About the Report
Base Year 2024Saudi Arabia Herceptin Market Overview
- The Saudi Arabia Herceptin market is valued at USD 30–40 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of HER2-positive breast cancer and the rising adoption of targeted therapies among healthcare providers. The market is also supported by advancements in healthcare infrastructure and increased awareness regarding early cancer detection and treatment options.
- Key cities dominating the market include Riyadh, Jeddah, and Dammam. Riyadh serves as the capital and a major healthcare hub, hosting numerous specialized oncology centers. Jeddah, with its strategic port, facilitates the import of pharmaceuticals, while Dammam benefits from its proximity to oil wealth, enabling investments in healthcare facilities and services.
- In recent years, the Saudi Food and Drug Authority (SFDA) has streamlined and clarified biosimilar approval requirements to improve access and affordability. This initiative aims to enhance patient access to affordable treatment options while ensuring safety and efficacy, thereby promoting competition and innovation in the market.
Saudi Arabia Herceptin Market Segmentation
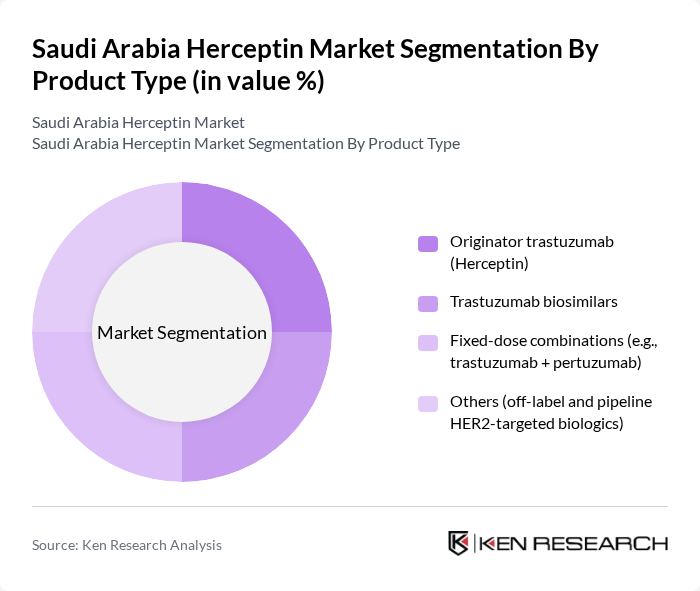
By Product Type:The product type segmentation includes various forms of trastuzumab, which cater to different patient needs and treatment protocols. The subsegments are Originator trastuzumab (Herceptin), Trastuzumab biosimilars, Fixed-dose combinations (e.g., trastuzumab + pertuzumab), and Others (off-label and pipeline HER2-targeted biologics). The market is primarily driven by the originator trastuzumab, which remains the preferred choice among oncologists due to its established efficacy and safety profile.
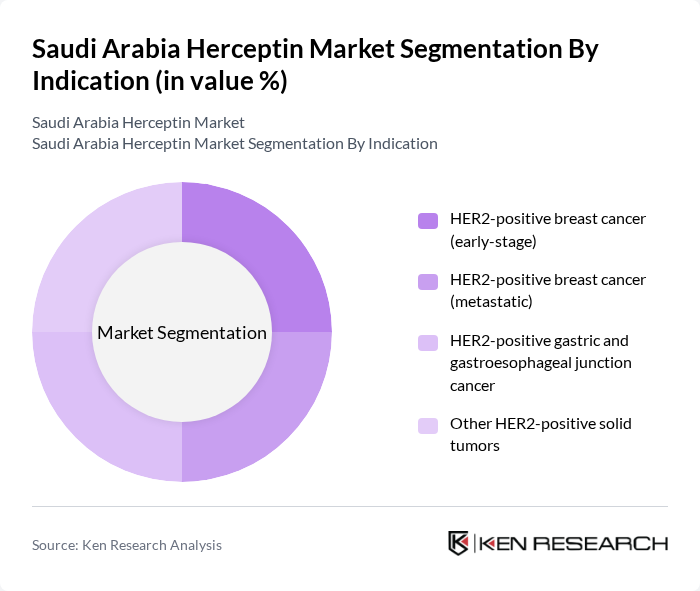
By Indication:The indication segmentation focuses on the specific types of cancers treated with trastuzumab. The subsegments include HER2-positive breast cancer (early-stage), HER2-positive breast cancer (metastatic), HER2-positive gastric and gastroesophageal junction cancer, and Other HER2-positive solid tumors. The HER2-positive breast cancer (metastatic) segment is the largest due to the high incidence of advanced-stage breast cancer and the effectiveness of trastuzumab in improving patient outcomes.
Saudi Arabia Herceptin Market Competitive Landscape
The Saudi Arabia Herceptin Market is characterized by a dynamic mix of regional and international players. Leading participants such as F. Hoffmann-La Roche Ltd (Herceptin originator; Phesgo), Celltrion Healthcare Co., Ltd, Samsung Bioepis Co., Ltd, Amgen Inc. (including biosimilar portfolio), Biocon Biologics Ltd, Mylan / Viatris Inc., Sandoz International GmbH, Pfizer Inc., Hikma Pharmaceuticals plc, Dr. Reddy’s Laboratories Ltd, STADA Arzneimittel AG, Accord Healthcare Ltd, Julphar (Gulf Pharmaceutical Industries), Tabuk Pharmaceuticals Manufacturing Company, Jamjoom Pharma contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Herceptin Market Industry Analysis
Growth Drivers
- Increasing Prevalence of HER2-positive Breast Cancer:The incidence of HER2-positive breast cancer in Saudi Arabia is rising, with approximately 20% of breast cancer cases classified as HER2-positive. In future, an estimated 20,000 new breast cancer cases are expected, leading to a growing demand for Herceptin. This increase is driven by factors such as lifestyle changes and genetic predispositions, necessitating effective treatment options to manage this aggressive cancer type.
- Rising Healthcare Expenditure:Saudi Arabia's healthcare expenditure is projected to reach SAR 250 billion (approximately USD 66 billion) in future, reflecting a commitment to improving healthcare services. This increase in funding supports the procurement of advanced cancer therapies, including Herceptin. Enhanced healthcare budgets facilitate better access to innovative treatments, ultimately improving patient outcomes and driving market growth in oncology.
- Growing Awareness and Early Detection Initiatives:The Saudi government has launched various awareness campaigns aimed at early detection of breast cancer, significantly impacting treatment outcomes. In future, over 1.5 million women are expected to participate in screening programs, leading to earlier diagnoses. This proactive approach not only increases the demand for Herceptin but also emphasizes the importance of timely intervention in managing HER2-positive breast cancer effectively.
Market Challenges
- High Cost of Herceptin:The cost of Herceptin remains a significant barrier to access, with annual treatment expenses exceeding SAR 120,000 (approximately USD 31,800). This high price limits affordability for many patients, particularly in lower-income segments. As healthcare systems strive to balance cost and quality, the financial burden of Herceptin poses a challenge to widespread adoption and treatment accessibility in Saudi Arabia.
- Limited Access in Rural Areas:Access to Herceptin is particularly challenging in rural regions of Saudi Arabia, where healthcare facilities are often under-resourced. Approximately 35% of the population resides in these areas, facing difficulties in obtaining specialized cancer treatments. This disparity in healthcare access exacerbates health inequalities and hinders the effective management of HER2-positive breast cancer, limiting the overall market potential for Herceptin.
Saudi Arabia Herceptin Market Future Outlook
The Saudi Arabia Herceptin market is poised for significant developments driven by advancements in healthcare infrastructure and increasing public-private partnerships. As the government invests in modernizing healthcare facilities, access to innovative cancer treatments will improve. Additionally, the integration of telemedicine and digital health solutions is expected to enhance patient engagement and monitoring, ultimately leading to better treatment adherence and outcomes for HER2-positive breast cancer patients in the region.
Market Opportunities
- Expansion of Healthcare Infrastructure:The ongoing expansion of healthcare facilities in Saudi Arabia presents a significant opportunity for Herceptin. With over 60 new hospitals planned in future, improved access to oncology services will facilitate the distribution of Herceptin, enhancing treatment availability for patients across the country.
- Collaborations with Local Healthcare Providers:Forming strategic partnerships with local healthcare providers can enhance the distribution and accessibility of Herceptin. Collaborations can lead to tailored treatment programs and educational initiatives, ultimately improving patient outcomes and fostering a supportive environment for HER2-positive breast cancer management in Saudi Arabia.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Originator trastuzumab (Herceptin) Trastuzumab biosimilars Fixed-dose combinations (e.g., trastuzumab + pertuzumab) Others (off?label and pipeline HER2?targeted biologics) |
| By Indication | HER2-positive breast cancer (early-stage) HER2-positive breast cancer (metastatic) HER2-positive gastric and gastroesophageal junction cancer Other HER2-positive solid tumors |
| By Route of Administration | Intravenous (IV) Subcutaneous (SC) |
| By Distribution Channel | Hospital pharmacies Specialty and oncology pharmacies Retail and online pharmacies Government tender and centralized procurement |
| By Type of Facility | Public tertiary hospitals Private hospitals and oncology centers Military and security forces hospitals Research and academic centers |
| By Region | Riyadh Region Makkah Region (including Jeddah) Eastern Province (including Dammam and Khobar) Other regions |
| By Line of Therapy | First-line therapy Second-line therapy Third-line and beyond |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Pharmaceutical Manufacturers
Healthcare Providers and Hospitals
Distributors and Wholesalers
Biotechnology Firms
Health Insurance Companies
Pharmaceutical Trade Associations
Players Mentioned in the Report:
F. Hoffmann-La Roche Ltd (Herceptin originator; Phesgo)
Celltrion Healthcare Co., Ltd
Samsung Bioepis Co., Ltd
Amgen Inc. (including biosimilar portfolio)
Biocon Biologics Ltd
Mylan / Viatris Inc.
Sandoz International GmbH
Pfizer Inc.
Hikma Pharmaceuticals plc
Dr. Reddys Laboratories Ltd
STADA Arzneimittel AG
Accord Healthcare Ltd
Julphar (Gulf Pharmaceutical Industries)
Tabuk Pharmaceuticals Manufacturing Company
Jamjoom Pharma
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Herceptin Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Herceptin Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Herceptin Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of HER2-positive breast cancer
3.1.2 Rising healthcare expenditure
3.1.3 Growing awareness and early detection initiatives
3.1.4 Advancements in treatment protocols
3.2 Market Challenges
3.2.1 High cost of Herceptin
3.2.2 Limited access in rural areas
3.2.3 Regulatory hurdles
3.2.4 Competition from biosimilars
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Collaborations with local healthcare providers
3.3.3 Increasing investment in cancer research
3.3.4 Potential for personalized medicine
3.4 Market Trends
3.4.1 Shift towards targeted therapies
3.4.2 Growth of telemedicine in oncology
3.4.3 Rising demand for patient-centric care
3.4.4 Integration of digital health solutions
3.5 Government Regulation
3.5.1 Pricing controls on pharmaceuticals
3.5.2 Approval processes for new treatments
3.5.3 Guidelines for clinical trials
3.5.4 Policies promoting local manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Herceptin Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Herceptin Market Segmentation
8.1 By Product Type
8.1.1 Originator trastuzumab (Herceptin)
8.1.2 Trastuzumab biosimilars
8.1.3 Fixed-dose combinations (e.g., trastuzumab + pertuzumab)
8.1.4 Others (off?label and pipeline HER2?targeted biologics)
8.2 By Indication
8.2.1 HER2-positive breast cancer (early-stage)
8.2.2 HER2-positive breast cancer (metastatic)
8.2.3 HER2-positive gastric and gastroesophageal junction cancer
8.2.4 Other HER2-positive solid tumors
8.3 By Route of Administration
8.3.1 Intravenous (IV)
8.3.2 Subcutaneous (SC)
8.4 By Distribution Channel
8.4.1 Hospital pharmacies
8.4.2 Specialty and oncology pharmacies
8.4.3 Retail and online pharmacies
8.4.4 Government tender and centralized procurement
8.5 By Type of Facility
8.5.1 Public tertiary hospitals
8.5.2 Private hospitals and oncology centers
8.5.3 Military and security forces hospitals
8.5.4 Research and academic centers
8.6 By Region
8.6.1 Riyadh Region
8.6.2 Makkah Region (including Jeddah)
8.6.3 Eastern Province (including Dammam and Khobar)
8.6.4 Other regions
8.7 By Line of Therapy
8.7.1 First-line therapy
8.7.2 Second-line therapy
8.7.3 Third-line and beyond
9. Saudi Arabia Herceptin Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name
9.2.2 Product portfolio in trastuzumab and HER2-targeted therapies (originator vs biosimilars)
9.2.3 Saudi Arabia trastuzumab revenue and 5-year CAGR
9.2.4 Volume share in HER2-positive treatment lines (by indication)
9.2.5 Market access breadth (formulary listings and SHC/cluster coverage)
9.2.6 Share of sales via government tenders vs private sector
9.2.7 Average treatment cost per patient per cycle
9.2.8 Price discount vs originator (for biosimilar players)
9.2.9 Clinical and real-world evidence footprint in Saudi Arabia
9.2.10 Distribution and cold-chain footprint within the Kingdom
9.2.11 Partnership footprint with local distributors and hospitals
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 F. Hoffmann-La Roche Ltd (Herceptin originator; Phesgo)
9.5.2 Celltrion Healthcare Co., Ltd
9.5.3 Samsung Bioepis Co., Ltd
9.5.4 Amgen Inc. (including biosimilar portfolio)
9.5.5 Biocon Biologics Ltd
9.5.6 Mylan / Viatris Inc.
9.5.7 Sandoz International GmbH
9.5.8 Pfizer Inc.
9.5.9 Hikma Pharmaceuticals plc
9.5.10 Dr. Reddy’s Laboratories Ltd
9.5.11 STADA Arzneimittel AG
9.5.12 Accord Healthcare Ltd
9.5.13 Julphar (Gulf Pharmaceutical Industries)
9.5.14 Tabuk Pharmaceuticals Manufacturing Company
9.5.15 Jamjoom Pharma
10. Saudi Arabia Herceptin Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Finance
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare facilities
10.2.2 Research and development
10.2.3 Training and education
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Patients
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness levels
10.4.2 Training needs
10.4.3 Infrastructure readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost savings
10.5.2 Treatment outcomes
10.5.3 Patient satisfaction
10.5.4 Others
11. Saudi Arabia Herceptin Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare authorities in Saudi Arabia
- Review of published articles and journals on Herceptin usage and trends
- Examination of demographic and epidemiological data related to breast cancer in the region
Primary Research
- Interviews with oncologists and healthcare professionals specializing in breast cancer treatment
- Surveys with hospital administrators regarding Herceptin procurement and usage
- Focus groups with patients to understand treatment experiences and preferences
Validation & Triangulation
- Cross-validation of findings with data from pharmaceutical sales reports
- Triangulation of insights from healthcare professionals and patient feedback
- Sanity checks through expert panel reviews comprising oncologists and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total breast cancer cases in Saudi Arabia and Herceptin's market share
- Analysis of healthcare expenditure trends and their impact on Herceptin sales
- Incorporation of government healthcare initiatives promoting cancer treatment
Bottom-up Modeling
- Volume estimates based on the number of patients receiving Herceptin annually
- Cost analysis of Herceptin treatment regimens across various healthcare settings
- Assessment of patient adherence rates and treatment cycles for accurate revenue projections
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors like population growth and healthcare access
- Scenario modeling based on potential changes in healthcare policies and drug pricing
- Baseline, optimistic, and pessimistic forecasts for Herceptin market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists in Major Hospitals | 120 | Medical Oncologists, Clinical Researchers |
| Pharmacy Directors in Healthcare Facilities | 90 | Pharmacy Managers, Procurement Officers |
| Patients Undergoing Herceptin Treatment | 100 | Breast Cancer Patients, Patient Advocates |
| Healthcare Policy Makers | 60 | Health Ministry Officials, Regulatory Bodies |
| Insurance Providers | 70 | Health Insurance Analysts, Claims Managers |
Frequently Asked Questions
What is the current value of the Herceptin market in Saudi Arabia?
The Saudi Arabia Herceptin market is valued between USD 30 million and USD 40 million, driven by the increasing prevalence of HER2-positive breast cancer and the adoption of targeted therapies among healthcare providers.