
APAC Propofol Market Outlook to 2030
Region:Asia
Author(s):Shubham Kashyap
Product Code:KROD8428
December 2024
84
About the Report
APAC Propofol Market Overview
- The APAC Propofol market is valued at USD 1.4 billion, driven primarily by the increasing demand for anesthesia solutions across diverse medical applications, such as surgeries and ICU sedation. The rapid expansion of healthcare infrastructure, along with rising surgical volumes, has significantly fueled the demand for Propofol in the region. Additionally, the growing preference for short-acting anesthetic agents among healthcare providers, due to their fast recovery time, contributes to the markets steady growth, positioning APAC as a leading region in the anesthetic drug market.
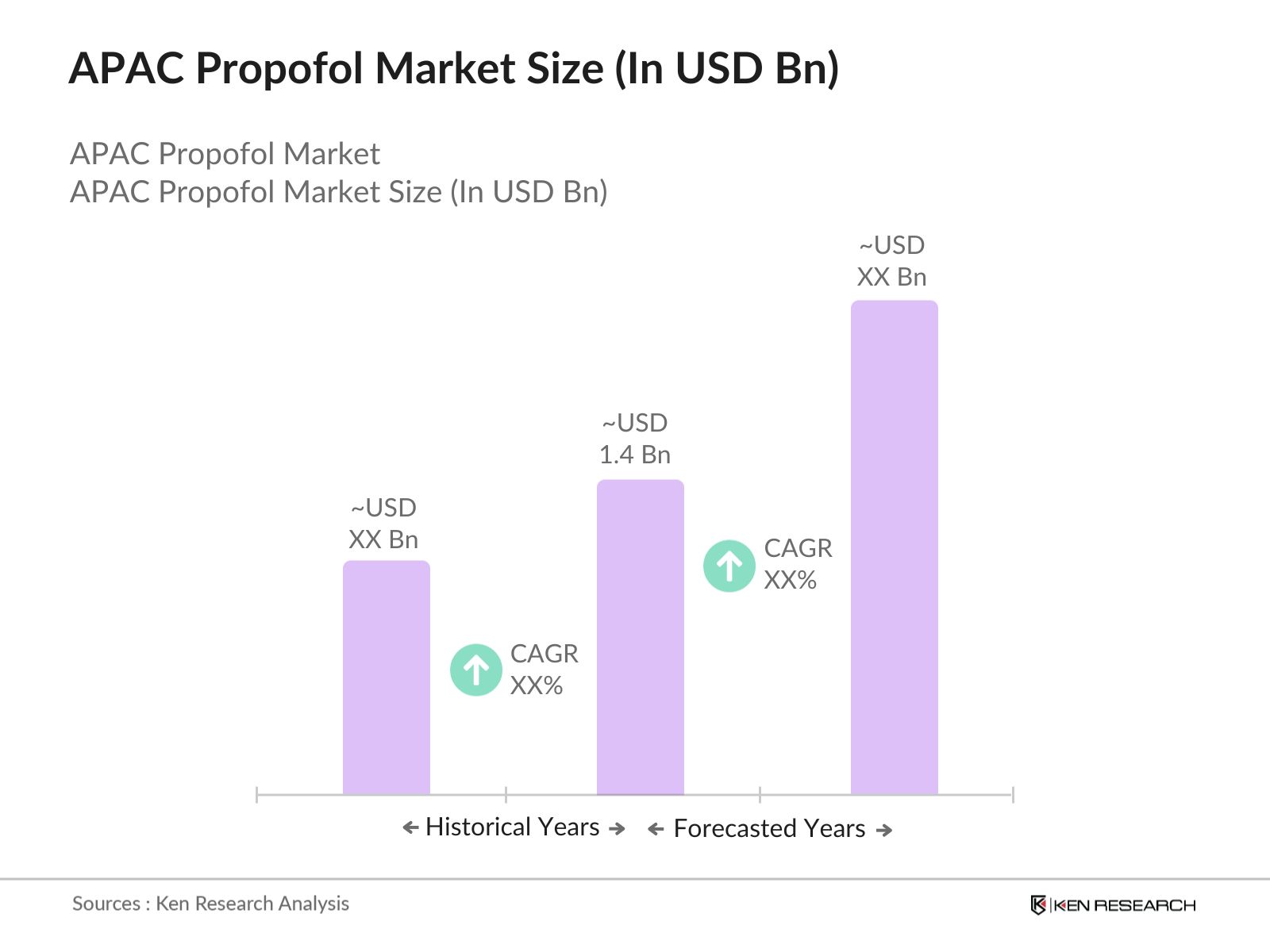
- China, Japan, and India are the major demand centers for Propofol in the APAC region. China leads due to its large population base, advanced healthcare facilities, and substantial investments in expanding surgical capabilities across hospitals. Japans dominance is driven by its high standards for healthcare and extensive use of advanced anesthetics in minimally invasive surgeries. Meanwhile, Indias growing medical tourism and increasing number of private hospitals have contributed to its rising demand for anesthetic drugs, establishing these countries as primary markets for Propofol in APAC.
- Propofol distribution in APAC is highly regulated, with each country implementing specific licensing and approval protocols. In 2023, Japans Ministry of Health processed over 600 applications for anesthetic licenses, while Australias TGA mandated rigorous testing for new propofol products. These regulatory standards, while essential for safety, extend time-to-market for propofol, impacting its availability across the region.
APAC Propofol Market Segmentation
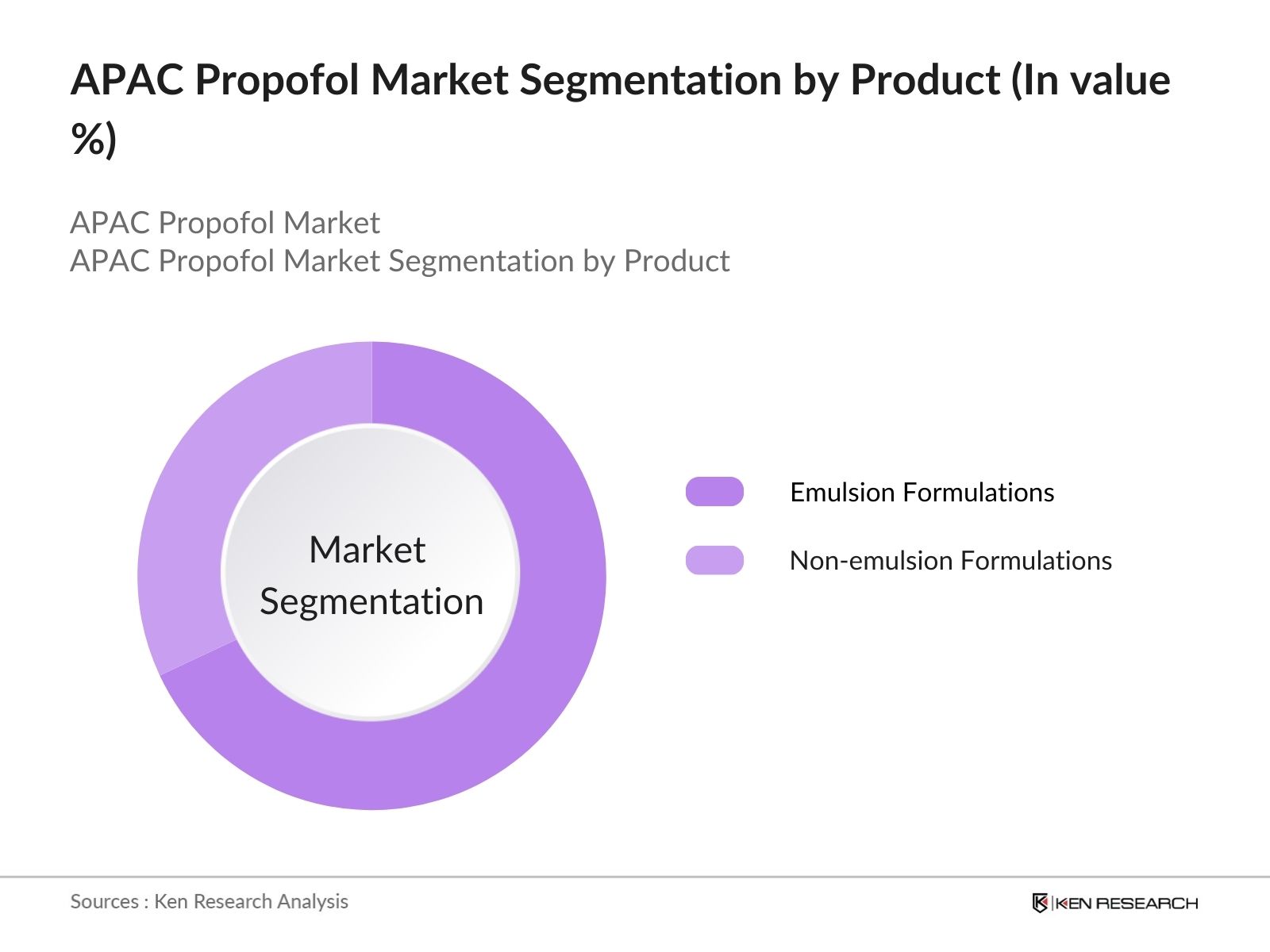
- By Product Type: The market is segmented by product type into emulsion formulations and non-emulsion formulations. Emulsion formulations hold the dominant market share within this segmentation due to their proven efficacy, quick onset, and safe profile in various medical applications. Emulsion-based Propofol formulations are widely adopted in surgeries, contributing to their high demand across hospitals and surgical centers. The preference for emulsion formulations stems from their stability and effectiveness in providing reliable anesthesia, making them a favored choice among healthcare professionals in APAC.
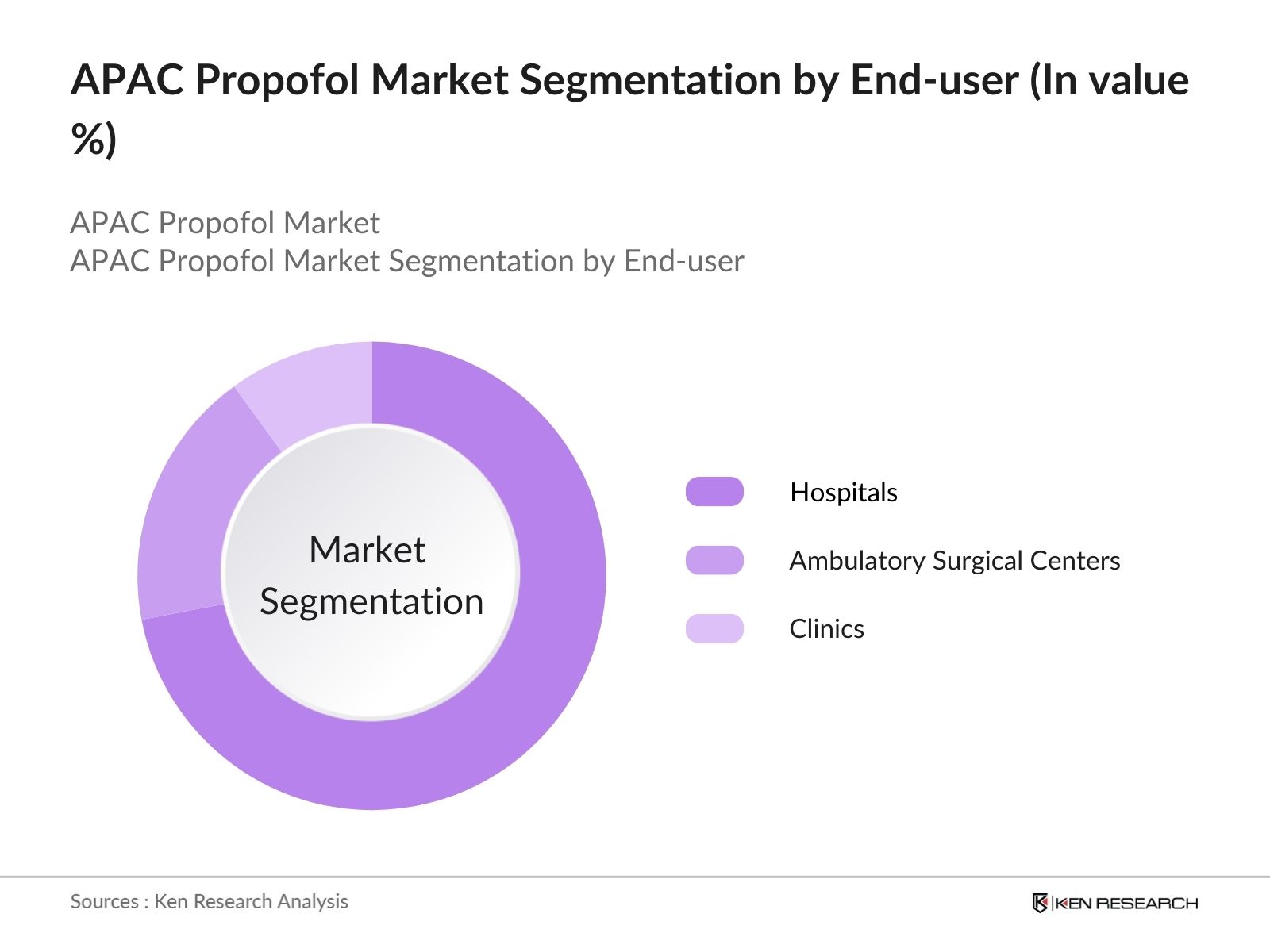
- By End-user: The market is segmented by end-user into hospitals, ambulatory surgical centers, and clinics. Hospitals represent the largest end-user segment, driven by the high frequency of surgical procedures requiring anesthesia, and the availability of intensive care units (ICUs) where Propofol is used for sedation. Additionally, hospitals generally have larger budgets, enabling bulk procurement of Propofol. Ambulatory surgical centers also show significant demand due to the increasing popularity of outpatient procedures, where Propofols rapid recovery time is advantageous, supporting its high usage in this segment.
APAC Propofol Market Competitive Landscape
The APAC Propofol market is highly competitive, with both global pharmaceutical giants and regional players striving to capture market share. Leading companies leverage their R&D capabilities, extensive distribution networks, and partnerships with healthcare facilities to maintain a competitive edge. 
APAC Propofol Market Analysis
Growth Drivers
- Increased Demand for Anesthesia: The APAC region is experiencing a notable rise in surgeries due to an aging population and the prevalence of chronic diseases. According to the World Bank, over 10 million surgeries were performed in the region in 2023, with the number expected to grow as healthcare access improves. This growth is directly influencing demand for propofol, as it remains the preferred anesthesia for surgical procedures due to its rapid onset and control over sedation depth. Improved healthcare infrastructure, especially in China and India, has contributed to increased operating capacities, thus driving anesthesia requirements.
- Rising Surgical Procedures (Healthcare Demand): The APAC region has seen a sharp rise in surgical procedures due to growing healthcare access, aging populations, and advancements in medical technology. This demand is particularly high in China, Japan, and India, where expanding hospital networks and specialized surgical centers have driven the need for anesthetic agents like Propofol. As of 2024, the increasing demand for both routine and complex surgeries continues to fuel the Propofol market, creating growth opportunities for manufacturers to expand their product offerings.
- Expanding Healthcare Infrastructure: Many APAC countries are investing in healthcare infrastructure to improve access and quality of care, contributing to the increased adoption of anesthetic drugs. Governments across the region have implemented policies to support the construction of new healthcare facilities, including hospitals and outpatient surgical centers. This infrastructure expansion supports the growth of the Propofol market, as newly established healthcare facilities require reliable anesthetic solutions for a range of medical procedures.
Challenges
- Strict Regulatory Compliance Requirements: The APAC Propofol market is subject to strict regulatory standards, with each country imposing its own set of requirements for drug approval and distribution. Manufacturers must meet stringent quality and safety standards to obtain approval, which can be a time-consuming and costly process. Regulatory compliance poses a significant challenge for companies, particularly those entering multiple APAC markets, as they need to navigate different regulatory frameworks.
- High Production and Raw Material Costs: The production of Propofol is cost-intensive, particularly due to the need for high-quality raw materials and advanced manufacturing processes. Additionally, fluctuations in raw material prices can impact the cost structure for manufacturers, leading to higher prices for consumers. The reliance on global suppliers for raw materials makes the Propofol market vulnerable to supply chain disruptions, which can affect availability and pricing stability in APAC markets.
APAC Propofol Market Future Outlook
The APAC Propofol market is expected to see substantial growth, supported by expanding healthcare infrastructure and the rising demand for efficient anesthesia solutions. Advancements in anesthetic drug formulations and increased focus on patient safety are likely to enhance Propofol adoption across medical institutions. Moreover, governments ongoing investments in healthcare facilities and services are projected to foster market expansion, with a shift toward innovative anesthetic practices that cater to evolving healthcare needs in APAC.
Future Market Opportunities
- Increasing Demand for Outpatient Surgical Procedures: The APAC region is witnessing a trend toward outpatient surgeries, as healthcare providers adopt minimally invasive techniques that reduce hospital stay durations. This trend aligns with the growing preference for quick-recovery anesthetic agents, making Propofol an ideal choice for outpatient procedures. This demand is further reinforced by government initiatives promoting cost-effective healthcare solutions, presenting an opportunity for Propofol manufacturers to cater to outpatient facilities.
- Development of Alternative Drug Delivery Methods: As part of ongoing innovation, there is a rising interest in alternative drug delivery methods for Propofol, such as patient-controlled intravenous sedation. This approach offers more control to healthcare providers and patients, potentially improving outcomes and expanding Propofols applications. Research and development efforts focused on improving drug delivery options could drive market growth by making Propofol more adaptable for various clinical settings.
Scope of the Report
|
By Product Type |
Emulsion Formulations |
|
By Application |
General Anesthesia |
|
By End-user |
Hospitals |
|
By Route of Administration |
Intravenous |
|
By Region |
China |
Products
Key Target Audience
Hospitals and Healthcare Providers
Ambulatory Surgical Centers
Pharmaceutical Distributors and Suppliers
Government and Regulatory Bodies (e.g., APAC Health Ministries, Food and Drug Administrations)
Anesthesiologists and Healthcare Professionals
Investors and Venture Capitalist Firms
Banks and Financial Institutions
Medical Research Institutes and Laboratories
Companies
Players Mentioned in the Report
Fresenius Kabi
Pfizer Inc.
AstraZeneca
Hikma Pharmaceuticals
Dr. Reddy's Laboratories
Baxter International Inc.
Teva Pharmaceuticals
Mylan N.V.
B. Braun Melsungen AG
Accord Healthcare
Piramal Healthcare
Jiangsu Hengrui Medicine Co., Ltd.
Abbott Laboratories
Sandoz (A Novartis Division)
Bharat Serums and Vaccines Limited
Table of Contents
APAC Propofol Market Overview
1.1 Definition and Scope
1.2 Market Taxonomy
1.3 Market Growth Rate
1.4 Market Segmentation Overview
APAC Propofol Market Size (In USD Bn)
2.1 Historical Market Size
2.2 Year-on-Year Growth Analysis
2.3 Key Market Developments and Milestones
APAC Propofol Market Analysis
3.1 Growth Drivers
3.1.1 Increased Demand for Anesthesia (Due to Growing Surgical Procedures)
3.1.2 Advancements in Drug Delivery Systems (Impact on Propofol Utilization)
3.1.3 Rising Healthcare Expenditure (Across Emerging Economies)
3.2 Market Challenges
3.2.1 Regulatory Constraints (Stringent FDA and Regional Regulatory Guidelines)
3.2.2 High Production Costs (Associated with Propofol Formulations)
3.2.3 Supply Chain Volatility (Impact on Raw Material Availability)
3.3 Opportunities
3.3.1 Expansion in Outpatient Surgery Centers (Growing Demand for Sedation)
3.3.2 Development of Alternative Delivery Forms (For Improved Efficacy)
3.3.3 Partnerships with Pharma Companies (To Address Market Demand)
3.4 Trends
3.4.1 Preference for Short-acting Anesthetics (Due to Faster Recovery Times)
3.4.2 Rise in Research for Safer Sedation Protocols (Impact on Propofol Usage)
3.4.3 Increased Focus on Biosimilar Development (Market Expansion)
3.5 Government Regulations
3.5.1 Licensing and Approval Processes (Regional Differences in APAC)
3.5.2 Propofol Manufacturing Standards (Impact on Production Quality)
3.5.3 Public Health Policies (Influencing Propofol Demand)
3.6 SWOT Analysis
3.7 Stakeholder Ecosystem
3.8 Porters Five Forces
3.9 Competitive Landscape Overview
APAC Propofol Market Segmentation
4.1 By Product Type (In Value %)
4.1.1 Emulsion Formulations
4.1.2 Non-emulsion Formulations
4.2 By Application (In Value %)
4.2.1 General Anesthesia
4.2.2 Sedation for Intensive Care Units (ICUs)
4.2.3 Short-term Sedation
4.3 By End-user (In Value %)
4.3.1 Hospitals
4.3.2 Ambulatory Surgical Centers
4.3.3 Clinics
4.4 By Route of Administration (In Value %)
4.4.1 Intravenous
4.4.2 Inhalation (Emerging Options)
4.5 By Region (In Value %)
4.5.1 China
4.5.2 India
4.5.3 Japan
4.5.4 Australia
4.5.5 South Korea
APAC Propofol Market Competitive Analysis
5.1 Detailed Profiles of Major Companies
5.1.1 Fresenius Kabi
5.1.2 Baxter International Inc.
5.1.3 Pfizer Inc.
5.1.4 AstraZeneca
5.1.5 Teva Pharmaceuticals
5.1.6 B. Braun Melsungen AG
5.1.7 Dr. Reddy's Laboratories
5.1.8 Abbott Laboratories
5.1.9 Hikma Pharmaceuticals
5.1.10 Mylan N.V.
5.1.11 Accord Healthcare
5.1.12 Piramal Healthcare
5.1.13 Jiangsu Hengrui Medicine Co., Ltd.
5.1.14 Sandoz (A Novartis Division)
5.1.15 Bharat Serums and Vaccines Limited
5.2 Cross Comparison Parameters (Market Presence, Product Portfolio, Manufacturing Capacity, R&D Investments, Strategic Partnerships, Revenue Growth, Market Reach, Innovation in Drug Delivery)
5.3 Market Share Analysis
5.4 Strategic Initiatives and Developments
5.5 Mergers and Acquisitions
5.6 Investment Analysis
5.7 Government Grants and Support
5.8 Private Equity and Venture Capital Investments
APAC Propofol Market Regulatory Framework
6.1 Drug Approval Processes (By Regional Regulatory Bodies)
6.2 Compliance with Quality Standards (ISO, GMP)
6.3 Certification Requirements (APAC Region-Specific Standards)
APAC Propofol Future Market Size (In USD Bn)
7.1 Future Market Size Projections
7.2 Key Factors Driving Future Market Growth
APAC Propofol Future Market Segmentation
8.1 By Product Type (In Value %)
8.2 By Application (In Value %)
8.3 By End-user (In Value %)
8.4 By Route of Administration (In Value %)
8.5 By Region (In Value %)
APAC Propofol Market Analysts Recommendations
9.1 TAM/SAM/SOM Analysis
9.2 Customer Cohort Analysis
9.3 Strategic Marketing Initiatives
9.4 White Space Opportunity Analysis
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
The initial stage involves identifying and defining the critical variables impacting the APAC Propofol market, such as demand drivers, supply chain dynamics, and regulatory requirements. This includes gathering data through desk research from secondary and proprietary sources to map the markets ecosystem.
Step 2: Market Analysis and Construction
In this phase, historical data on the APAC Propofol market is compiled and analyzed to assess market penetration, demand from healthcare segments, and revenue contributions. An analysis of distribution channels is conducted to ensure accurate market estimations.
Step 3: Hypothesis Validation and Expert Consultation
Hypotheses are developed regarding market trends and growth drivers and validated through consultations with industry experts. These consultations provide insights into operational aspects and allow for refinement of market data through firsthand perspectives from healthcare providers and manufacturers.
Step 4: Research Synthesis and Final Output
The final phase includes consolidating findings to provide a comprehensive market outlook. Data validation is achieved through collaboration with Propofol manufacturers, ensuring accuracy in the reports analysis and insights, ready for presentation to APAC healthcare professionals and market stakeholders.
Frequently Asked Questions
01. How big is the APAC Propofol Market?
The APAC Propofol Market is valued at USD 1.4 billion, driven by increasing demand for surgical procedures and improvements in healthcare infrastructure across major APAC economies.
02. What are the major growth drivers in the APAC Propofol Market?
Key growth drivers in the APAC Propofol Market include the expansion of healthcare facilities, the rising number of surgeries, and technological advancements in anesthetic drug formulations, making Propofol a preferred choice in various medical procedures.
03. Which countries dominate the APAC Propofol Market?
China and Japan dominate the APAC Propofol Market due to their advanced healthcare infrastructure, high surgical volumes, and the presence of leading pharmaceutical companies that manufacture and distribute Propofol.
04. What challenges does the APAC Propofol Market face?
Challenges in the APAC Propofol Market include regulatory compliance, high production costs, and volatility in the supply chain, particularly for raw materials essential in manufacturing.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















