
Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Outlook to 2030
Region:Asia
Author(s):Paribhasha Tiwari
Product Code:KROD6232
December 2024
93
About the Report
Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Overview
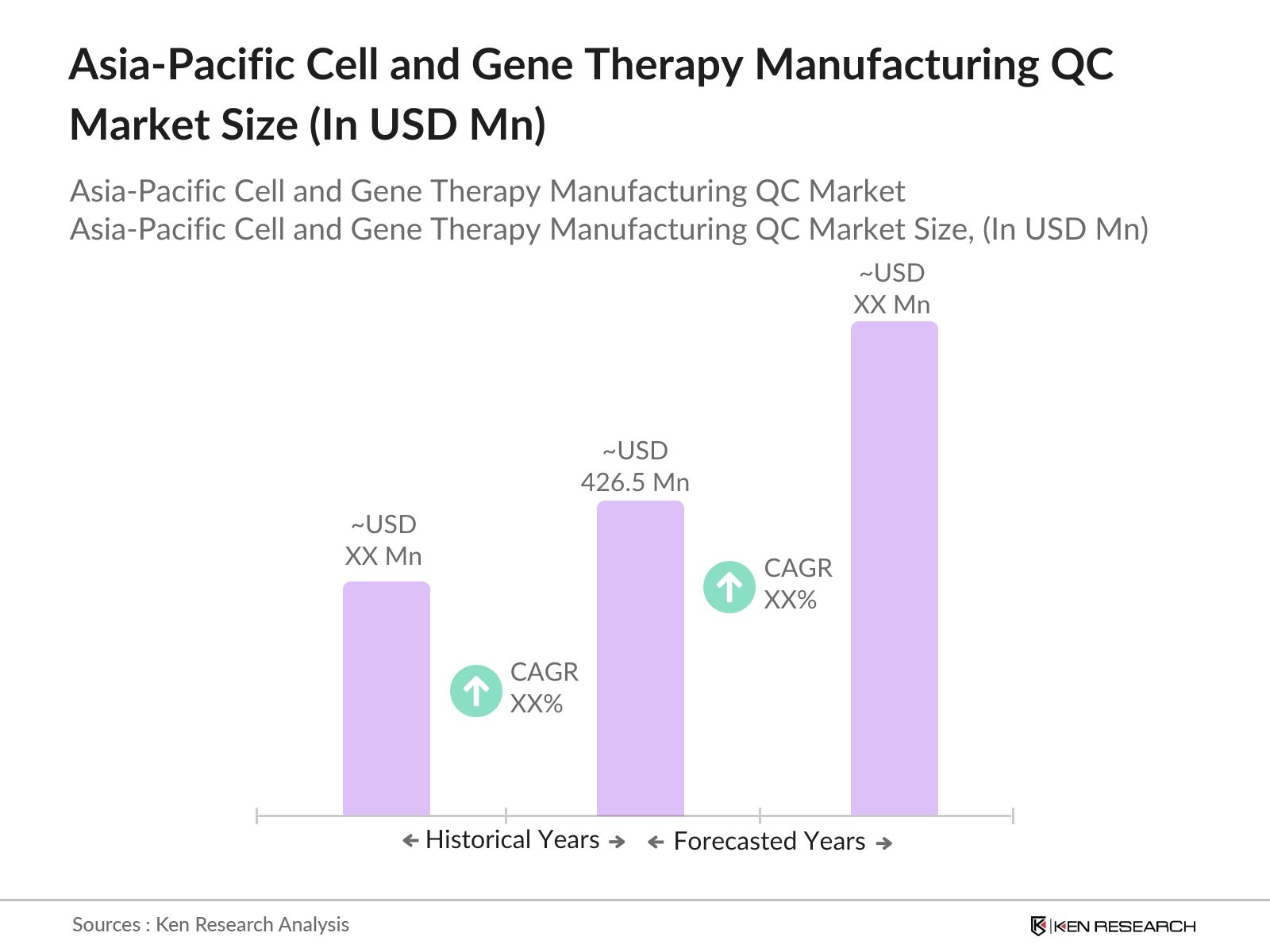
- The Asia-Pacific cell and gene therapy manufacturing QC market is valued at USD 426.5 million, driven by rising investments in personalized medicine and advanced biotechnology across the region. Growth factors include increasing clinical trials and an expanding network of biomanufacturing facilities meeting rigorous regulatory standards, making QC processes essential. This market is further propelled by public and private sector support to meet demand for gene-modified therapies.
- Countries such as China and Japan are dominant due to their advanced biomanufacturing infrastructure and government support. China has a growing network of research institutions specializing in cell therapy, while Japan benefits from a streamlined regulatory environment. These factors enable both countries to play leading roles in market development.
- Japan has introduced a comprehensive plan for advanced therapies, allocating over 100 billion toward the development of infrastructure for QC in cell and gene therapy. This initiative, launched in 2024, will see the establishment of new testing labs and workforce training centers to enhance the regional QC landscape for innovative therapies.
Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Segmentation
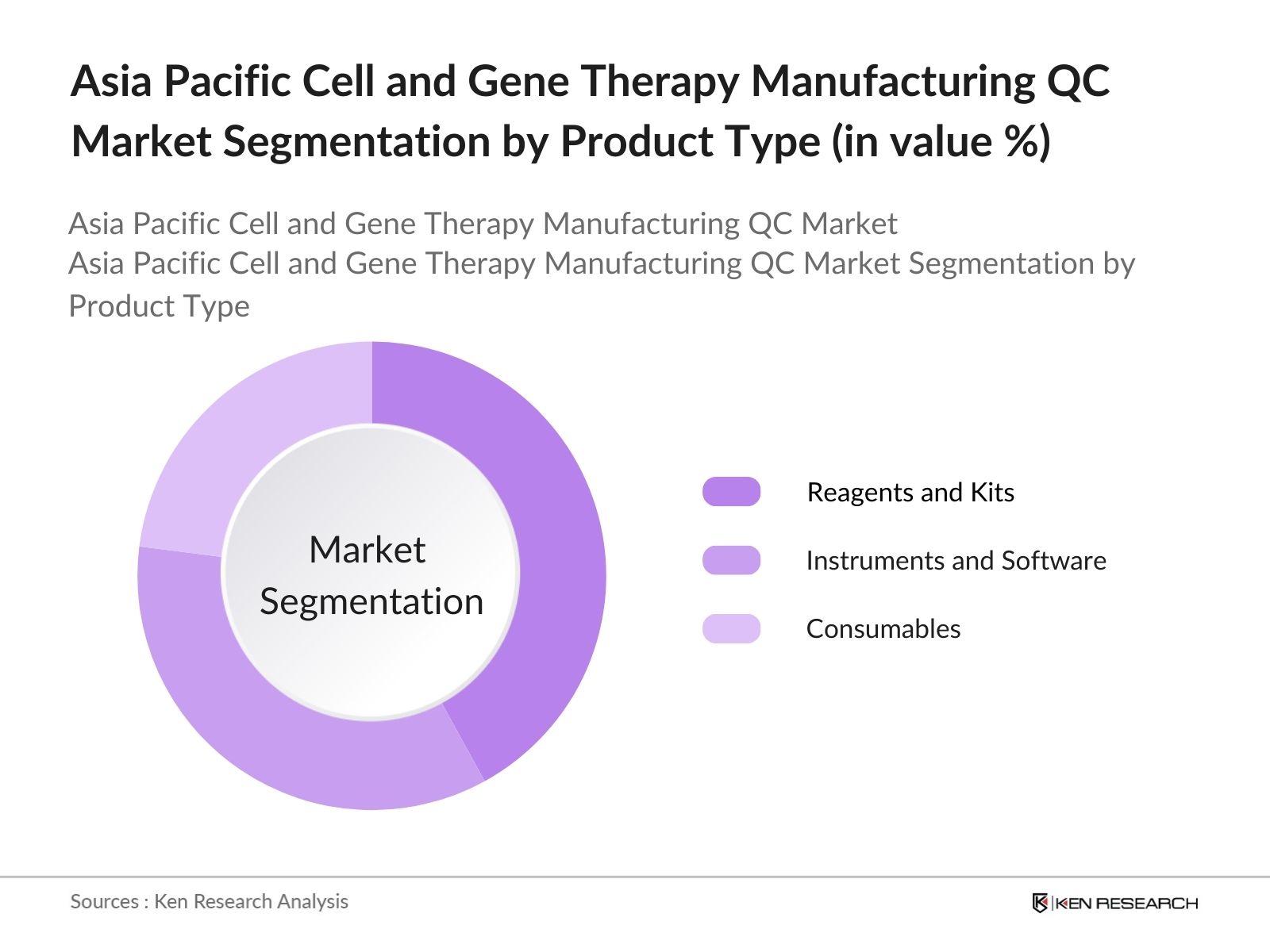
By Product Type: The Asia-Pacific cell and gene therapy manufacturing QC market is segmented by product type into Reagents & Kits, Instruments & Software, and Consumables. Reagents & Kits dominate the product type segmentation, holding a 42% market share in 2023 due to their essential role in ensuring cell viability and contamination control in biomanufacturing. Their frequent application in quality assurance processes has solidified their market lead.
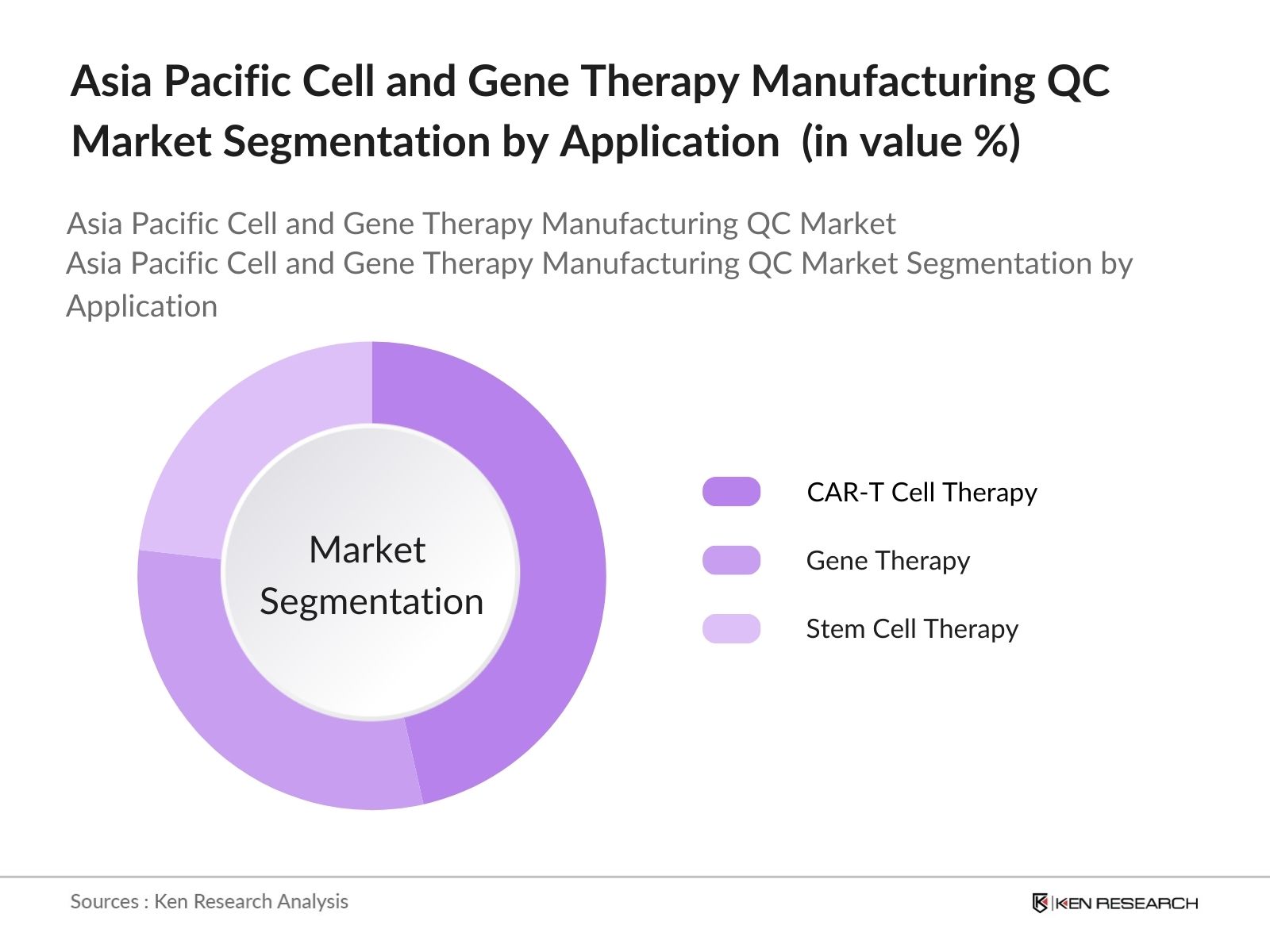
By Application: The market is further segmented by application into CAR-T Cell Therapy, Gene Therapy, and Stem Cell Therapy. CAR-T Cell Therapy leads with a 46% share within the application segment, primarily due to its increasing adoption in cancer treatment and the stringent QC standards required. Specialized QC protocols for CAR-T ensure the therapy's potency and specificity, solidifying its dominance in this category.
Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Competitive Landscape
The Asia-Pacific cell and gene therapy manufacturing QC market has a competitive landscape dominated by both regional and international companies. Key players include Charles River Laboratories, Catalent Inc., and WuXi AppTec, which leverage their infrastructure, partnerships, and advanced technology to maintain a stronghold in the market. These companies are frequently involved in collaborations with regulatory bodies, which helps streamline approval for innovative QC solutions.
Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Analysis
Growth Drivers
- Increase in Clinical Trials for Cell and Gene Therapies: The Asia-Pacific region has seen a surge in clinical trials for cell and gene therapies, with over 1,500 trials initiated by mid-2024, demonstrating strong regional interest in innovative therapies. The availability of large patient populations in countries like China and India has fueled this growth, enabling quicker recruitment and more extensive trial phases. This growth is also reflected in government support, as seen in Japan's substantial investments in regenerative medicine, with nearly 50 billion JPY dedicated to advancing clinical trials. The rise in clinical trials highlights an escalating need for specialized QC services to ensure efficacy and safety in cell and gene therapies.
- Advances in Cell and Gene Therapy Technologies: The development of next-generation gene-editing technologies, such as CRISPR and CAR-T, has significantly advanced the efficacy of cell and gene therapies. In 2024, Asia-Pacific countries have recorded over 800 laboratories actively employing CRISPR-based gene editing, underscoring the technology's impact in clinical applications. These technological advancements increase the complexity of QC processes, demanding robust, high-precision quality control solutions. Funding support from governments, as seen with Chinas recent investment of 20 billion in genetic research infrastructure, underscores the region's commitment to scaling advanced QC frameworks in line with technological progress.
- Rising Demand for Specialized QC Services: As cell and gene therapy products progress from research to commercialization, the need for specialized QC services is escalating. Countries like South Korea have reported over 500 ongoing manufacturing processes for cell therapies, all requiring stringent quality checks, reinforcing the need for tailored QC protocols. The regional market is set to benefit from investments in specialized QC facilities, including Singapores cell manufacturing units, which have allocated over S$30 million toward QC process development. This trend ensures that the region meets the rising demand for advanced quality controls in cell and gene therapy manufacturing.
Market Challenges
- High Costs of Quality Control (QC) in Cell and Gene Therapy: The costs associated with QC in cell and gene therapy remain a significant barrier, with an average expenditure of $500,000 per batch reported in clinical-stage products. This high cost is attributed to advanced techniques and equipment necessary to meet regulatory standards, particularly in emerging economies like Vietnam, where manufacturing costs have grown by 15% due to additional QC measures. Addressing these cost challenges is crucial for sustaining the regional expansion of cell and gene therapy manufacturing.
- Limited Skilled Workforce for Advanced QC Methods: A shortage of trained professionals in Asia-Pacific to handle sophisticated QC processes is a growing concern, with reports indicating a workforce gap of nearly 20,000 specialists across the region. This scarcity is exacerbated by the complexity of QC requirements in gene therapy manufacturing, where skilled personnel must conduct rigorous testing protocols. Efforts are underway in Japan and South Korea to establish training programs, but the lack of an adequately skilled workforce remains a substantial challenge for sustained QC compliance.
Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Future Outlook
The Asia-Pacific cell and gene therapy manufacturing QC market is projected to grow substantially over the next five years, driven by advancements in QC technologies, government support for biopharmaceutical innovation, and the increasing need for specialized therapies. Further integration of AI in QC processes and automation will enhance efficiency, supporting market expansion and ensuring rigorous quality standards.
Market Opportunities
- Expansion of GMP-Compliant Manufacturing Facilities: The expansion of GMP-compliant (Good Manufacturing Practice) facilities across Asia-Pacific is a promising opportunity, with over 300 new facilities expected to be operational by 2025. Countries like China and India have announced significant investments in building GMP-certified units to meet global standards. For example, Chinas biopharmaceutical sector plans to inject $2 billion into GMP infrastructure, facilitating compliance with rigorous QC standards and enhancing manufacturing capacity for cell and gene therapies.
- Increasing Investments from Pharmaceutical Firms: Pharmaceutical companies are investing heavily in cell and gene therapy manufacturing across Asia-Pacific, with annual investments exceeding $1.5 billion by the end of 2024. This includes Pfizers establishment of a $200 million facility in Singapore aimed at enhancing QC in gene therapy production. Such investments from prominent industry players create an avenue for growth, fostering advancements in QC methodologies and infrastructure throughout the region.
Scope of the Report
|
By Product Type |
Reagents & Kits |
|
By Application |
CAR-T Cell Therapy |
|
By QC Methodology |
Potency Testing |
|
By End User |
Pharmaceutical Companies |
|
By Region |
China |
Products
Key Target Audience
Pharmaceutical and Biopharmaceutical Companies
Research and Development Institutions
Contract Manufacturing Organizations (CMOs)
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., PMDA, NMPA)
Clinical Laboratories
Quality Control Solution Providers
Healthcare Providers and Hospitals
Companies
Players Mentioned in the Report:
Charles River Laboratories
Catalent Inc.
WuXi AppTec
Lonza Group AG
Thermo Fisher Scientific
BioReliance (Merck KGaA)
SGS SA
Eurofins Scientific
ICON plc
Covance Inc.
Table of Contents
1. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Overview
1.1 Definition and Scope
1.2 Market Taxonomy
1.3 Key Industry Standards and Regulations
1.4 Market Segmentation Overview
2. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Size (in USD Mn)
2.1 Historical Market Size
2.2 Year-On-Year Growth Analysis
2.3 Milestones in Market Development
2.4 Key Technological Advancements
3. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Analysis
3.1 Growth Drivers
3.1.1 Increase in Clinical Trials for Cell and Gene Therapies
3.1.2 Advances in Cell and Gene Therapy Technologies
3.1.3 Rising Demand for Specialized QC Services
3.1.4 Government and Regulatory Support
3.2 Market Challenges
3.2.1 High Costs of Quality Control (QC) in Cell and Gene Therapy
3.2.2 Limited Skilled Workforce for Advanced QC Methods
3.2.3 Regulatory Compliance Complexities
3.3 Opportunities
3.3.1 Expansion of GMP-Compliant Manufacturing Facilities
3.3.2 Increasing Investments from Pharmaceutical Firms
3.3.3 Emerging Markets for Cell and Gene Therapy
3.4 Trends
3.4.1 Adoption of Automated and Digital QC Methods
3.4.2 Integration with AI and Big Data in QC Processes
3.4.3 Growth of Decentralized Manufacturing Models
3.5 Government Regulation
3.5.1 Regulatory Bodies and Quality Standards (FDA, EMA, PMDA)
3.5.2 Guidelines on Cell and Gene Therapy Manufacturing
3.5.3 Compliance Standards for QC Laboratories
3.6 SWOT Analysis
3.7 Ecosystem Mapping
3.8 Porters Five Forces Analysis
3.9 Competitive Landscape
4. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Segmentation
4.1 By Product Type (in Value %)
4.1.1 Reagents & Kits
4.1.2 Instruments & Software
4.1.3 Consumables
4.2 By Application (in Value %)
4.2.1 CAR-T Cell Therapy
4.2.2 Gene Therapy
4.2.3 Stem Cell Therapy
4.3 By QC Methodology (in Value %)
4.3.1 Potency Testing
4.3.2 Purity Testing
4.3.3 Safety and Identity Testing
4.4 By End User (in Value %)
4.4.1 Pharmaceutical Companies
4.4.2 Research Institutions
4.4.3 Contract Manufacturing Organizations (CMOs)
4.5 By Region (in Value %)
4.5.1 China
4.5.2 Japan
4.5.3 South Korea
4.5.4 India
4.5.5 Australia
5. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Competitive Analysis
5.1 Profiles of Key Competitors
5.1.1 Charles River Laboratories
5.1.2 Catalent Inc.
5.1.3 WuXi AppTec
5.1.4 Lonza Group AG
5.1.5 BioReliance (Merck KGaA)
5.1.6 Thermo Fisher Scientific Inc.
5.1.7 SGS SA
5.1.8 Eurofins Scientific
5.1.9 ICON plc
5.1.10 Covance Inc.
5.1.11 PPD, Inc.
5.1.12 Novartis AG
5.1.13 Roche Diagnostics
5.1.14 Fujifilm Diosynth Biotechnologies
5.1.15 Takara Bio Inc.
5.2 Cross-Comparison Parameters (Employee Count, Revenue, R&D Investment, Market Share, Facility Presence, Key Partnerships, Regulatory Certifications, Innovation Focus)
5.3 Market Share Analysis
5.4 Strategic Initiatives
5.5 Mergers & Acquisitions
5.6 Investment Trends and Funding Analysis
5.7 Government Grants and Support Programs
6. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Regulatory Framework
6.1 Compliance Standards for Cell and Gene Therapy Manufacturing
6.2 Certification Requirements for QC Laboratories
6.3 Impact of Emerging Regulations
7. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Future Market Size (in USD Mn)
7.1 Projected Market Growth
7.2 Key Factors Impacting Future Market Growth
8. Asia-Pacific Cell and Gene Therapy Manufacturing QC Market Analysts Recommendations
8.1 Total Addressable Market (TAM), Serviceable Available Market (SAM), and Serviceable Obtainable Market (SOM)
8.2 Target Market Entry Strategies
8.3 Key Areas for Investment
8.4 White Space Opportunity Analysis
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
The initial phase involves constructing an ecosystem map encompassing all primary stakeholders in the Asia-Pacific cell and gene therapy manufacturing QC market. This includes gathering industry-level information to define critical variables impacting market dynamics, such as technology integration and regulatory changes.
Step 2: Market Analysis and Construction
In this phase, historical data for the market is compiled and analyzed. This includes examining the distribution and demand for QC solutions, as well as revenue trends, ensuring a reliable foundation for revenue estimates.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are validated through interviews with industry experts. Insights from consultations provide operational perspectives, refining the accuracy of data and clarifying market dynamics.
Step 4: Research Synthesis and Final Output
The final phase synthesizes all gathered data, integrating input from biomanufacturers to verify product segment details and performance statistics. This ensures a comprehensive and validated analysis of the Asia-Pacific cell and gene therapy manufacturing QC market.
Frequently Asked Questions
1. How big is the Asia-Pacific Cell and Gene Therapy Manufacturing QC Market?
The Asia-Pacific cell and gene therapy manufacturing QC market is valued at USD 426.5 Million, driven by an increase in biomanufacturing facilities and demand for precise quality control solutions.
2. What are the challenges in the Asia-Pacific Cell and Gene Therapy Manufacturing QC Market?
Key challenges in the Asia-Pacific cell and gene therapy manufacturing QC market include high operational costs for QC compliance, a limited skilled workforce, and complex regulatory standards across countries.
3. Who are the major players in the Asia-Pacific Cell and Gene Therapy Manufacturing QC Market?
Major players in the Asia-Pacific cell and gene therapy manufacturing QC market include Charles River Laboratories, Catalent Inc., WuXi AppTec, Lonza Group AG, and Thermo Fisher Scientific, leading due to advanced infrastructure and regulatory partnerships.
4. What are the growth drivers for the Asia-Pacific Cell and Gene Therapy Manufacturing QC Market?
Growth drivers in the Asia-Pacific cell and gene therapy manufacturing QC market include an increase in R&D investments for cell and gene therapy, supportive government policies, and advances in QC technologies, particularly automation.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















