
Asia-Pacific Airway Management Device Market Outlook to 2030
Region:Asia
Author(s):Meenakshi
Product Code:KROD2154
November 2024
92
About the Report
Asia-Pacific Airway Management Device Market Overview
- The Asia-Pacific airway management device market is valued at USD 1.95 billion based on a five-year historical analysis. This market is driven by the rising incidence of respiratory diseases, increased surgical procedures, and growing awareness about advanced airway management techniques. The increasing prevalence of chronic obstructive pulmonary disease (COPD) and asthma across major countries in the region has led to a high demand for airway management devices.
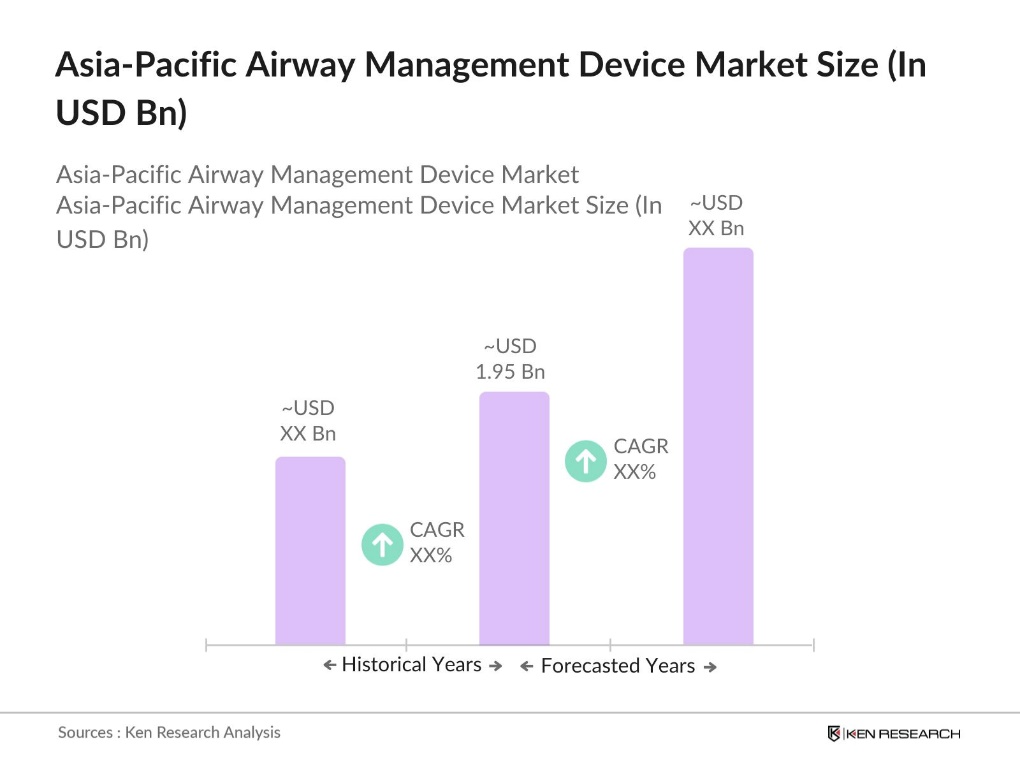
- Dominant countries such as China, Japan, and India lead the airway management device market in the Asia-Pacific region. The dominance of these countries is attributed to their large populations, well-established healthcare infrastructure, and increasing healthcare expenditure. Additionally, China and Japans significant investments in medical technology and innovation further consolidate their leading positions in the market.
- Medical device reporting systems have become a key focus for healthcare regulators in the region. In 2023, China updated its Medical Device Adverse Event Reporting system, requiring more detailed post-market surveillance. Compliance rates among medical device manufacturers reached 85% that year, reflecting the increased pressure to monitor and report device performance, especially for airway management tools.
Asia-Pacific Airway Management Device Market Segmentation
By Device Type: The Asia-Pacific airway management device market is segmented by device type into endotracheal tubes, tracheostomy tubes, supraglottic airway devices, nasopharyngeal airways, and oropharyngeal airways. Endotracheal tubes hold a dominant market share due to their frequent use in surgeries and emergency situations. This sub-segment's dominance is driven by the increasing number of surgeries, the growing adoption of ventilators, and their critical role in airway management during anesthesia and critical care. Additionally, the development of single-use and disposable endotracheal tubes has boosted demand, especially in infection-control-sensitive environments like hospitals and clinics.

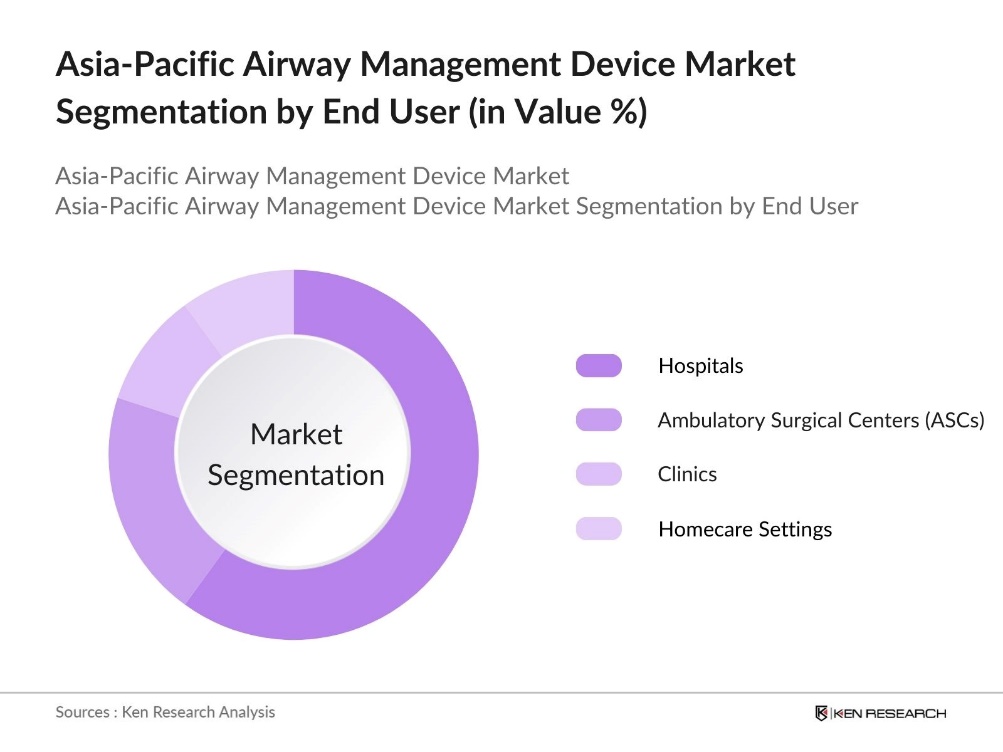
By End User: The Asia-Pacific airway management device market is segmented by end user into hospitals, ambulatory surgical centers (ASCs), clinics, and homecare settings. Hospitals dominate this segment, with a substantial share of airway management devices being used in hospital settings due to the high volume of surgeries, emergency treatments, and respiratory care provided. Hospitals are the primary users of advanced airway devices because they offer a broader range of services that require specialized equipment, from routine surgeries to critical care for severe respiratory disorders.
Asia-Pacific Airway Management Device Market Competitive Landscape
The Asia-Pacific airway management device market is characterized by the presence of several key players. The market is dominated by large global medical device companies and regional players that have developed a strong foothold due to innovation and strategic partnerships. These companies invest heavily in research and development to introduce new and innovative products, which helps them stay competitive in the market.
|
Company |
Establishment Year |
Headquarters |
R&D Spending |
Global Reach |
Product Portfolio |
Patents |
Market Share |
No. of Employees |
|
Teleflex Inc. |
1943 |
Wayne, Pennsylvania |
||||||
|
Medtronic PLC |
1949 |
Dublin, Ireland |
||||||
|
Ambu A/S |
1937 |
Copenhagen, Denmark |
||||||
|
Fisher & Paykel Healthcare |
1934 |
Auckland, New Zealand |
||||||
|
Smiths Medical |
1940 |
London, United Kingdom |
Asia-Pacific Airway Management Device Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Respiratory Diseases: The Asia-Pacific region has seen a significant rise in chronic respiratory diseases such as asthma, COPD, and bronchitis, particularly in countries like China and India. By 2020, it was estimated that the East Asia and Pacific region had around 136 million cases of COPD, with projections indicating significant increases by 2050. This rise in patient demographics has increased the demand for airway management devices in hospital settings across the region.
- Aging Population: The aging population across Asia-Pacific has also driven the demand for airway management devices, as older adults are more prone to respiratory complications. Japan's elderly population ratio is reported as 29.1% of its total population, with approximately 36.2 million individuals aged 65 or older as of September 2023. This surge is mirrored across countries like South Korea, China, and Australia. A higher geriatric population results in greater hospital admissions for surgeries and respiratory issues, driving airway management demand. The increased demand for geriatric care is expected to further stretch healthcare resources in the region.
- Increasing Surgical Procedures: The growing number of surgeries in Asia-Pacific, particularly in countries like Australia, South Korea, and China, has significantly increased the demand for airway management devices. With a rise in both elective and emergency procedures, healthcare facilities are relying more on advanced airway devices to ensure patient safety and operational efficiency during surgeries, driving the need for effective airway management solutions across the region.
Market Challenges
- High Cost of Devices: The high cost of airway management devices presents a significant barrier in the Asia-Pacific region, especially in low- and middle-income countries. Advanced devices are often expensive, making them less accessible in countries with lower healthcare spending. This price disparity between developed and emerging markets limits the widespread adoption of advanced technologies, as healthcare providers in lower-income regions struggle to afford and integrate these devices into their systems.
- Stringent Regulatory Requirements: The regulatory landscape for medical devices in Asia-Pacific varies by country, often making market entry challenging. Strict regulations can lead to longer approval times, especially for advanced medical devices. Companies looking to expand into the region must navigate complex compliance requirements, which can slow down product launches. This regulatory variability complicates market access and increases the cost and time associated with introducing new airway management devices.
Asia-Pacific Airway Management Device Market Future Outlook
Over the next five years, the Asia-Pacific airway management device market is expected to grow significantly. The markets growth will be driven by continuous technological advancements, increasing awareness about respiratory health, and growing government support to improve healthcare access. The rising elderly population in the region will also contribute to the increased demand for airway management devices, especially in critical care settings.
Market Opportunities
- Expanding Medical Tourism: Medical tourism is rapidly growing in Asia-Pacific, with countries like Thailand, Singapore, and India attracting a large number of international patients seeking cost-effective, high-quality healthcare. This surge in medical tourism has boosted the demand for advanced airway management devices, particularly in surgical centers that cater to these patients. As more tourists seek medical procedures, the need for modern medical devices presents significant opportunities for market expansion in the region.
- Untapped Markets in Emerging Economies: Emerging economies in Asia-Pacific, such as Vietnam, the Philippines, and Indonesia, offer promising opportunities for the airway management device market. These nations are progressively investing in healthcare infrastructure, aiming to improve access to advanced medical technologies. As healthcare systems in these countries expand, there is a growing potential for the adoption of airway management devices, opening up new market prospects.
Scope of the Report
|
Device Type |
Endotracheal Tubes Tracheostomy Tubes Supraglottic Airway Devices Nasopharyngeal Airways Oropharyngeal Airways |
|
Application |
Anesthesia Emergency Medicine Critical Care Neonatal and Pediatric Care |
|
End User |
Hospitals Ambulatory Surgical Centers (ASCs) Clinics Homecare Settings |
|
Patient Type |
Adult Pediatric Geriatric |
|
Region |
East Asia South Asia Southeast Asia Oceania |
Products
Key Target Audience
Medical Device Manufacturers
Healthcare Equipment Manufactures
Telemedicine companies
Medical Equipment Leasing Companies
Government and Regulatory Bodies (Ministry of Health, Asia-Pacific Medical Device Regulatory Authority)
Investors and venture capital Firms
Banks and Financial Institutions
Companies
Players Mentioned in the Report
Teleflex Inc.
Medtronic PLC
Ambu A/S
Smiths Medical
Fisher & Paykel Healthcare
Cook Medical Inc.
VBM Medizintechnik GmbH
SunMed LLC
Intersurgical Ltd.
Boston Scientific Corporation
Table of Contents
1. Asia-Pacific Airway Management Device Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2. Asia-Pacific Airway Management Device Market Size (In USD Mn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
3. Asia-Pacific Airway Management Device Market Analysis
3.1. Growth Drivers
3.1.1. Increasing Prevalence of Chronic Respiratory Diseases (incidence rate, patient demographics)
3.1.2. Aging Population (aging index, geriatric care demand)
3.1.3. Increasing Surgical Procedures (surgical volumes, types of surgeries)
3.1.4. Technological Advancements in Airway Management Devices (R&D investments, patents filed)
3.2. Market Challenges
3.2.1. High Cost of Devices (pricing metrics, cost comparisons)
3.2.2. Stringent Regulatory Requirements (FDA/EMA regulations, compliance hurdles)
3.2.3. Risk of Complications (device failure rates, clinical outcomes)
3.3. Opportunities
3.3.1. Expanding Medical Tourism (growth regions, demand indicators)
3.3.2. Untapped Markets in Emerging Economies (penetration rates, disposable income)
3.3.3. Increasing Demand for Homecare Settings (home healthcare expenditures, device adoption)
3.4. Trends
3.4.1. Use of Video Laryngoscopy (adoption rates, user demographics)
3.4.2. Shift Towards Single-Use Airway Devices (environmental impact, cost-benefit analysis)
3.4.3. Integration of AI and Machine Learning (AI-based airway management systems, innovation rate)
3.5. Government Regulation
3.5.1. Asia-Pacific Medical Device Regulations (Asia-Pacific regulatory framework, approvals timeline)
3.5.2. Medical Device Reporting Systems (adverse event reporting, compliance rates)
3.5.3. National Reimbursement Policies (coverage rate, government reimbursements)
3.6. SWOT Analysis
3.7. Stakeholder Ecosystem
3.8. Porters Five Forces Analysis
3.9. Competition Ecosystem
4. Asia-Pacific Airway Management Device Market Segmentation
4.1. By Device Type (In Value %)
4.1.1. Endotracheal Tubes
4.1.2. Tracheostomy Tubes
4.1.3. Supraglottic Airway Devices
4.1.4. Nasopharyngeal Airways
4.1.5. Oropharyngeal Airways
4.2. By Application (In Value %)
4.2.1. Anesthesia
4.2.2. Emergency Medicine
4.2.3. Critical Care
4.2.4. Neonatal and Pediatric Care
4.3. By End User (In Value %)
4.3.1. Hospitals
4.3.2. Ambulatory Surgical Centers (ASCs)
4.3.3. Clinics
4.3.4. Homecare Settings
4.4. By Patient Type (In Value %)
4.4.1. Adult
4.4.2. Pediatric
4.4.3. Geriatric
4.5. By Region (In Value %)
4.5.1. East Asia
4.5.2. South Asia
4.5.3. Southeast Asia
4.5.4. Oceania
5. Asia-Pacific Airway Management Device Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. Teleflex Inc.
5.1.2. Medtronic PLC
5.1.3. Ambu A/S
5.1.4. Smiths Medical
5.1.5. Cook Medical Inc.
5.1.6. VBM Medizintechnik GmbH
5.1.7. SunMed LLC
5.1.8. Intersurgical Ltd.
5.1.9. Fisher & Paykel Healthcare
5.1.10. Boston Scientific Corporation
5.1.11. ConvaTec Group PLC
5.1.12. Karl Storz SE & Co. KG
5.1.13. Armstrong Medical
5.1.14. Ventlab Corporation
5.1.15. Verathon Inc.
5.2. Cross Comparison Parameters (Revenue, R&D Spending, Global Reach, Product Portfolio, Market Share, No. of Patents, No. of Employees, Headquarters)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers and Acquisitions
5.6. Investment Analysis
5.7. Government Grants and Funding
5.8. Venture Capital Funding
6. Asia-Pacific Airway Management Device Market Regulatory Framework
6.1. Medical Device Classification
6.2. Regulatory Approvals
6.3. Post-market Surveillance
6.4. Compliance with International Standards
7. Asia-Pacific Airway Management Device Future Market Size (In USD Mn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. Asia-Pacific Airway Management Device Future Market Segmentation
8.1. By Device Type (In Value %)
8.2. By Application (In Value %)
8.3. By End User (In Value %)
8.4. By Patient Type (In Value %)
8.5. By Region (In Value %)
9. Asia-Pacific Airway Management Device Market Analysts Recommendations
9.1. TAM/SAM/SOM Analysis
9.2. Customer Cohort Analysis
9.3. Marketing Initiatives
9.4. White Space Opportunity Analysis
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
The initial phase involves constructing an ecosystem map encompassing all major stakeholders within the Asia-Pacific Airway Management Device Market. This step is underpinned by extensive desk research, utilizing a combination of secondary and proprietary databases to gather comprehensive industry-level information. The primary objective is to identify and define the critical variables that influence market dynamics.
Step 2: Market Analysis and Construction
In this phase, we will compile and analyze historical data pertaining to the Asia-Pacific Airway Management Device Market. This includes assessing market penetration, the ratio of devices to service providers, and the resultant revenue generation. Furthermore, an evaluation of service quality statistics will be conducted to ensure the reliability and accuracy of the revenue estimates.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses will be developed and subsequently validated through computer-assisted telephone interviews (CATI) with industry experts representing a diverse array of companies. These consultations will provide valuable operational and financial insights directly from industry practitioners, which will be instrumental in refining and corroborating the market data.
Step 4: Research Synthesis and Final Output
The final phase involves direct engagement with multiple airway management device manufacturers to acquire detailed insights into product segments, sales performance, consumer preferences, and other pertinent factors. This interaction will serve to verify and complement the statistics derived from the bottom-up approach, thereby ensuring a comprehensive, accurate, and validated analysis of the Asia-Pacific airway management device market.
Frequently Asked Questions
01. How big is the Asia-Pacific Airway Management Device Market?
The Asia-Pacific airway management device market is valued at USD 1.95 billion, driven by increasing demand for surgical and emergency care, rising cases of respiratory diseases, and advances in medical technologies.
02. What are the challenges in the Asia-Pacific Airway Management Device Market?
Challenges in Asia-Pacific airway management device market include high costs of advanced airway devices, regulatory hurdles across different countries, and a shortage of skilled healthcare workers trained in airway management techniques.
03. Who are the major players in the Asia-Pacific Airway Management Device Market?
Key players in the Asia-Pacific airway management device market include Teleflex Inc., Medtronic PLC, Ambu A/S, Fisher & Paykel Healthcare, and Smiths Medical. These companies dominate due to their extensive product portfolios, strong R&D investments, and established distribution networks.
04. What are the growth drivers of the Asia-Pacific Airway Management Device Market?
The Asia-Pacific airway management device market is propelled by factors such as the increasing prevalence of respiratory diseases, growing elderly population, and rising healthcare expenditure in countries like China, India, and Japan.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















