Region:Asia
Author(s):Rebecca
Product Code:KRAD4934
Pages:83
Published On:December 2025
By Type of Plastic:The market is segmented into various types of plastics used in medical 3D printing, including standard thermoplastics, engineering-grade thermoplastics, photopolymer resins, bioresorbable and bio-based medical polymers, and others. Each type serves specific applications and has unique properties that cater to the diverse needs of the medical industry, in line with global medical 3D printing plastics usage patterns.
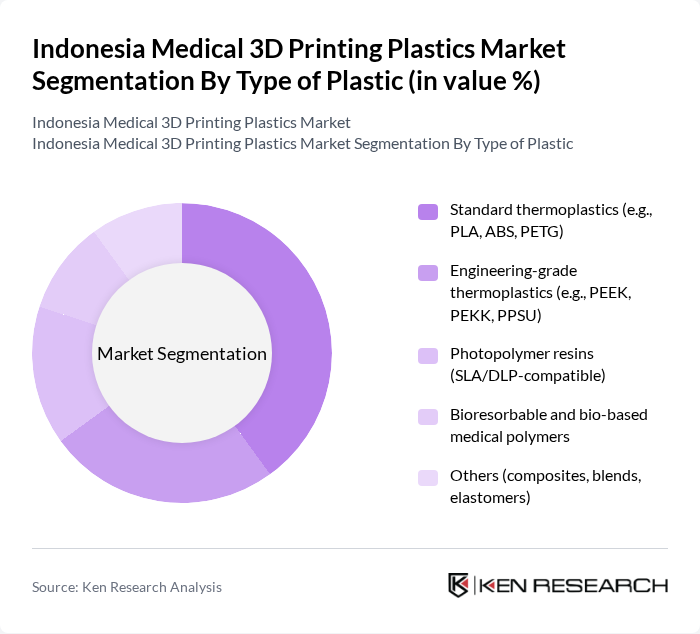
The leading subsegment in the type of plastic category is standard thermoplastics, particularly PLA and ABS, due to their versatility, ease of use, and cost-effectiveness for FDM/FFF printers, which dominate installed 3D printing capacity in Indonesia. These materials are widely adopted for producing surgical guides, anatomical models, training aids, and certain non-implantable medical devices. The growing trend towards sustainable materials and bio-based polymers has also boosted the demand for bio-based options, while engineering-grade thermoplastics such as PEEK and PEKK gain traction for high-performance medical applications requiring sterilizability and biocompatibility.
By End-User:The market is segmented based on end-users, including public hospitals, private hospitals and specialty clinics, dental laboratories and dental chains, medical device manufacturers and service bureaus, academic and research institutions, and others. Each end-user category has distinct requirements and applications for 3D printing technologies, broadly consistent with the way healthcare is treated as a key end-use sector in Indonesia’s 3D printing materials market.
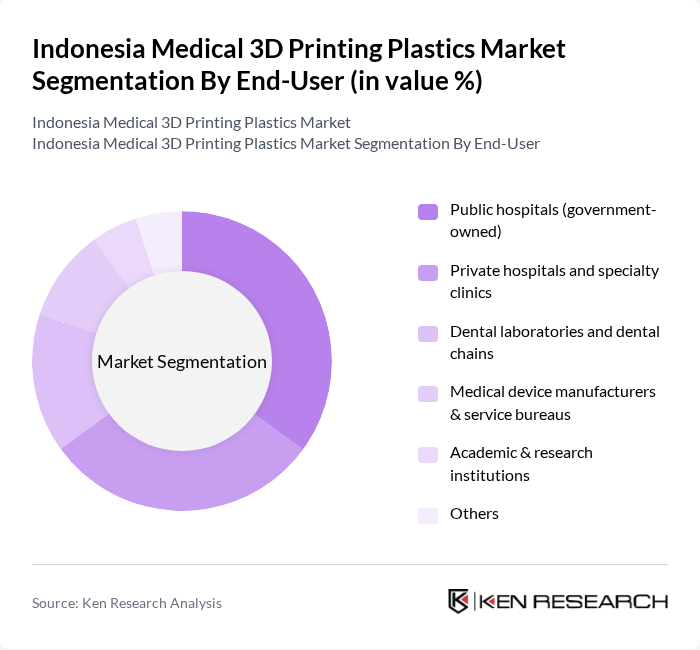
Public hospitals are the dominant end-user segment, driven by government efforts to upgrade referral hospitals, expand access to advanced diagnostics and surgical care, and integrate digital and additive technologies into public healthcare delivery. Private hospitals and specialty clinics also contribute significantly to the market as they seek innovative solutions for patient-specific implants, orthotics, and complex surgical planning. The dental sector is witnessing strong growth due to the rising demand for customized dental restorations, aligners, and surgical guides, further supporting the expansion of 3D printing technologies in the medical field.
The Indonesia Medical 3D Printing Plastics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Stratasys Ltd., 3D Systems Corporation, Materialise NV, EOS GmbH, Evonik Industries AG, Arkema S.A., BASF SE (BASF 3D Printing Solutions), Evonik Indonesia (local subsidiary operations), PT Formulatrix Indonesia, PT Bio Farma (Persero), PT Kimia Farma Tbk, PT Phapros Tbk, PT Multi Filament Indonesia, Digital Dental Solutions Indonesia (DDS Indonesia), PT Indoprinting & 3D Solutions contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Indonesian medical 3D printing plastics market appears promising, driven by technological advancements and increasing healthcare investments. In future, the integration of artificial intelligence in 3D printing processes is expected to streamline production and enhance customization capabilities. Furthermore, the rise of telemedicine is likely to create new avenues for 3D printed medical devices, facilitating remote patient care and expanding market reach. These trends indicate a dynamic evolution in the industry, fostering innovation and improved patient outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type of Plastic | Standard thermoplastics (e.g., PLA, ABS, PETG) Engineering-grade thermoplastics (e.g., PEEK, PEKK, PPSU) Photopolymer resins (SLA/DLP-compatible) Bioresorbable and bio-based medical polymers Others (composites, blends, elastomers) |
| By End-User | Public hospitals (government-owned) Private hospitals and specialty clinics Dental laboratories and dental chains Medical device manufacturers & service bureaus Academic & research institutions Others |
| By Application | Surgical guides and anatomical models Orthopedic implants, braces, and prosthetics Dental models, aligners, and restorations Patient-specific instruments and device components Wearables and external support devices Others |
| By Material Class | PLA (Polylactic Acid) – medical and dental grades ABS (Acrylonitrile Butadiene Styrene) PETG (Polyethylene Terephthalate Glycol) PEEK/PEKK and other high?performance polymers Biocompatible resins (Class I & IIa) Others |
| By 3D Printing Technology Used | Fused Deposition Modeling (FDM/FFF) Stereolithography (SLA/DLP) Selective Laser Sintering (SLS) Material Jetting / PolyJet Multi Jet Fusion (MJF) and others |
| By Distribution Channel | Direct sales by material manufacturers Local distributors and resellers Online platforms and e?commerce OEM-tied sales (printer–material bundles) Others |
| By Region | Java (Jakarta, West Java, Central Java, East Java) Sumatra Bali & Nusa Tenggara Kalimantan Sulawesi & Eastern Indonesia Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Prosthetics Manufacturing | 90 | Product Managers, R&D Engineers |
| Dental Applications | 80 | Dentists, Dental Technicians |
| Surgical Models Production | 70 | Surgeons, Medical Device Developers |
| Bioprinting Research | 50 | Biomedical Researchers, University Professors |
| Healthcare Facility Adoption | 90 | Hospital Administrators, Procurement Officers |
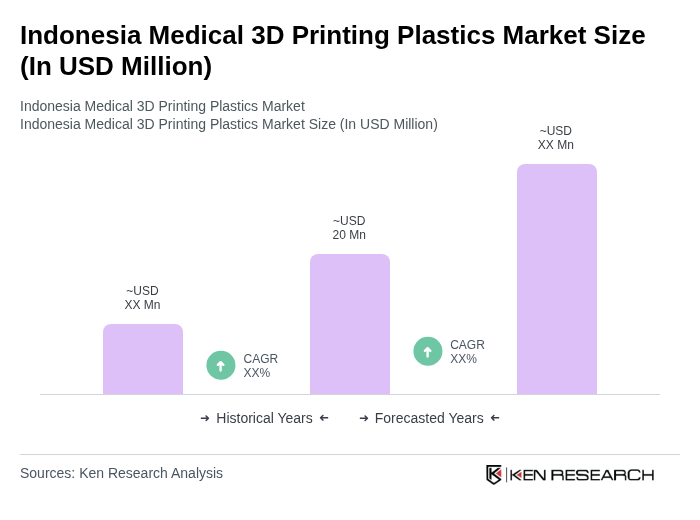
The Indonesia Medical 3D Printing Plastics Market is valued at approximately USD 20 million, reflecting significant growth driven by advancements in healthcare technology and increasing demand for personalized medical solutions.