
Asia-Pacific Non-Invasive Prenatal Testing (NIPT) Market Outlook to 2030
Region:Asia
Author(s):Paribhasha Tiwari
Product Code:KROD7365
December 2024
86
About the Report
Asia-Pacific Non-Invasive Prenatal Testing (NIPT) Market Overview
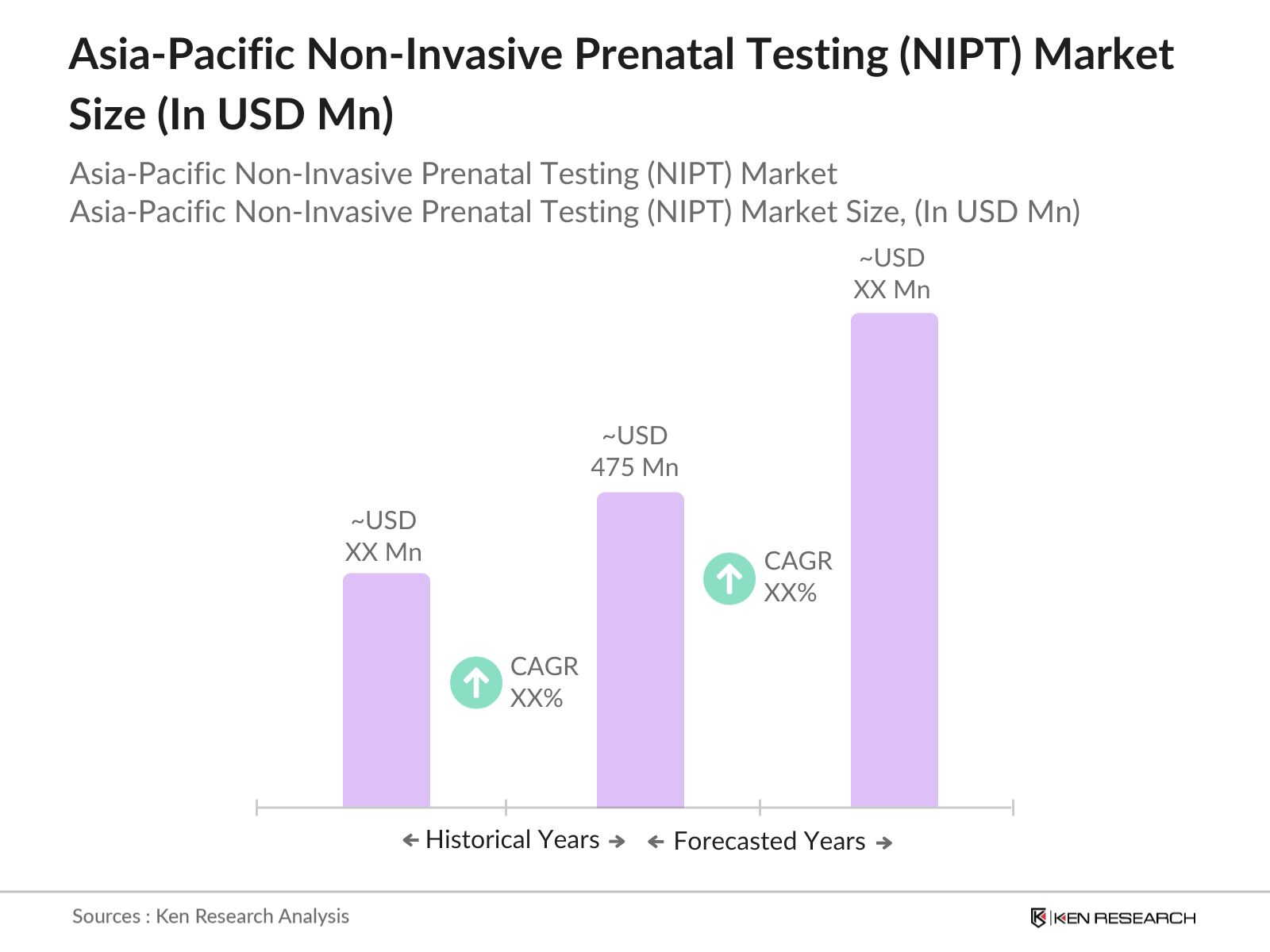
- The Asia-Pacific NIPT market, valued at USD 475 million, shows steady growth driven by a rising awareness of prenatal health and advances in genetic screening technologies. The availability of highly accurate, non-invasive procedures has increased patient preference for NIPT, especially among the urban population across high-growth markets. This demand is further fueled by a growing healthcare infrastructure and supportive government policies that emphasize prenatal screening.
- Key markets within the Asia-Pacific region include China, Japan, and Australia, where high levels of healthcare spending, advanced genetic research capabilities, and supportive regulatory frameworks bolster the markets growth. China, in particular, shows dominance due to its large population, expanding healthcare sector, and increasing government initiatives aimed at improving prenatal health.
- Regulatory bodies in countries like Australia are providing workshops and training programs focused on food safety standards for small mobile food truck operators. In 2024, these workshops helped over 2,000 vendors in Australia meet compliance requirements, reducing the risk of fines and fostering a reputation for safe food practices.
Asia-Pacific Non-Invasive Prenatal Testing (NIPT) Market Segmentation
By Product Type: The Asia-Pacific NIPT market is segmented by product type into kits and consumables, instruments, and software solutions. Recently, kits and consumables have a dominant market share under this segmentation, attributed to their frequent usage in routine testing and the recurrent need to replace them. This segment benefits from high demand due to the established infrastructure and ease of integration into existing healthcare workflows. Consumable manufacturers have also strengthened supply chains, making products readily available across urban and rural settings.
By Testing Technology: The Asia-Pacific NIPT market is categorized by testing technology into cfDNA testing, next-generation sequencing (NGS), and quantitative real-time PCR (qPCR). cfDNA testing holds a significant market share due to its high accuracy, minimally invasive nature, and expanding application in detecting a variety of genetic anomalies. This technology is preferred by healthcare providers as it allows early detection and thus enables timely medical interventions, enhancing patient outcomes and lowering long-term healthcare costs.
Asia-Pacific Non-Invasive Prenatal Testing (NIPT) Market Competitive Landscape
The Asia-Pacific NIPT market is dominated by a few key players with established global and regional footprints. These companies focus on innovations in testing technologies and collaborative partnerships with regional healthcare providers to expand their market presence.
Asia-Pacific Non-Invasive Prenatal Testing (NIPT) Market Analysis
Growth Drivers
- Increasing Prevalence of Genetic Disorders: The prevalence of genetic disorders in the Asia-Pacific region has been escalating, with more than 5 million newborns affected by genetic or congenital conditions annually. This surge has prompted greater demand for non-invasive prenatal testing (NIPT) as early diagnosis becomes critical for effective intervention. National health statistics from regional healthcare bodies highlight that genetic screening and diagnostics are increasingly prioritized, particularly in countries with high birth rates such as India and China.
- Rising Awareness on Prenatal Screening: Across Asia-Pacific, governments and health organizations are intensifying efforts to raise awareness about prenatal screening, resulting in higher acceptance rates for NIPT. Data from regional health ministries show a 20% increase in prenatal consultations related to NIPT in major urban centers, as populations increasingly seek non-invasive options for early-stage diagnosis of genetic disorders.
- Technological Advancements in Genomic Medicine: Advances in genomic sequencing and bioinformatics within Asia-Pacific are significantly boosting NIPTs diagnostic capabilities, enabling more precise screening for a broader range of genetic conditions. Over 1,000 healthcare institutions across the region have now incorporated next-generation sequencing technology, according to governmental health agency reports, enhancing the reliability and accessibility of NIPT services.
Market Challenges
- High Cost of NIPT Procedures: The average cost of NIPT across Asia-Pacific remains high, with estimates around USD 800-1,200 per test, making it unaffordable for a significant portion of the population. In countries where health insurance penetration is low, affordability becomes a major constraint, limiting access to NIPT services and slowing market growth.
- Limited Reimbursement Policies: A lack of comprehensive reimbursement frameworks for NIPT in many Asia-Pacific countries presents a substantial barrier to market expansion. In only a few countries, such as South Korea and Japan, are reimbursements available, covering about 30% of testing costs. For the majority of countries, out-of-pocket expenses remain the norm, limiting accessibility for low- and middle-income populations.
Asia-Pacific Non-Invasive Prenatal Testing (NIPT) Market Future Outlook
The Asia-Pacific NIPT market is anticipated to experience significant growth over the next five years, driven by technological advancements in non-invasive testing and increasing government support for prenatal healthcare initiatives. As awareness of genetic screening expands across both urban and rural regions, and more countries integrate NIPT into their national healthcare guidelines, the market is likely to see continued adoption and innovation in testing solutions.
Market Opportunities
- Expansion of Coverage in Government Health Plans: Government health plans in Asia-Pacific are increasingly incorporating NIPT services, with initiatives in countries like Singapore and Australia to expand coverage to broader population segments. For instance, Singapores government health services have introduced subsidies for NIPT, making it accessible to approximately 100,000 additional individuals annually.
- Growth in Non-Invasive Testing Technologies: There is a marked increase in investments in non-invasive testing technology across the region, with R&D expenditures in this field crossing USD 3 billion annually. These investments have led to the development of advanced NIPT methods, enhancing diagnostic accuracy and making them viable options for public health adoption.
Scope of the Report
|
By Product Type |
Instruments |
|
By Application |
Trisomy Detection |
|
By Testing Technology |
cfDNA Testing |
|
By End-User |
Hospitals and Clinics |
|
By Region |
China |
Products
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (Health Ministries, FDA-equivalent agencies)
Healthcare Providers (hospitals, diagnostic laboratories)
Research Institutions in Genomics
Biotechnology Firms
Medical Device Manufacturers
Public Health and Epidemiology Organizations
Pharmaceutical Companies
Companies
Players Mentioned in the Report:
Illumina Inc.
Roche Diagnostics
Thermo Fisher Scientific
Agilent Technologies
Natera Inc.
PerkinElmer Inc.
BGI Genomics
Sequenom Laboratories
Quest Diagnostics
Berry Genomics
Table of Contents
1. Asia-Pacific NIPT Market Overview
1.1 Definition and Scope
1.2 Market Taxonomy
1.3 Market Growth Rate
1.4 Market Segmentation Overview
2. Asia-Pacific NIPT Market Size (USD Million)
2.1 Historical Market Size
2.2 Year-On-Year Growth Analysis
2.3 Key Market Milestones and Developments
3. Asia-Pacific NIPT Market Dynamics
3.1 Growth Drivers
3.1.1 Increasing Prevalence of Genetic Disorders
3.1.2 Rising Awareness on Prenatal Screening
3.1.3 Technological Advancements in Genomic Medicine
3.1.4 Expanding Healthcare Infrastructure in Emerging Economies
3.2 Market Challenges
3.2.1 High Cost of NIPT Procedures
3.2.2 Limited Reimbursement Policies
3.2.3 Ethical and Regulatory Issues
3.2.4 Lack of Skilled Genetic Counselors
3.3 Opportunities
3.3.1 Expansion of Coverage in Government Health Plans
3.3.2 Growth in Non-Invasive Testing Technologies
3.3.3 Increase in Research and Development Investments
3.4 Trends
3.4.1 Integration of AI in Genetic Testing
3.4.2 Increased Demand for Customized Testing Options
3.4.3 Use of cfDNA-based Screening Methods
3.5 Regulatory Framework
3.5.1 Guidelines by Regulatory Authorities
3.5.2 Compliance and Standardization Requirements
3.6 Competitive Ecosystem
3.6.1 Market Stakeholders and Role Distribution
3.6.2 Porters Five Forces Analysis
3.6.3 SWOT Analysis
4. Asia-Pacific NIPT Market Segmentation
4.1 By Product Type
4.1.1 Instruments
4.1.2 Kits and Consumables
4.1.3 Software Solutions
4.2 By Application
4.2.1 Trisomy Detection
4.2.2 Sex Chromosome Aneuploidy Detection
4.2.3 Microdeletions Syndrome Detection
4.3 By Testing Technology
4.3.1 cfDNA Testing
4.3.2 Next-Generation Sequencing (NGS)
4.3.3 Quantitative Real-Time PCR (qPCR)
4.4 By End-User
4.4.1 Hospitals and Clinics
4.4.2 Diagnostic Laboratories
4.4.3 Specialty Clinics
4.5 By Region
4.5.1 China
4.5.2 Japan
4.5.3 Australia
4.5.4 South Korea
4.5.5 India
5.Asia-Pacific NIPT Market Competitive Landscape
5.1 Detailed Company Profiles
5.1.1 Illumina Inc.
5.1.2 Roche Diagnostics
5.1.3 Thermo Fisher Scientific
5.1.4 Agilent Technologies
5.1.5 Natera Inc.
5.1.6 PerkinElmer Inc.
5.1.7 BGI Genomics
5.1.8 Sequenom Laboratories
5.1.9 Quest Diagnostics
5.1.10 Myriad Genetics
5.1.11 Eurofins Scientific
5.1.12 Berry Genomics
5.1.13 Laboratory Corporation of America Holdings
5.1.14 Bio-Rad Laboratories
5.1.15 Fulgent Genetics
5.2 Cross-Comparison Parameters (Revenue, No. of Employees, Global Reach, Product Portfolio, R&D Investment, Collaborations, Market Position, Customer Base)
5.3 Market Share Analysis
5.4 Strategic Initiatives
5.5 Mergers and Acquisitions
5.6 Joint Ventures and Partnerships
6. Asia-Pacific NIPT Market Regulatory and Ethical Framework
6.1 Regional Regulatory Bodies and Guidelines
6.2 Compliance Requirements and Standards
6.3 Ethical Considerations in Prenatal Testing
7. Asia-Pacific NIPT Future Market Size (USD Million)
7.1 Projected Market Size
7.2 Key Factors Influencing Future Market Growth
8. Analyst Recommendations for Asia-Pacific NIPT Market
8.1 Total Addressable Market (TAM), Serviceable Available Market (SAM), and Serviceable Obtainable Market (SOM) Analysis
8.2 Patient Cohort Analysis
8.3 Growth and Investment Opportunities
8.4 Innovation White Space Identification
Research Methodology
Step 1: Identification of Key Variables
This initial phase entails mapping out the NIPT market ecosystem within Asia-Pacific, including stakeholders such as genetic testing companies, healthcare providers, and regulatory agencies. A thorough review of secondary sources is conducted to identify the primary factors influencing market growth and dynamics.
Step 2: Market Analysis and Construction
In this phase, a compilation of historical data on market penetration and adoption trends within the Asia-Pacific region is undertaken. Parameters such as market revenue, product usage statistics, and regional adoption rates are analyzed to construct accurate market forecasts.
Step 3: Hypothesis Validation and Expert Consultation
Key market hypotheses are validated through consultations with industry experts using CATI methods. Insights from leading genetic testing companies and healthcare providers help refine market data, ensuring precision in projections.
Step 4: Research Synthesis and Final Output
Final data integration involves corroborating bottom-up and top-down research findings. Extensive interviews with NIPT product providers enable a synthesis of market insights, producing a comprehensive and validated analysis of the Asia-Pacific NIPT market.
Frequently Asked Questions
01. How big is the Asia-Pacific Non-Invasive Prenatal Testing (NIPT) Market?
The Asia-Pacific NIPT market is valued at USD 475 million, driven by technological advancements in prenatal diagnostics and increasing healthcare investments across the region.
02. What are the primary challenges in the Asia-Pacific NIPT Market?
Key challenges in the Asia-Pacific NIPT market include high procedural costs, limited accessibility in certain regions, and regulatory hurdles. Additionally, the demand for skilled genetic counselors remains unmet in some countries, impacting market reach.
03. Who are the leading players in the Asia-Pacific NIPT Market?
Major players in the Asia-Pacific NIPT market include Illumina Inc., Roche Diagnostics, Thermo Fisher Scientific, and BGI Genomics, known for their extensive product portfolios, innovative technologies, and established market presence.
04. What drives the growth of the Asia-Pacific NIPT Market?
Growth of Asia-Pacific NIPT market is fueled by factors such as rising awareness of prenatal health, government initiatives for maternal health programs, and advancements in genetic sequencing technology, enhancing diagnostic precision.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.