
Asia Pacific Psoriasis Drugs Market Outlook to 2030
Region:Asia
Author(s):Paribhasha Tiwari
Product Code:KROD10547
November 2024
93
About the Report
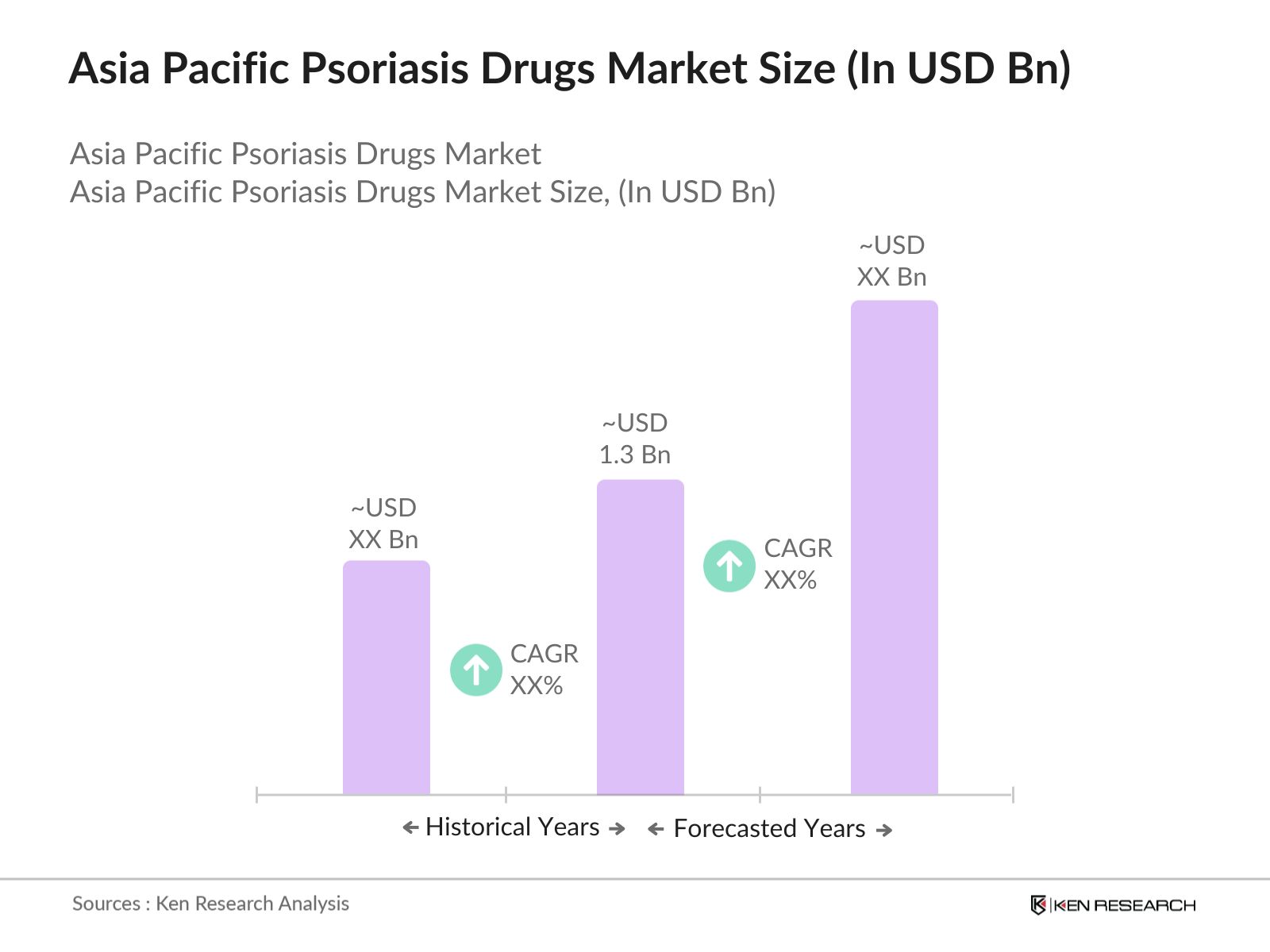
Asia Pacific Psoriasis Drugs Market Overview
- The Asia Pacific Psoriasis Drugs Market is valued at USD 1.3 billion, driven by several key factors. One of the primary growth drivers is the increasing prevalence of psoriasis across the region. Governments in countries such as China and India are allocating more funds toward healthcare infrastructure, and investments in research and development are promoting the development of biologics and other advanced therapies for psoriasis treatment. This consistent investment and rising awareness among patients about the importance of early treatment are pushing the market forward.
- The dominant markets in the Asia Pacific region include China, Japan, and India. China leads due to its vast population and expanding healthcare infrastructure, while Japan's dominance is attributed to its advanced healthcare system and high demand for biologics. India's growing pharmaceutical sector and government initiatives to improve access to healthcare are also critical factors. These countries dominate the market due to their robust pharmaceutical industries, government support, and large patient populations.
- Japan has launched a new program in 2024 promoting the use of biologic therapies through public healthcare. Under this initiative, biologic treatments for psoriasis will be covered up to 75% by insurance, allowing nearly 150,000 patients to access these therapies by 2025.
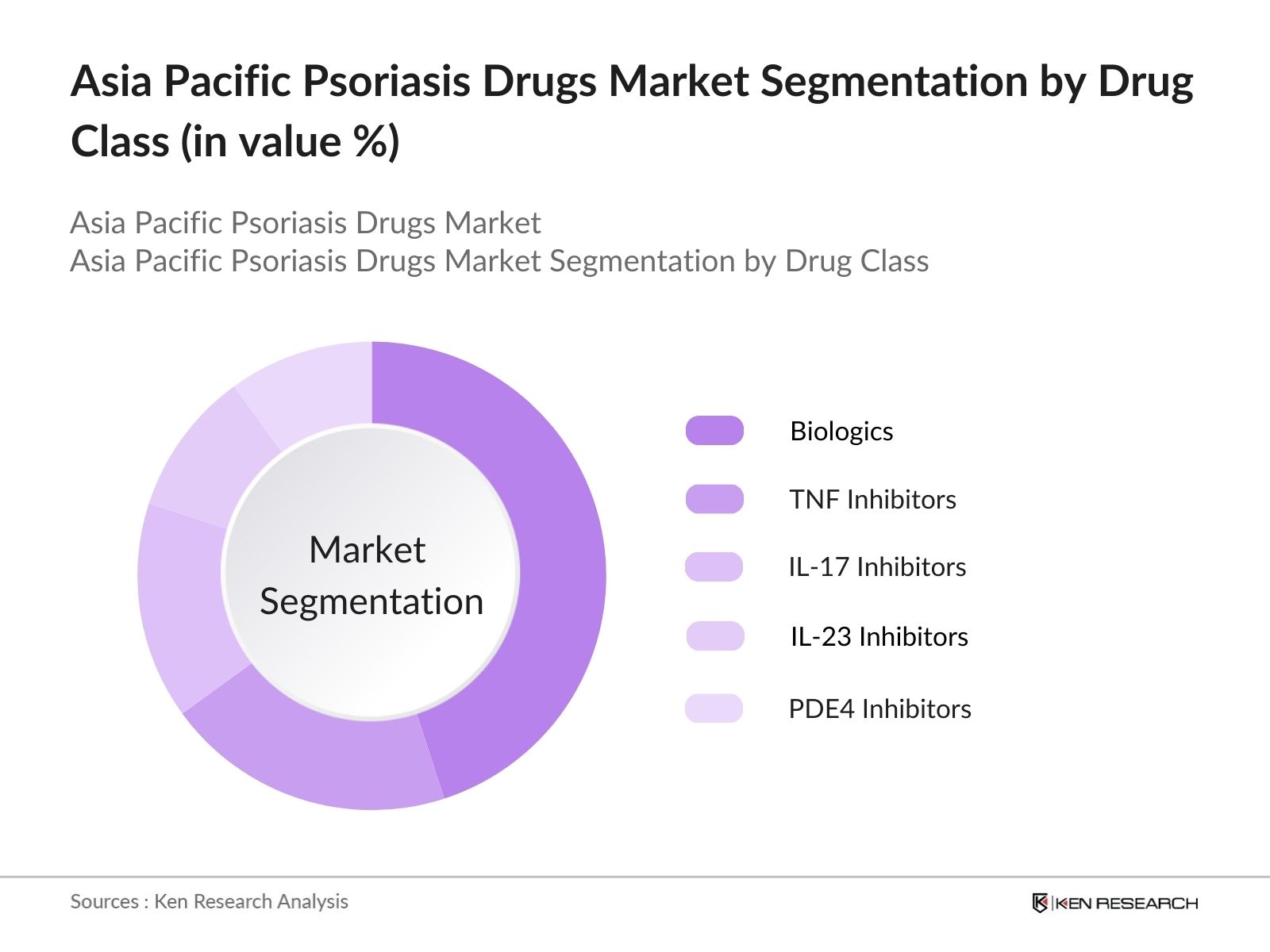
Asia Pacific Psoriasis Drugs Market Segmentation
By Drug Class: The Asia Pacific Psoriasis Drugs Market is segmented by drug class into biologics, TNF inhibitors, IL-17 inhibitors, IL-23 inhibitors, and PDE4 inhibitors. Recently, biologics have dominated the market in this segment, owing to their targeted approach in treating moderate to severe psoriasis. Biologics, which include drugs like adalimumab and secukinumab, have shown high efficacy rates and fewer side effects compared to traditional systemic therapies. This has led to an increasing adoption of biologics among dermatologists in key countries like China and Japan. The dominance of biologics is also supported by ongoing clinical research and new product launches.
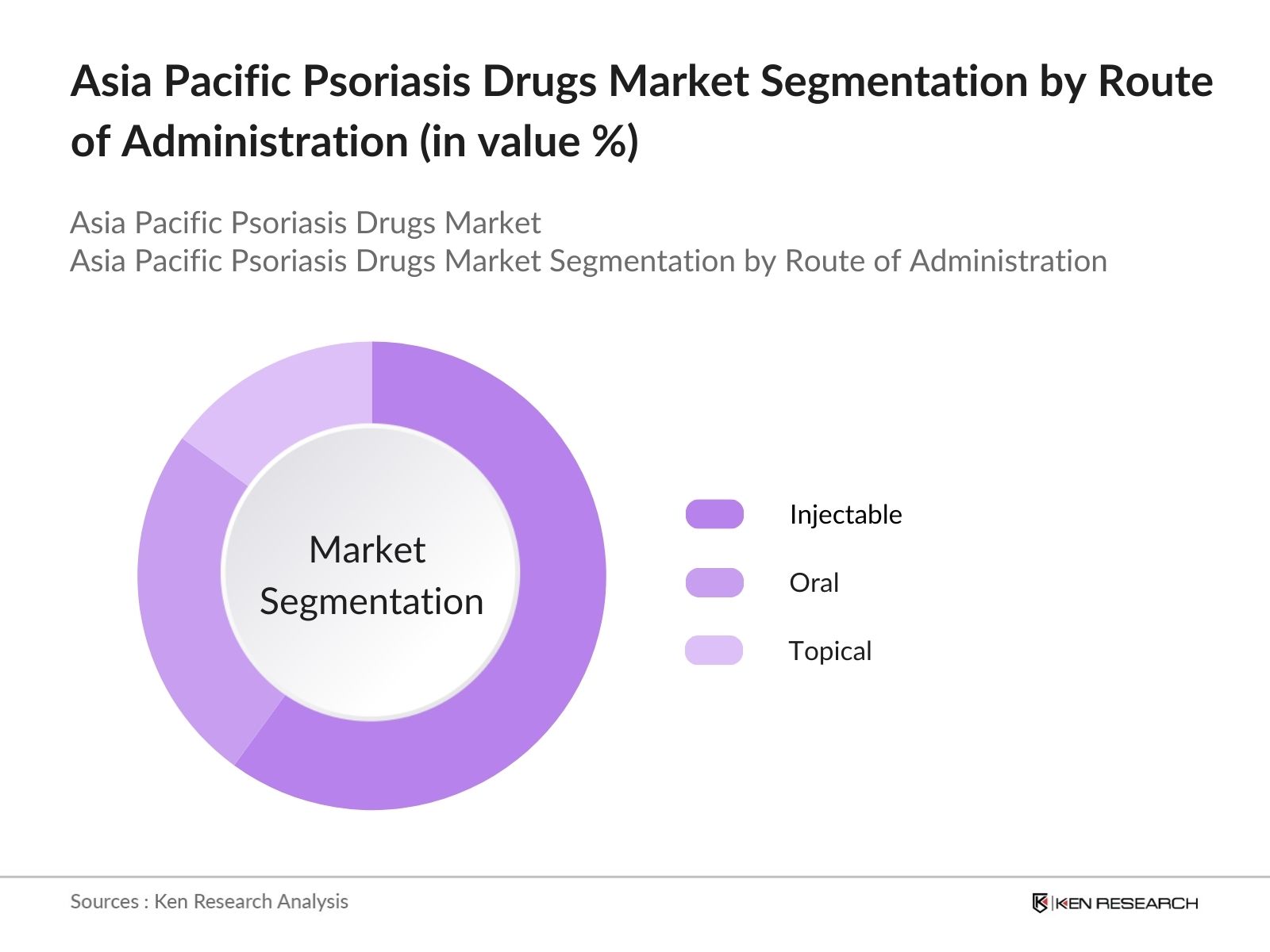
By Route of Administration: The market is segmented by route of administration into oral, injectable, and topical treatments. Injectable therapies have gained the largest market share in 2023, driven by the increased adoption of biologics and the ease of administration offered by self-injecting devices. This growth is also attributed to the high effectiveness of injectable therapies for patients with moderate to severe psoriasis. Moreover, the development of long-acting injectables has contributed to patient adherence and better outcomes, further driving this segment's dominance in the Asia Pacific region.
Asia Pacific Psoriasis Drugs Market Competitive Landscape
The Asia Pacific Psoriasis Drugs Market is dominated by several global and regional players. These companies are focusing on expanding their product portfolios and investing in research and development to enhance the efficacy of their therapies. The market is relatively consolidated, with a few key players leading the competition due to their established market presence and innovative product offerings.
The Asia Pacific Psoriasis Drugs Market is highly competitive, with companies like AbbVie Inc., Amgen Inc., and Novartis AG leading the charge due to their strong presence in biologic therapies. Regional companies such as Sun Pharmaceuticals are also making significant strides in the market, particularly in the generic drugs and biosimilar segments. This competition is driving innovation, with key players investing heavily in R&D to maintain a competitive edge.
Asia Pacific Psoriasis Drugs Market Analysis
Growth Drivers
- Increasing Prevalence of Psoriasis The Asia Pacific region is witnessing a growing number of psoriasis cases, with an estimated 8-12 million people affected across countries like India, China, and Japan. Rising awareness and better diagnostic tools have contributed to an increase in reported cases. According to a 2024 WHO report, healthcare institutions in India alone are seeing over 250,000 new psoriasis cases annually, driving the need for effective treatment options.
- Advancements in Biologic Therapies Biologic drugs are becoming more accessible in the Asia Pacific market, with countries like Japan and Australia leading the adoption. By 2023, approximately 450,000 patients in the region had been treated with biologics, a 20% increase in usage compared to five years ago, as noted by government healthcare databases. These therapies are designed to target specific immune responses in patients, making them highly effective in controlling psoriasis.
- Rising Government Healthcare Expenditure Government spending on healthcare across Asia Pacific is increasing, with a combined investment of over $500 billion in 2024 aimed at improving treatment accessibility. Countries such as China and India have ramped up their healthcare budgets, facilitating better patient access to psoriasis treatments through public health schemes and insurance coverage.
Market Challenges
- High Cost of Biologics The cost of biologic treatments remains a significant barrier in the region, particularly in developing countries like Indonesia and Vietnam, where annual treatment costs can range between $10,000 and $20,000 per patient. While biologics offer effective results, limited government subsidies make them unaffordable for a large portion of the population, curbing widespread adoption.
- Lack of Skilled Dermatologists in Emerging Economies The availability of qualified dermatologists in countries like the Philippines and Bangladesh is insufficient to meet the rising demand for psoriasis treatments. According to 2024 government health reports, there are less than 0.5 dermatologists per 100,000 population in these countries, severely limiting patient access to specialized care and delaying diagnosis.
Asia Pacific Psoriasis Drugs Market Future Outlook
The Asia Pacific Psoriasis Drugs Market is expected to witness robust growth over the next five years. Factors such as increased healthcare spending, continuous advancements in biologic therapies, and rising awareness among patients will drive this growth. Moreover, the introduction of biosimilars and innovative treatment options, such as small-molecule drugs, will open up new opportunities for companies operating in the market. Governments across the region are also supporting the development of healthcare infrastructure, which will further boost access to psoriasis treatments.
Market Opportunities
- Expansion of Healthcare Infrastructure in Developing Economies Countries like India, Thailand, and Malaysia are investing in expanding their healthcare infrastructure, with over $15 billion allocated for 2024 projects aimed at building new healthcare centers and hospitals in rural areas. This will significantly improve access to psoriasis treatment in remote regions and increase the patient pool.
- Increasing Focus on Personalized Medicine Governments and healthcare institutions in Japan and South Korea are pushing for advancements in personalized medicine, which offers tailored treatment plans based on individual patient profiles. By 2025, it is estimated that over 150,000 patients in these countries will receive personalized treatments, which is expected to enhance treatment efficacy for psoriasis sufferers.
Scope of the Report
|
By Drug Class |
Biologics TNF Inhibitors IL-17 Inhibitors IL-23 Inhibitors PDE4 Inhibitors |
|
By Route of Administration |
Oral Injectable Topical |
|
By Disease Type |
Plaque Psoriasis Guttate Psoriasis Inverse Psoriasis Pustular Psoriasis Erythrodermic Psoriasis |
|
By Distribution Channel |
Hospital Pharmacies Retail Pharmacies Online Pharmacies |
|
By Region |
China India Japan Australia Southeast Asia |
Products
Key Target Audience
Pharmaceutical Manufacturers
Biologic Drug Developers
Dermatology Clinics
Healthcare Providers
Hospitals and Specialty Clinics
Government and Regulatory Bodies (e.g., National Medical Products Administration, Pharmaceuticals and Medical Devices Agency)
Investors and Venture Capitalist Firms
Healthcare Technology Providers
Companies
Players Mentioned in the Report:
AbbVie Inc.
Amgen Inc.
Novartis AG
Pfizer Inc.
Sun Pharmaceutical Industries Ltd.
Eli Lilly and Company
Johnson & Johnson
UCB S.A.
Merck & Co., Inc.
Bausch Health Companies Inc.
Table of Contents
1. Asia Pacific Psoriasis Drugs Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2. Asia Pacific Psoriasis Drugs Market Size (In USD Bn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
3. Asia Pacific Psoriasis Drugs Market Analysis
3.1. Growth Drivers (Regulatory Approvals, Technological Advancements, Increasing Patient Population)
3.1.1. Increasing Prevalence of Psoriasis
3.1.2. Advancements in Biologic Therapies
3.1.3. Rising Government Healthcare Expenditure
3.1.4. Growing R&D Investments in Immunotherapy
3.2. Market Challenges (High Treatment Costs, Limited Accessibility in Remote Areas)
3.2.1. High Cost of Biologics
3.2.2. Lack of Skilled Dermatologists in Emerging Economies
3.2.3. Regulatory Hurdles in Drug Approvals
3.3. Opportunities (Emerging Biosimilars, Expansion of Healthcare Infrastructure)
3.3.1. Expansion of Healthcare Infrastructure in Developing Economies
3.3.2. Increasing Focus on Personalized Medicine
3.3.3. Favorable Reimbursement Policies
3.3.4. Opportunities in Untapped Markets
3.4. Trends (Shift Towards Biologics, Rise of Telemedicine in Dermatology)
3.4.1. Adoption of Biologics for Severe Psoriasis
3.4.2. Increasing Use of Telehealth for Psoriasis Management
3.4.3. Development of Oral Small-Molecule Drugs
3.4.4. Digital Platforms for Patient Engagement and Monitoring
3.5. Government Initiatives (Healthcare Regulations, Drug Price Control)
3.5.1. National Drug Reimbursement Policies
3.5.2. Price Regulations on Biologics
3.5.3. Psoriasis Awareness Programs
3.5.4. Government Support for Research & Development
3.6. SWOT Analysis
3.7. Stakeholder Ecosystem (Psoriasis Drug Manufacturers, Healthcare Providers)
3.8. Porters Five Forces
3.9. Competition Ecosystem
4. Asia Pacific Psoriasis Drugs Market Segmentation
4.1. By Drug Class (In Value %)
4.1.1. Biologics
4.1.2. TNF Inhibitors
4.1.3. IL-17 Inhibitors
4.1.4. IL-23 Inhibitors
4.1.5. PDE4 Inhibitors
4.2. By Route of Administration (In Value %)
4.2.1. Oral
4.2.2. Injectable
4.2.3. Topical
4.3. By Disease Type (In Value %)
4.3.1. Plaque Psoriasis
4.3.2. Guttate Psoriasis
4.3.3. Inverse Psoriasis
4.3.4. Pustular Psoriasis
4.3.5. Erythrodermic Psoriasis
4.4. By Distribution Channel (In Value %)
4.4.1. Hospital Pharmacies
4.4.2. Retail Pharmacies
4.4.3. Online Pharmacies
4.5. By Region (In Value %)
4.5.1. China
4.5.2. India
4.5.3. Japan
4.5.4. Australia
4.5.5. Southeast Asia
5. Asia Pacific Psoriasis Drugs Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. AbbVie Inc.
5.1.2. Amgen Inc.
5.1.3. Novartis AG
5.1.4. Pfizer Inc.
5.1.5. Johnson & Johnson
5.1.6. Eli Lilly and Company
5.1.7. UCB S.A.
5.1.8. Merck & Co., Inc.
5.1.9. Sun Pharmaceutical Industries Ltd.
5.1.10. Bausch Health Companies Inc.
5.1.11. LEO Pharma
5.1.12. Biocon Ltd.
5.1.13. Cipla Inc.
5.1.14. Glenmark Pharmaceuticals
5.1.15. Dr. Reddys Laboratories Ltd.
5.2. Cross Comparison Parameters (Number of Approved Drugs, R&D Investment, Strategic Collaborations, Number of Patents, Revenue, Psoriasis Drug Sales, Market Penetration, Geographical Presence)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers And Acquisitions
5.6. Investment Analysis
5.7. Government Grants
5.8. Venture Capital Funding
6. Asia Pacific Psoriasis Drugs Market Regulatory Framework
6.1. Drug Approval Process
6.2. Psoriasis Drug Pricing Regulations
6.3. Regulatory Compliance for Psoriasis Drug Manufacturers
7. Asia Pacific Psoriasis Drugs Future Market Size (In USD Bn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. Asia Pacific Psoriasis Drugs Future Market Segmentation
8.1. By Drug Class (In Value %)
8.2. By Route of Administration (In Value %)
8.3. By Disease Type (In Value %)
8.4. By Distribution Channel (In Value %)
8.5. By Region (In Value %)
9. Asia Pacific Psoriasis Drugs Market Analysts Recommendations
9.1. TAM/SAM/SOM Analysis
9.2. White Space Opportunity Analysis
9.3. Customer Cohort Analysis
9.4. Marketing InitiativesDisclaimer
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
The initial phase of the research involves constructing an ecosystem map that includes all stakeholders within the Asia Pacific Psoriasis Drugs Market. Extensive desk research is conducted using secondary databases to gather comprehensive market-level data. This helps in identifying and defining the variables that drive the market, such as drug development timelines, treatment costs, and regulatory frameworks.
Step 2: Market Analysis and Construction
In this phase, historical data related to psoriasis drug adoption and market penetration is analyzed. The analysis also includes evaluating regional differences in drug availability, healthcare infrastructure, and treatment preferences across key countries in the region. This step ensures accuracy in estimating market performance.
Step 3: Hypothesis Validation and Expert Consultation
Industry experts, including dermatologists, pharmaceutical executives, and healthcare regulators, are consulted to validate the hypotheses developed during the research. This process is conducted through interviews and focus group discussions, which provide insights into market dynamics, challenges, and opportunities from industry insiders.
Step 4: Research Synthesis and Final Output
The final phase involves synthesizing the data and insights collected during the previous steps. The report includes a comprehensive analysis of product segments, market trends, and competitor strategies, ensuring that the final output is detailed, accurate, and actionable for stakeholders in the Asia Pacific Psoriasis Drugs Market.
Frequently Asked Questions
01. How big is the Asia Pacific Psoriasis Drugs Market?
The Asia Pacific Psoriasis Drugs Market is valued at USD 1.3 billion, driven by increasing healthcare investments and the growing prevalence of psoriasis across the region.
02. What are the challenges in the Asia Pacific Psoriasis Drugs Market?
Challenges in the Asia Pacific Psoriasis Drugs Market include the high cost of biologic therapies, regulatory barriers, and limited access to advanced treatments in rural areas.
03. Who are the major players in the Asia Pacific Psoriasis Drugs Market?
Key players in the Asia Pacific Psoriasis Drugs Market in the market include AbbVie Inc., Amgen Inc., Novartis AG, Pfizer Inc., and Sun Pharmaceutical Industries Ltd.
04. What are the growth drivers of the Asia Pacific Psoriasis Drugs Market?
The Asia Pacific Psoriasis Drugs Market is driven by rising awareness of psoriasis treatment options, increased R&D investments in biologics, and supportive government policies.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















