About the Report
Base Year 2024Indonesia Herceptin Market Overview
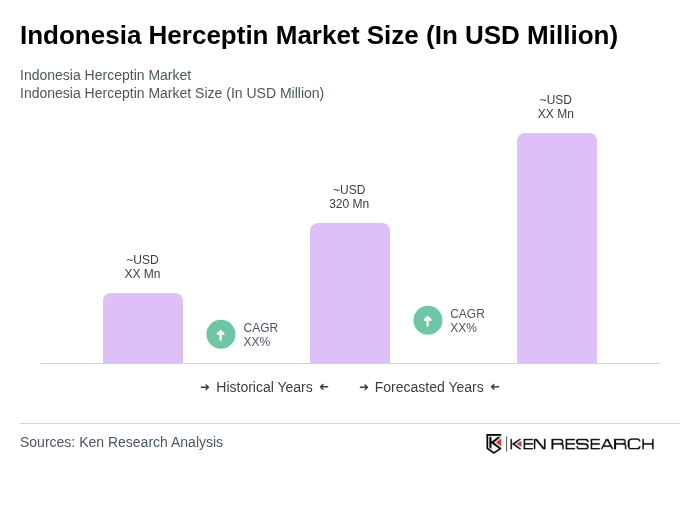
- The Indonesia Herceptin market is valued at USD 320 million, based on a five-year historical analysis, in line with regional benchmarks where Asia Pacific Herceptin revenues account for a substantial share of the global market. This growth is primarily driven by the increasing prevalence of HER2-positive breast cancer and the rising awareness of targeted therapies among healthcare professionals and patients, with breast cancer remaining the most common cancer in Indonesian women and HER2-positive disease representing a significant subset. The market is also supported by advancements in healthcare infrastructure, expansion of BPJS-Kesehatan coverage, and the increasing availability of innovative treatment options including trastuzumab biosimilars and subcutaneous fixed-dose combinations that improve convenience and affordability.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their advanced healthcare facilities, concentration of referral hospitals, and higher patient populations. Jakarta, being the capital, has the most comprehensive oncology services with national referral centers and teaching hospitals, while Surabaya and Bandung serve as significant healthcare hubs in East and West Java, respectively, attracting cancer patients from surrounding provinces for diagnosis, systemic therapy, and follow-up care.
- Trastuzumab has been included in the National Formulary and reimbursed under the National Health Insurance (Jaminan Kesehatan Nasional, JKN) scheme through BPJS-Kesehatan based on the Minister of Health Decree on National Formulary (Keputusan Menteri Kesehatan Republik Indonesia tentang Formularium Nasional) issued by the Ministry of Health, with subsequent updates aligning with oncology clinical guidelines to ensure access for HER2-positive breast cancer and gastric cancer patients. This regulatory framework aims to improve access to essential cancer treatments by mandating reimbursement of trastuzumab within INA-CBGs-based payment, thereby supporting broader utilization and better treatment outcomes in eligible patients.
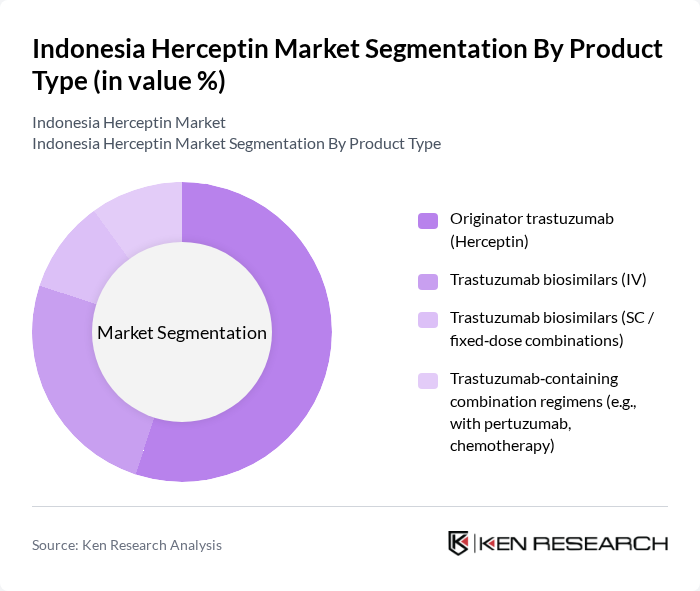
Indonesia Herceptin Market Segmentation
By Product Type:The product type segmentation includes various forms of trastuzumab, which cater to different patient needs and treatment protocols. The dominant sub-segment is the originator trastuzumab (Herceptin), which has historically maintained strong use due to its established efficacy, safety profile, and early availability in Indonesia. Trastuzumab biosimilars are gaining traction as they offer cost-effective alternatives and are supported by national and regional tenders, consistent with global trends where biosimilars now represent the largest share of Herceptin-class products in Asia Pacific. Subcutaneous and fixed-dose trastuzumab-based combinations are increasingly utilized in clinical practice because they shorten chair time, improve patient convenience, and facilitate combination regimens with agents such as pertuzumab and chemotherapy to enhance treatment outcomes.
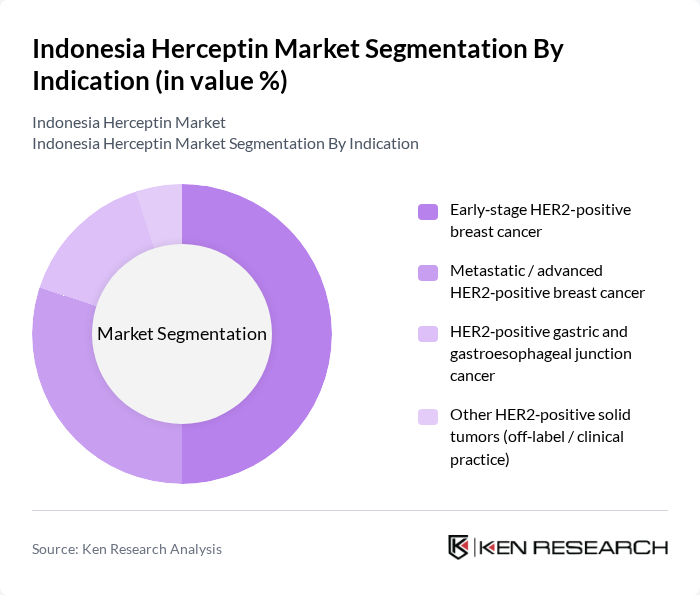
By Indication:The indication segmentation highlights the various cancer types treated with trastuzumab. The early-stage HER2-positive breast cancer sub-segment leads the market, reflecting international and Indonesian clinical practice where adjuvant trastuzumab is standard of care for eligible early-stage HER2-positive cases and breast cancer remains the primary indication for Herceptin globally. Metastatic HER2-positive breast cancer also represents a significant portion, as patients typically require prolonged or sequential lines of HER2-targeted therapy, often in combination with chemotherapy or other biologics. The inclusion of HER2-positive gastric and gastroesophageal junction cancer in international and regional treatment guidelines, aligned with trastuzumab’s labeled indications, further expands the market potential, while limited off-label use in other HER2-positive solid tumors remains a small but emerging segment.
Indonesia Herceptin Market Competitive Landscape
The Indonesia Herceptin market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Roche Indonesia (Roche / Genentech), PT Kalbe Farma Tbk (Kalbe Farma), PT Novartis Indonesia, PT Pfizer Indonesia, PT Merck Sharp & Dohme Pharma Tbk (MSD Indonesia), PT AstraZeneca Indonesia, Biocon Biologics Ltd. (including Ogivri / Canmab supply into Indonesia), Viatris Inc. (including Mylan?origin biosimilars portfolio), Sandoz (Indonesia operations / regional supply of trastuzumab biosimilars), PT Kimia Farma Tbk, PT Bio Farma (Persero), Celltrion Healthcare Co., Ltd. (Herzuma supplier to Indonesia), Amgen Inc. (regional HER2 biologics and biosimilars presence), Samsung Bioepis Co., Ltd. (Samfenet / trastuzumab biosimilar developer), PT Soho Global Health Tbk and other local oncology distributors contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Herceptin Market Industry Analysis
Growth Drivers
- Increasing Prevalence of HER2-positive Breast Cancer:The incidence of HER2-positive breast cancer in Indonesia is rising, with approximately 22,000 new cases reported annually. This increase is attributed to factors such as lifestyle changes and genetic predispositions. The World Health Organization (WHO) estimates that breast cancer accounts for 25% of all cancer cases in women in Indonesia, driving demand for targeted therapies like Herceptin. This growing patient population is a significant growth driver for the Herceptin market.
- Rising Healthcare Expenditure:Indonesia's healthcare expenditure is projected to reach IDR 1,600 trillion (approximately USD 104 billion) in future, reflecting a 7% increase from the previous period. This rise is fueled by government initiatives to improve healthcare access and quality. Increased spending on healthcare services and pharmaceuticals enhances the affordability and availability of Herceptin, thereby supporting market growth. The government’s commitment to healthcare reform is expected to further bolster this trend.
- Growing Awareness and Early Diagnosis:Awareness campaigns and educational programs have significantly improved early diagnosis rates of breast cancer in Indonesia. In future, it is estimated that 65% of breast cancer cases will be diagnosed at an early stage, compared to 45% in previous years. This shift is largely due to increased public health initiatives and the establishment of screening programs. Early diagnosis leads to higher treatment rates with Herceptin, driving market growth as patients seek effective therapies.
Market Challenges
- High Cost of Treatment:The cost of Herceptin treatment in Indonesia can exceed IDR 35 million (approximately USD 2,300) per patient per year, which poses a significant barrier for many patients. With the average annual income in Indonesia being around IDR 70 million (USD 4,600), affordability remains a critical issue. This high cost limits access to treatment, particularly for low-income populations, thereby hindering market growth and patient outcomes.
- Limited Access in Rural Areas:Approximately 60% of Indonesia's population resides in rural areas, where healthcare infrastructure is often inadequate. Many rural regions lack access to specialized cancer treatment facilities, making it difficult for patients to receive Herceptin. The Indonesian Ministry of Health reports that only 35% of rural health centers are equipped to handle cancer treatments. This disparity in access creates a significant challenge for the Herceptin market, limiting its reach and potential growth.
Indonesia Herceptin Market Future Outlook
The future of the Herceptin market in Indonesia appears promising, driven by advancements in healthcare infrastructure and increasing government support for cancer treatment initiatives. As the population becomes more health-conscious, the demand for targeted therapies is expected to rise. Additionally, the integration of telemedicine and digital health solutions will enhance patient access to treatment. These trends indicate a robust growth trajectory for the Herceptin market, with potential for innovative treatment approaches and improved patient outcomes.
Market Opportunities
- Increasing Demand for Personalized Medicine:The shift towards personalized medicine presents a significant opportunity for the Herceptin market. With advancements in genetic testing, more patients can be identified as suitable candidates for Herceptin therapy. This trend is expected to enhance treatment efficacy and patient satisfaction, driving market growth as healthcare providers increasingly adopt personalized treatment plans.
- Collaborations with Local Healthcare Providers:Forming partnerships with local healthcare providers can enhance distribution and accessibility of Herceptin. Collaborations can facilitate better patient education and support services, improving treatment adherence. By leveraging local expertise and networks, pharmaceutical companies can expand their market presence and effectively address the unique challenges faced by Indonesian patients.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Originator trastuzumab (Herceptin) Trastuzumab biosimilars (IV) Trastuzumab biosimilars (SC / fixed?dose combinations) Trastuzumab?containing combination regimens (e.g., with pertuzumab, chemotherapy) |
| By Indication | Early?stage HER2?positive breast cancer Metastatic / advanced HER2?positive breast cancer HER2?positive gastric and gastroesophageal junction cancer Other HER2?positive solid tumors (off?label / clinical practice) |
| By Payer / Reimbursement | National Health Insurance (JKN/BPJS?Kesehatan) funded Private insurance funded Out?of?pocket / self?pay Charity / NGO and hospital assistance programs |
| By Distribution Channel | Hospital pharmacies (public) Hospital pharmacies (private) Specialty / oncology pharmacies Tender and centralized procurement channels |
| By Treatment Setting | Tertiary referral hospitals (RSUP / national centers) Secondary / provincial and district hospitals Private oncology centers Day?care infusion and ambulatory clinics |
| By Geographic Region | Java (including Jakarta) Sumatra Kalimantan Sulawesi, Bali, Nusa Tenggara, Papua & other islands |
| By Line of Therapy | Neoadjuvant therapy Adjuvant therapy First?line metastatic therapy Later?line metastatic therapy |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Badan Pengawas Obat dan Makanan - BPOM)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Hospitals
Industry Associations (e.g., Indonesian Pharmaceutical Association)
Health Insurance Companies
Players Mentioned in the Report:
PT Roche Indonesia (Roche / Genentech)
PT Kalbe Farma Tbk (Kalbe Farma)
PT Novartis Indonesia
PT Pfizer Indonesia
PT Merck Sharp & Dohme Pharma Tbk (MSD Indonesia)
PT AstraZeneca Indonesia
Biocon Biologics Ltd. (including Ogivri / Canmab supply into Indonesia)
Viatris Inc. (including Mylanorigin biosimilars portfolio)
Sandoz (Indonesia operations / regional supply of trastuzumab biosimilars)
PT Kimia Farma Tbk
PT Bio Farma (Persero)
Celltrion Healthcare Co., Ltd. (Herzuma supplier to Indonesia)
Amgen Inc. (regional HER2 biologics and biosimilars presence)
Samsung Bioepis Co., Ltd. (Samfenet / trastuzumab biosimilar developer)
PT Soho Global Health Tbk and other local oncology distributors
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Herceptin Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Herceptin Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Herceptin Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of HER2-positive breast cancer
3.1.2 Rising healthcare expenditure
3.1.3 Growing awareness and early diagnosis
3.1.4 Expansion of healthcare infrastructure
3.2 Market Challenges
3.2.1 High cost of treatment
3.2.2 Limited access in rural areas
3.2.3 Regulatory hurdles
3.2.4 Competition from biosimilars
3.3 Market Opportunities
3.3.1 Increasing demand for personalized medicine
3.3.2 Collaborations with local healthcare providers
3.3.3 Expansion into untapped markets
3.3.4 Investment in research and development
3.4 Market Trends
3.4.1 Shift towards targeted therapies
3.4.2 Growth of telemedicine
3.4.3 Rising patient advocacy and support groups
3.4.4 Increased focus on patient-centric care
3.5 Government Regulation
3.5.1 National Health Insurance Program (JKN)
3.5.2 Drug registration and approval processes
3.5.3 Pricing regulations for pharmaceuticals
3.5.4 Import regulations for medical products
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Herceptin Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Herceptin Market Segmentation
8.1 By Product Type
8.1.1 Originator trastuzumab (Herceptin)
8.1.2 Trastuzumab biosimilars (IV)
8.1.3 Trastuzumab biosimilars (SC / fixed?dose combinations)
8.1.4 Trastuzumab?containing combination regimens (e.g., with pertuzumab, chemotherapy)
8.2 By Indication
8.2.1 Early?stage HER2?positive breast cancer
8.2.2 Metastatic / advanced HER2?positive breast cancer
8.2.3 HER2?positive gastric and gastroesophageal junction cancer
8.2.4 Other HER2?positive solid tumors (off?label / clinical practice)
8.3 By Payer / Reimbursement
8.3.1 National Health Insurance (JKN/BPJS?Kesehatan) funded
8.3.2 Private insurance funded
8.3.3 Out?of?pocket / self?pay
8.3.4 Charity / NGO and hospital assistance programs
8.4 By Distribution Channel
8.4.1 Hospital pharmacies (public)
8.4.2 Hospital pharmacies (private)
8.4.3 Specialty / oncology pharmacies
8.4.4 Tender and centralized procurement channels
8.5 By Treatment Setting
8.5.1 Tertiary referral hospitals (RSUP / national centers)
8.5.2 Secondary / provincial and district hospitals
8.5.3 Private oncology centers
8.5.4 Day?care infusion and ambulatory clinics
8.6 By Geographic Region
8.6.1 Java (including Jakarta)
8.6.2 Sumatra
8.6.3 Kalimantan
8.6.4 Sulawesi, Bali, Nusa Tenggara, Papua & other islands
8.7 By Line of Therapy
8.7.1 Neoadjuvant therapy
8.7.2 Adjuvant therapy
8.7.3 First?line metastatic therapy
8.7.4 Later?line metastatic therapy
9. Indonesia Herceptin Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Product portfolio breadth in HER2?targeted therapies (originator vs biosimilars)
9.2.2 Indonesia Herceptin / trastuzumab revenue and revenue growth (last 3–5 years)
9.2.3 Volume share in JKN/BPJS?Kesehatan tenders and hospital procurement
9.2.4 Average treatment cost per patient (Herceptin and biosimilars)
9.2.5 Geographic coverage (number of provinces / hospitals served)
9.2.6 Market access strength (formulary listings, INA?CBGs inclusion, tender wins)
9.2.7 Pricing and discount strategy in public and private segments
9.2.8 Local manufacturing / fill?finish and partnership footprint
9.2.9 Regulatory and approval status of trastuzumab products in Indonesia
9.2.10 Investment in clinical evidence, real?world data, and medical education in Indonesia
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT Roche Indonesia (Roche / Genentech)
9.5.2 PT Kalbe Farma Tbk (Kalbe Farma)
9.5.3 PT Novartis Indonesia
9.5.4 PT Pfizer Indonesia
9.5.5 PT Merck Sharp & Dohme Pharma Tbk (MSD Indonesia)
9.5.6 PT AstraZeneca Indonesia
9.5.7 Biocon Biologics Ltd. (including Ogivri / Canmab supply into Indonesia)
9.5.8 Viatris Inc. (including Mylan?origin biosimilars portfolio)
9.5.9 Sandoz (Indonesia operations / regional supply of trastuzumab biosimilars)
9.5.10 PT Kimia Farma Tbk
9.5.11 PT Bio Farma (Persero)
9.5.12 Celltrion Healthcare Co., Ltd. (Herzuma supplier to Indonesia)
9.5.13 Amgen Inc. (regional HER2 biologics and biosimilars presence)
9.5.14 Samsung Bioepis Co., Ltd. (Samfenet / trastuzumab biosimilar developer)
9.5.15 PT Soho Global Health Tbk and other local oncology distributors
10. Indonesia Herceptin Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for cancer treatment
10.1.2 Procurement processes and timelines
10.1.3 Collaboration with healthcare providers
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in oncology facilities
10.2.2 Funding for research initiatives
10.2.3 Partnerships with NGOs
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to treatment
10.3.2 Affordability of medications
10.3.3 Quality of care
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of HER2-positive breast cancer
10.4.2 Acceptance of new treatment protocols
10.4.3 Training for healthcare professionals
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of treatment outcomes
10.5.2 Patient feedback mechanisms
10.5.3 Expansion into new patient demographics
10.5.4 Others
11. Indonesia Herceptin Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Operational framework design
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategies
2.5 Digital marketing initiatives
2.6 Event marketing opportunities
2.7 Feedback and adaptation mechanisms
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 E-commerce integration
3.4 Logistics and supply chain management
3.5 Distribution partnerships
3.6 Inventory management practices
3.7 Performance metrics for distribution
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Value-based pricing models
4.6 Discount and promotion strategies
4.7 Pricing adjustments based on market feedback
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends and needs
5.4 Product development opportunities
5.5 Market entry strategies for unmet needs
5.6 Collaboration opportunities for innovation
5.7 Feedback loops for continuous improvement
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer engagement strategies
6.4 Feedback collection mechanisms
6.5 Relationship management tools
6.6 Community building initiatives
6.7 Customer education programs
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling points of Herceptin
7.4 Patient-centric approaches
7.5 Cost-effectiveness analysis
7.6 Long-term health benefits
7.7 Partnerships for enhanced value
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Market research and analysis
8.5 Training and development
8.6 Stakeholder engagement
8.7 Performance monitoring
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band strategies
9.1.3 Packaging innovations
9.2 Export Entry Strategy
9.2.1 Target countries analysis
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers and Acquisitions
10.4 Distributor Model
10.5 Risk assessment for each mode
10.6 Strategic fit evaluation
10.7 Long-term sustainability considerations
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Financial projections
11.4 Funding sources identification
11.5 Budget allocation strategies
11.6 Risk management plans
11.7 Milestones for evaluation
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
12.3 Risk mitigation strategies
12.4 Control mechanisms
12.5 Long-term strategic alignment
12.6 Performance metrics for partnerships
12.7 Exit strategies
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin projections
13.4 Cost management strategies
13.5 Revenue diversification opportunities
13.6 Market share growth potential
13.7 Financial health indicators
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisitio
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health organizations and pharmaceutical associations in Indonesia
- Review of published articles and journals focusing on Herceptin usage and breast cancer treatment trends
- Examination of government health statistics and pharmaceutical sales data from the Ministry of Health
Primary Research
- Interviews with oncologists and healthcare professionals specializing in breast cancer treatment
- Surveys conducted with hospital procurement departments regarding Herceptin purchasing patterns
- Focus group discussions with patients undergoing treatment to understand their experiences and preferences
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and secondary data sources
- Triangulation of market data with clinical trial results and treatment guidelines
- Sanity checks through feedback from a panel of healthcare industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total breast cancer cases in Indonesia and the percentage eligible for Herceptin treatment
- Analysis of historical sales data to identify growth trends in the Herceptin market
- Incorporation of demographic factors and healthcare access in urban vs. rural settings
Bottom-up Modeling
- Collection of data on Herceptin pricing from hospitals and pharmacies across major cities
- Estimation of treatment cycles per patient and average duration of therapy
- Calculation of total market value based on patient population and treatment frequency
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as healthcare policy changes and drug accessibility
- Scenario modeling based on potential market disruptions, including new entrants and generics
- Baseline, optimistic, and pessimistic forecasts for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists in Major Hospitals | 60 | Medical Oncologists, Breast Cancer Specialists |
| Pharmacy Managers in Urban Areas | 50 | Pharmacy Directors, Procurement Managers |
| Patients Undergoing Herceptin Treatment | 75 | Breast Cancer Patients, Caregivers |
| Healthcare Policy Makers | 40 | Health Ministry Officials, Regulatory Bodies |
| Clinical Trial Coordinators | 45 | Clinical Researchers, Trial Managers |
Frequently Asked Questions
What is the current value of the Indonesia Herceptin market?
The Indonesia Herceptin market is valued at approximately USD 320 million, reflecting a significant share of the global market, driven by the increasing prevalence of HER2-positive breast cancer and advancements in healthcare infrastructure.