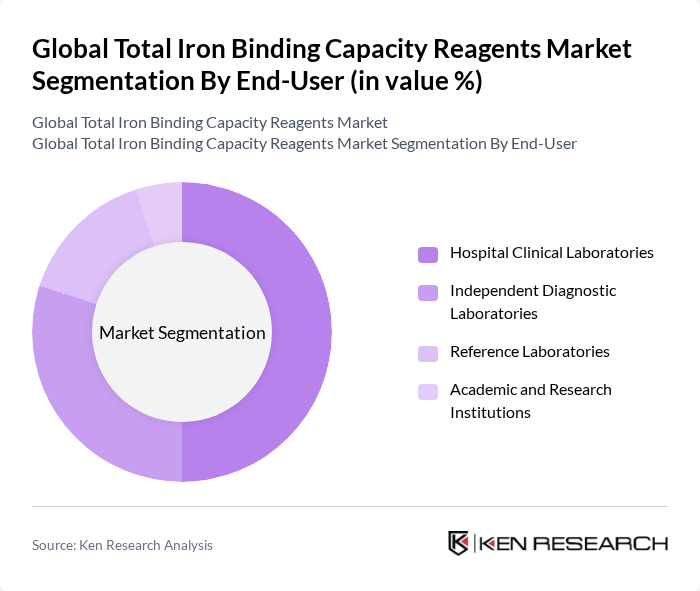
By End-User:The end-user segmentation includes Hospital Clinical Laboratories, Independent Diagnostic Laboratories, Reference Laboratories, and Academic and Research Institutions. Hospital Clinical Laboratories dominate this segment due to their high volume of routine testing and the need for reliable and efficient diagnostic solutions. The increasing number of hospitals and healthcare facilities globally further supports the growth of this subsegment .
Global Total Iron Binding Capacity Reagents Market Competitive Landscape
The Global Total Iron Binding Capacity Reagents Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Siemens Healthineers, Abbott Laboratories, Roche Diagnostics, Beckman Coulter, Inc., Bio-Rad Laboratories, Inc., Ortho Clinical Diagnostics (QuidelOrtho Corporation), Revvity, Inc. (formerly PerkinElmer, Inc.), Merck KGaA (MilliporeSigma), DiaSorin S.p.A., Mindray Bio-Medical Electronics Co., Ltd., Sysmex Corporation, FUJIFILM Wako Pure Chemical Corporation, Randox Laboratories Ltd, Sekisui Diagnostics, LLC, BioSystems S.A., DiaSys Diagnostic Systems GmbH, Sentinel Diagnostics S.r.l., Beckman Coulter Diagnostics (a Danaher company), Pointe Scientific, Inc. contribute to innovation, geographic expansion, and service delivery in this space .
Global Total Iron Binding Capacity Reagents Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Iron Deficiency Disorders:The World Health Organization reported that approximately 1.62 billion people globally are affected by anemia, with iron deficiency being the leading cause. In None, the prevalence of iron deficiency anemia is estimated at 20% among women of reproductive age, driving the demand for total iron binding capacity (TIBC) testing. This growing health concern necessitates the use of TIBC reagents, thereby propelling market growth significantly.
- Rising Demand for Diagnostic Testing in Healthcare:The global diagnostic testing market is projected to reach USD 70 billion in future, with a significant portion attributed to iron-related tests. In None, healthcare expenditure is expected to increase by 5% annually, leading to enhanced diagnostic capabilities. This surge in demand for accurate and timely diagnostic testing directly influences the uptake of TIBC reagents, fostering market expansion.
- Technological Advancements in Laboratory Testing:Innovations in laboratory technologies, such as automated analyzers and point-of-care testing devices, are transforming diagnostic processes. In None, the adoption of advanced testing technologies is projected to grow by 15% in future. These advancements enhance the efficiency and accuracy of TIBC testing, driving the demand for sophisticated reagents and contributing to market growth.
Market Challenges
- High Costs Associated with Advanced Testing Reagents:The cost of advanced TIBC reagents can be prohibitive, with prices ranging from USD 200 to USD 500 per kit. In None, healthcare budgets are constrained, limiting the adoption of these high-cost reagents. This financial barrier poses a significant challenge to market penetration, particularly in resource-limited settings where cost-effective solutions are essential.
- Stringent Regulatory Requirements:The regulatory landscape for diagnostic reagents is becoming increasingly stringent, with compliance costs rising. In None, the approval process for new TIBC reagents can take up to 24 months, delaying market entry. These regulatory hurdles can stifle innovation and limit the availability of new products, posing a challenge to market growth and competitiveness.
Global Total Iron Binding Capacity Reagents Market Future Outlook
The future of the TIBC reagents market appears promising, driven by ongoing advancements in diagnostic technologies and an increasing focus on preventive healthcare. As healthcare systems in None continue to evolve, the integration of digital solutions and point-of-care testing will likely enhance accessibility and efficiency. Furthermore, the growing emphasis on personalized medicine will create opportunities for tailored diagnostic solutions, fostering innovation and expanding the market landscape in the coming years.
Market Opportunities
- Expansion of Healthcare Infrastructure in Emerging Markets:The healthcare infrastructure in None is projected to grow by 10% annually, creating a favorable environment for TIBC reagent adoption. This expansion will facilitate better access to diagnostic services, increasing the demand for iron binding capacity testing and associated reagents.
- Development of Innovative and Cost-Effective Reagents:There is a significant opportunity for companies to develop cost-effective TIBC reagents that meet regulatory standards. With a growing focus on affordability in None, innovative solutions can capture market share, addressing the needs of healthcare providers and patients alike.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Colorimetric TIBC Reagents (e.g., Ferrozine-based) Spectrophotometric/Enzymatic TIBC Reagents Automated Chemistry Analyzer TIBC Kits Calibrators, Controls, and Standards for TIBC |
| By End-User | Hospital Clinical Laboratories Independent Diagnostic Laboratories Reference Laboratories Academic and Research Institutions |
| By Application | Routine Clinical Diagnostics (Anemia/Iron Disorders) Population Screening and Preventive Health Check-ups Research and Method Development Quality Control/Proficiency Testing |
| By Distribution Channel | Direct Sales (Manufacturers to Labs/Health Systems) Authorized Distributors/Dealers Group Purchasing Organizations (GPOs) and Tenders E?commerce and Procurement Platforms |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Packaging Type | Ready-to-Use Liquid Reagent Bottles Concentrates and Diluent Packs Controls/Calibrators in Vials Bulk/Institutional Packs |
| By Price Range | Economy (Value Tier) Mid-Priced Premium (High-Throughput/Automation-Ready) Contract/Payer-Negotiated Pricing |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers of Diagnostic Reagents
Clinical Laboratories and Diagnostic Centers
Pharmaceutical Companies
Healthcare Providers and Hospitals
Industry Associations and Trade Organizations
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Thermo Fisher Scientific Inc.
Siemens Healthineers
Abbott Laboratories
Roche Diagnostics
Beckman Coulter, Inc.
Bio-Rad Laboratories, Inc.
Ortho Clinical Diagnostics (QuidelOrtho Corporation)
Revvity, Inc. (formerly PerkinElmer, Inc.)
Merck KGaA (MilliporeSigma)
DiaSorin S.p.A.
Mindray Bio-Medical Electronics Co., Ltd.
Sysmex Corporation
FUJIFILM Wako Pure Chemical Corporation
Randox Laboratories Ltd
Sekisui Diagnostics, LLC
BioSystems S.A.
DiaSys Diagnostic Systems GmbH
Sentinel Diagnostics S.r.l.
Beckman Coulter Diagnostics (a Danaher company)
Pointe Scientific, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Total Iron Binding Capacity Reagents Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Total Iron Binding Capacity Reagents Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Total Iron Binding Capacity Reagents Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of iron deficiency disorders
3.1.2 Rising demand for diagnostic testing in healthcare
3.1.3 Technological advancements in laboratory testing
3.1.4 Growing awareness of preventive healthcare
3.2 Market Challenges
3.2.1 High costs associated with advanced testing reagents
3.2.2 Stringent regulatory requirements
3.2.3 Limited access to healthcare in developing regions
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure in emerging markets
3.3.2 Development of innovative and cost-effective reagents
3.3.3 Strategic partnerships with healthcare providers
3.3.4 Increasing focus on personalized medicine
3.4 Market Trends
3.4.1 Shift towards point-of-care testing
3.4.2 Integration of digital technologies in diagnostics
3.4.3 Growing emphasis on rapid testing solutions
3.4.4 Rising investments in research and development
3.5 Government Regulation
3.5.1 Compliance with international quality standards
3.5.2 Implementation of health and safety regulations
3.5.3 Guidelines for the approval of diagnostic reagents
3.5.4 Policies promoting access to healthcare diagnostics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Total Iron Binding Capacity Reagents Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Total Iron Binding Capacity Reagents Market Segmentation
8.1 By Type
8.1.1 Colorimetric TIBC Reagents (e.g., Ferrozine-based)
8.1.2 Spectrophotometric/Enzymatic TIBC Reagents
8.1.3 Automated Chemistry Analyzer TIBC Kits
8.1.4 Calibrators, Controls, and Standards for TIBC
8.2 By End-User
8.2.1 Hospital Clinical Laboratories
8.2.2 Independent Diagnostic Laboratories
8.2.3 Reference Laboratories
8.2.4 Academic and Research Institutions
8.3 By Application
8.3.1 Routine Clinical Diagnostics (Anemia/Iron Disorders)
8.3.2 Population Screening and Preventive Health Check-ups
8.3.3 Research and Method Development
8.3.4 Quality Control/Proficiency Testing
8.4 By Distribution Channel
8.4.1 Direct Sales (Manufacturers to Labs/Health Systems)
8.4.2 Authorized Distributors/Dealers
8.4.3 Group Purchasing Organizations (GPOs) and Tenders
8.4.4 E?commerce and Procurement Platforms
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.6 By Packaging Type
8.6.1 Ready-to-Use Liquid Reagent Bottles
8.6.2 Concentrates and Diluent Packs
8.6.3 Controls/Calibrators in Vials
8.6.4 Bulk/Institutional Packs
8.7 By Price Range
8.7.1 Economy (Value Tier)
8.7.2 Mid-Priced
8.7.3 Premium (High-Throughput/Automation-Ready)
8.7.4 Contract/Payer-Negotiated Pricing
9. Global Total Iron Binding Capacity Reagents Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name (e.g., Thermo Fisher Scientific; Siemens Healthineers; Abbott; Roche Diagnostics; Beckman Coulter; Randox Laboratories; Biosystems S.A.; Sekisui Diagnostics; DiaSys Diagnostic Systems; Sentinel Diagnostics)
9.2.2 Group Size (Global Large-cap; Mid-cap IVD; Specialized Reagent SME)
9.2.3 TIBC Reagents Revenue and Growth (3–5 year CAGR, USD)
9.2.4 Installed Analyzer Base Compatibility (number of chemistry analyzer models supported)
9.2.5 Geographic Footprint (countries served; regulatory approvals: FDA 510(k), CE-IVD, etc.)
9.2.6 Assay Performance KPIs (linearity range, precision/CV%, LoD, turnaround time)
9.2.7 Commercial KPIs (average selling price band, contract win rate/GPO penetration)
9.2.8 Distribution Network Coverage (number of distributors; direct vs indirect share)
9.2.9 Portfolio Breadth (TIBC with UIBC/Serum Iron panels; controls/calibrators availability)
9.2.10 Quality/Service Metrics (on-time delivery rate, complaint rate per 1,000 kits, service response time)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Thermo Fisher Scientific Inc.
9.5.2 Siemens Healthineers
9.5.3 Abbott Laboratories
9.5.4 Roche Diagnostics
9.5.5 Beckman Coulter, Inc.
9.5.6 Bio-Rad Laboratories, Inc.
9.5.7 Ortho Clinical Diagnostics (QuidelOrtho Corporation)
9.5.8 Revvity, Inc. (formerly PerkinElmer, Inc.)
9.5.9 Merck KGaA (MilliporeSigma)
9.5.10 DiaSorin S.p.A.
9.5.11 Mindray Bio-Medical Electronics Co., Ltd.
9.5.12 Sysmex Corporation
9.5.13 FUJIFILM Wako Pure Chemical Corporation
9.5.14 Randox Laboratories Ltd
9.5.15 Sekisui Diagnostics, LLC
9.5.16 BioSystems S.A.
9.5.17 DiaSys Diagnostic Systems GmbH
9.5.18 Sentinel Diagnostics S.r.l.
9.5.19 Beckman Coulter Diagnostics (a Danaher company)
9.5.20 Pointe Scientific, Inc.
10. Global Total Iron Binding Capacity Reagents Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government health departments
10.1.2 Public health organizations
10.1.3 Research funding agencies
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in laboratory infrastructure
10.2.2 Funding for diagnostic equipment
10.2.3 Budget allocation for research and development
10.3 Pain Point Analysis by End-User Category
10.3.1 Limited access to quality reagents
10.3.2 High costs of diagnostic tests
10.3.3 Delays in test results
10.4 User Readiness for Adoption
10.4.1 Training and education needs
10.4.2 Infrastructure readiness
10.4.3 Acceptance of new technologies
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of cost savings
10.5.2 Expansion into new diagnostic areas
10.5.3 Long-term partnerships with suppliers
11. Global Total Iron Binding Capacity Reagents Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape assessment
1.6 Key partnerships identification
1.7 Operational plan outline
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategy
2.5 Digital marketing approach
2.6 Trade show participation
2.7 Customer engagement tactics
3. Distribution Plan
3.1 Urban retail strategy
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales force deployment
3.5 Partnerships with distributors
3.6 Logistics and supply chain management
4. Channel & Pricing Gaps
4.1 Underserved routes analysis
4.2 Pricing bands evaluation
4.3 Competitor pricing comparison
4.4 Customer willingness to pay
4.5 Discount strategies
4.6 Bundling opportunities
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends exploration
5.4 Feedback from end-users
5.5 Future needs forecasting
6. Customer Relationship
6.1 Loyalty programs design
6.2 After-sales service strategies
6.3 Customer feedback mechanisms
6.4 Community engagement initiatives
6.5 Relationship management tools
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Cost-effectiveness analysis
7.4 Quality assurance measures
7.5 Innovation in product offerings
8. Key Activities
8.1 Regulatory compliance strategies
8.2 Branding initiatives
8.3 Distribution setup plans
8.4 Training and development programs
8.5 Performance monitoring systems
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries selection
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model evaluation
11. Capital and Timeline Estimation
11.1 Capital requirements analysis
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships analysis
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
14. Potential Partner List
14.1 Distributors identification
14.2 Joint Ventures opportunities
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone planning
15.2.2 Activity scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from global health organizations and market research firms
- Review of scientific literature on iron metabolism and total iron binding capacity
- Examination of regulatory frameworks and guidelines from health authorities regarding iron supplements
Primary Research
- Interviews with laboratory managers and quality control experts in clinical settings
- Surveys with healthcare professionals specializing in hematology and nutrition
- Focus groups with end-users, including patients and healthcare providers, to understand usage patterns
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and clinical studies
- Triangulation of market insights from primary interviews and secondary data analysis
- Sanity checks conducted through expert panels comprising industry veterans and academic researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on global healthcare expenditure and iron supplement sales
- Segmentation by geographical regions and healthcare sectors (hospitals, clinics, pharmacies)
- Incorporation of trends in anemia prevalence and public health initiatives promoting iron testing
Bottom-up Modeling
- Collection of sales data from leading manufacturers of total iron binding capacity reagents
- Estimation of market share based on product types (liquid, solid, diagnostic kits)
- Volume and pricing analysis based on historical sales data and projected growth rates
Forecasting & Scenario Analysis
- Multi-variable forecasting using factors such as population growth, dietary changes, and healthcare access
- Scenario analysis based on potential changes in healthcare policies and iron supplementation guidelines
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Managers, Clinical Pathologists |
| Healthcare Providers | 80 | General Practitioners, Hematologists |
| Pharmaceutical Distributors | 60 | Supply Chain Managers, Product Managers |
| Research Institutions | 50 | Research Scientists, Academic Professors |
| Patient Advocacy Groups | 40 | Patient Representatives, Health Educators |
Frequently Asked Questions
What is the current value of the Global Total Iron Binding Capacity Reagents Market?
The Global Total Iron Binding Capacity Reagents Market is valued at approximately USD 360 million, reflecting a significant demand driven by the increasing prevalence of iron deficiency anemia and the need for diagnostic testing in clinical laboratories.