
Global Centesis Device Market Outlook to 2030
Region:Global
Author(s):Shubham
Product Code:KROD5573
November 2024
90
About the Report
Global Centesis Device Market Overview
The Global Centesis Device Market is valued at USD 430 million and is driven by the increasing prevalence of chronic diseases that require centesis procedures, such as pleural effusion, ascites, and pericardial effusion. Centesis devices play a crucial role in minimally invasive procedures, allowing healthcare providers to perform fluid drainage safely. Advancements in imaging technologies, such as ultrasound-guided centesis, have further fueled the demand for these devices. Additionally, the rising geriatric population, coupled with advancements in diagnostic tools, continues to drive the markets growth. 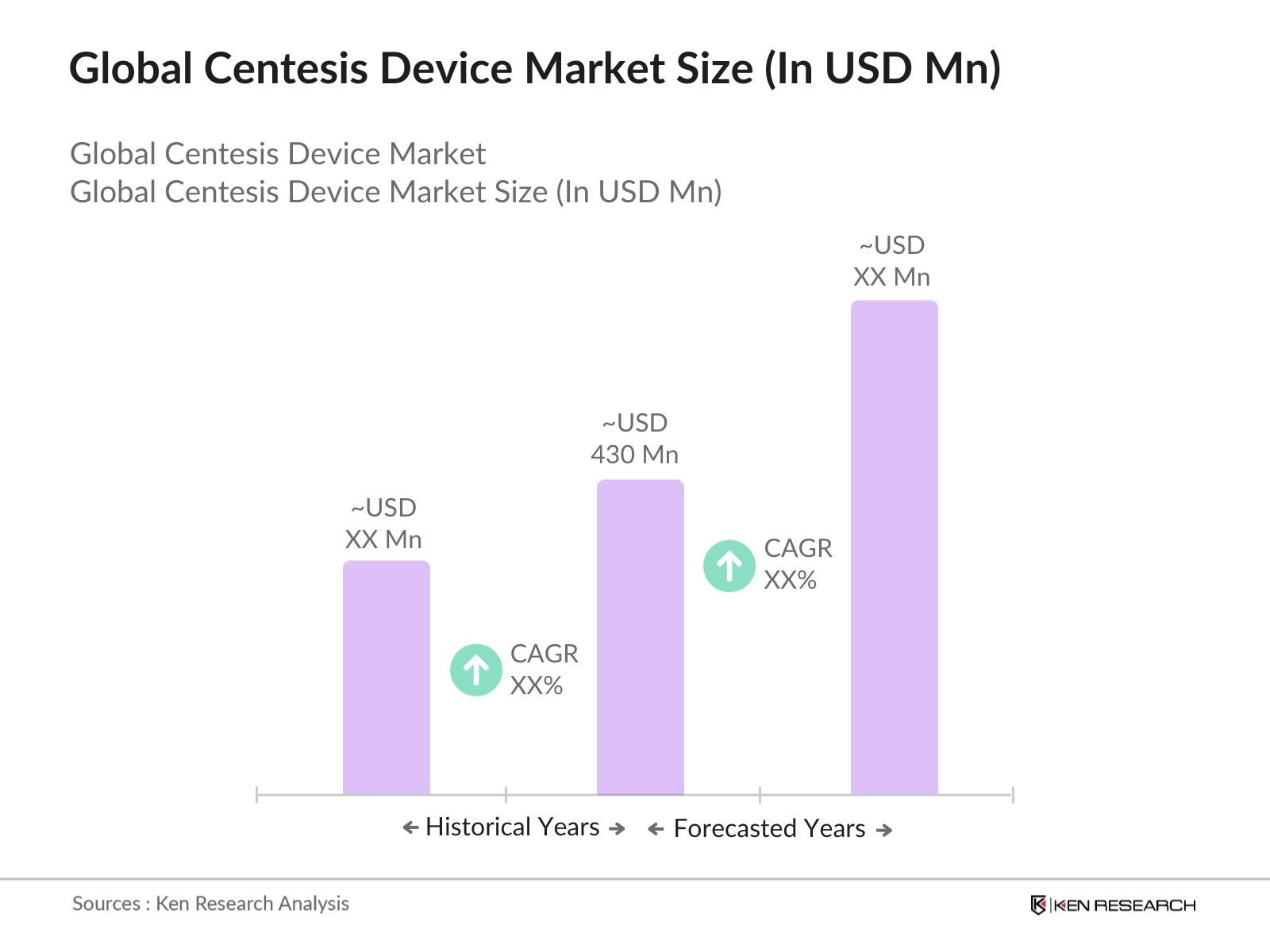 Key regions driving growth in the centesis device market include North America, Europe, and Asia Pacific. North America dominates the market due to a high prevalence of chronic diseases, advanced healthcare infrastructure, and robust healthcare spending. In Europe, demand is growing due to the regions aging population and advancements in medical technology. Meanwhile, the Asia Pacific region, led by China and India, is experiencing growth due to increasing healthcare investments, expanding healthcare infrastructure, and rising awareness of minimally invasive procedures.The U.S. FDA regulates centesis devices as Class II or III medical devices, depending on their intended use and complexity. As of 2024, the FDA's approval process requires extensive clinical data for devices like thoracentesis and paracentesis kits, which typically takes 12-18 months for approval. The FDA approved 15 new centesis-related devices in 2023, emphasizing the importance of compliance with stringent safety and efficacy standards to ensure market entry.
Key regions driving growth in the centesis device market include North America, Europe, and Asia Pacific. North America dominates the market due to a high prevalence of chronic diseases, advanced healthcare infrastructure, and robust healthcare spending. In Europe, demand is growing due to the regions aging population and advancements in medical technology. Meanwhile, the Asia Pacific region, led by China and India, is experiencing growth due to increasing healthcare investments, expanding healthcare infrastructure, and rising awareness of minimally invasive procedures.The U.S. FDA regulates centesis devices as Class II or III medical devices, depending on their intended use and complexity. As of 2024, the FDA's approval process requires extensive clinical data for devices like thoracentesis and paracentesis kits, which typically takes 12-18 months for approval. The FDA approved 15 new centesis-related devices in 2023, emphasizing the importance of compliance with stringent safety and efficacy standards to ensure market entry.
Global Centesis Device Market Segmentation
- By Procedure Type: The Market is segmented by procedure type into thoracentesis, paracentesis, amniocentesis, and arthrocentesis. Thoracentesis holds the dominant market share due to the increasing incidence of pleural effusion, often caused by lung cancer, heart failure, and infections. Thoracentesis is widely used for diagnostic and therapeutic purposes, making it a routine procedure in hospitals and diagnostic centers. The growing adoption of ultrasound guidance for thoracentesis has further strengthened its dominance in the market.
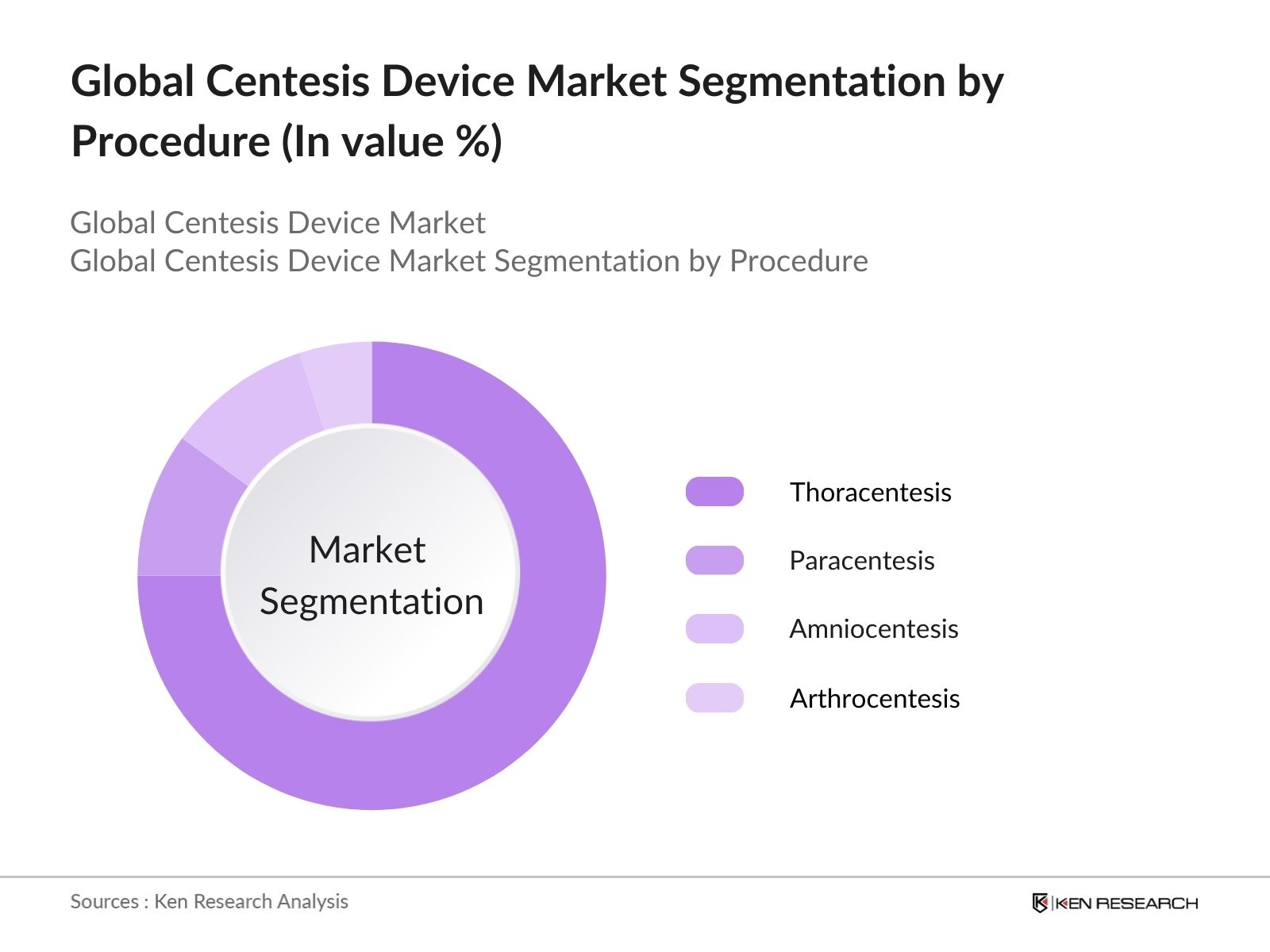
- By Product Type: The market is segmented by product type into centesis needles, drainage catheters, procedure trays, and other accessories. Centesis needles dominate the market due to their widespread use in various centesis procedures, such as thoracentesis and paracentesis. The increasing focus on patient safety and the introduction of innovative designs aimed at reducing pain and discomfort during procedures have contributed to the dominance of centesis needles. They are also favored for their precision in diagnostic and therapeutic applications.
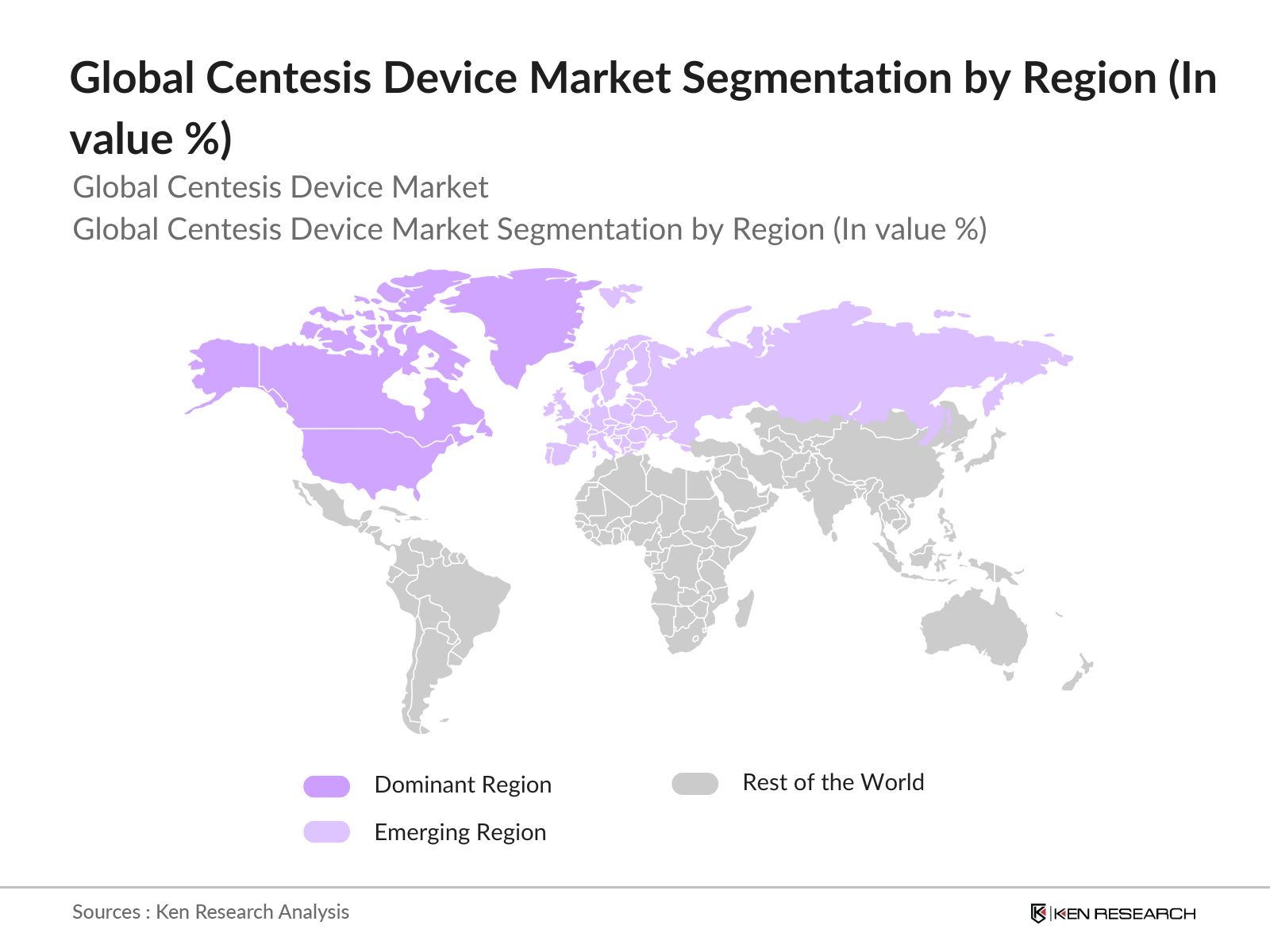
- By Region: The Market is segmented by region into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. North America holds the largest market share due to advanced healthcare infrastructure, high prevalence of chronic diseases such as cancer and cardiovascular conditions, and the growing number of minimally invasive procedures. The U.S. leads the market with increasing adoption of centesis devices for diagnostic and therapeutic purposes, supported by favorable reimbursement policies and well-established medical device manufacturers.
Global Centesis Device Market Competitive Landscape
The Global Centesis Device Market is highly consolidated, with a few major players dominating the market due to their extensive product portfolios, technological innovation, and strong relationships with healthcare providers. Key companies are continuously investing in research and development to enhance the precision and efficiency of their devices, particularly in improving patient safety and procedural outcomes. 
Global Centesis Device Market Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases: Chronic diseases such as cancer, kidney disorders, and liver diseases are becoming more prevalent worldwide. According to the World Health Organization, there were 19.3 million new cases of cancer globally in 2020, and this number is projected to rise as the population ages. Chronic kidney disease affected nearly 850 million people worldwide, with a significant portion requiring centesis procedures for diagnostic and therapeutic purposes. This increasing burden on healthcare systems highlights the demand for centesis devices, with a growing patient base needing minimally invasive treatments for fluid removal and biopsy procedures.
- Rising Demand for Minimally Invasive Procedures: The global healthcare sector has seen a shift towards minimally invasive procedures due to reduced recovery times, lower risks of complications, and improved patient outcomes. The number of minimally invasive procedures conducted in the U.S. increased by 25% between 2017 and 2022, with over 18 million procedures performed in 2022 alone. Centesis procedures, which include thoracentesis and paracentesis, have gained prominence in diagnostic and therapeutic interventions, aligning with the increased demand for less invasive medical interventions across healthcare systems.
- Growing Geriatric Population: The global geriatric population is rising steadily, and with age, the prevalence of diseases requiring centesis procedures also increases. In 2024, there were 800 million people aged 65 years or older worldwide, according to the United Nations. Aging increases susceptibility to conditions such as pleural effusions, ascites, and joint effusions, all of which may require centesis procedures. The growing elderly population in regions like Europe and Japan is leading to higher demand for centesis devices, contributing to the global market's growth.
Challenges
- High Costs Associated with Devices and Procedures: The costs associated with centesis devices and the procedures themselves pose significant barriers to market growth, particularly in low- and middle-income countries. These high costs limit the adoption of centesis procedures in regions with lower healthcare budgets or insufficient insurance coverage. The disparity in access to minimally invasive treatments creates challenges in expanding the use of centesis devices across different global markets.
- Stringent Regulatory Requirements: Centesis devices are classified as Class II or Class III medical devices in many regions, which necessitates rigorous regulatory scrutiny before they can be approved for market entry. In the U.S., the FDA mandates clinical trials for Class III devices to prove safety and efficacy, often resulting in lengthy approval processes. Similarly, in the European Union, the Medical Device Regulation (MDR) requires stricter post-market surveillance and compliance, increasing development costs and slowing the introduction of new devices.
Global Centesis Device Market Future Outlook
The Global Centesis Device Market is expected to experience steady growth, driven by increasing demand for minimally invasive procedures and the continuous advancements in medical imaging technology. As healthcare systems around the world continue to adopt more patient-centric approaches, the need for safe and effective centesis devices will rise. Moreover, emerging markets in Asia Pacific and Latin America are anticipated to present significant growth opportunities due to expanding healthcare infrastructure and rising investments in healthcare technology.
Future Market Opportunities
- Emerging Markets for Centesis Procedures: Emerging markets, particularly in Asia-Pacific and Latin America, are witnessing a surge in demand for centesis devices as healthcare infrastructure expands and medical tourism grows. Countries like India and Brazil are investing heavily in upgrading their healthcare systems, with India allocating USD 10.7 billion to healthcare in 2023. This growing healthcare expenditure is creating new opportunities for centesis device manufacturers to tap into underpenetrated markets, offering affordable and accessible solutions for minimally invasive procedures.
- Technological Advancements in Device Design: Technological advancements in the design and functionality of centesis devices are paving the way for more precise and efficient procedures. The introduction of automated and disposable centesis needles in 2022 has reduced the time required for procedures substantially. These innovations have enabled healthcare professionals to perform centesis procedures with improved accuracy, minimizing patient discomfort and reducing the risk of complications. The continuous evolution of device technology is expected to enhance the global adoption of centesis devices.
Scope of the Report
|
By Procedure Type |
Thoracentesis Paracentesis Amniocentesis Arthrocentesis Other Procedures |
|
By Product Type |
Centesis Needles Drainage Catheters Procedure Trays Other Accessories |
|
By Application |
Diagnostic Therapeutic |
|
By End-User |
Hospitals ASCs Diagnostic Centers Specialty Clinics |
|
By Region |
North America Europe Asia-Pacific Latin America Middle East & Africa |
Products
Key Target Audience
Hospitals and Diagnostic Centers
Centesis Device Manufacturers
Medical Device Distributors
Government and Regulatory Bodies (e.g., U.S. FDA, European Medical Device Regulation)
Ambulatory Surgical Centers (ASCs)
Investors and Venture Capitalist Firms
Banks and Financial Institutions
Healthcare Providers and Practitioners
Companies
Players Mentioned in the Report
Cardinal Health
BD (Becton Dickinson and Company)
Teleflex Incorporated
Medline Industries, Inc.
Merit Medical Systems
Smiths Medical
Argon Medical Devices, Inc.
Cook Medical
Stryker Corporation
Rocket Medical plc
Biometrix
Halyard Health
Medtronic
SOMATEX Medical Technologies GmbH
PFM Medical
Table of Contents
1. Global Centesis Device Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2. Global Centesis Device Market Size (In USD Mn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
3. Global Centesis Device Market Analysis
3.1. Growth Drivers
3.1.1. Increasing Prevalence of Chronic Diseases
3.1.2. Rising Demand for Minimally Invasive Procedures
3.1.3. Growing Geriatric Population
3.1.4. Advancements in Imaging Technologies
3.2. Market Challenges
3.2.1. High Costs Associated with Devices and Procedures
3.2.2. Stringent Regulatory Requirements
3.2.3. Risk of Infection and Complications
3.3. Opportunities
3.3.1. Emerging Markets for Centesis Procedures
3.3.2. Technological Advancements in Device Design
3.3.3. Expansion of Healthcare Infrastructure in Developing Regions
3.4. Trends
3.4.1. Integration of Ultrasound Guidance with Centesis Devices
3.4.2. Increasing Focus on Patient-Centric Solutions
3.4.3. Product Innovation and New Launches in Centesis Needles
3.5. Government Regulations
3.5.1. U.S. FDA Approval Process for Centesis Devices
3.5.2. European Union Medical Device Regulation (MDR)
3.5.3. Compliance with ISO Standards for Medical Devices
3.6. SWOT Analysis
3.7. Stake Ecosystem (Hospitals, Clinics, Diagnostic Centers)
3.8. Porters Five Forces
3.9. Competition Ecosystem
4. Global Centesis Device Market Segmentation
4.1. By Procedure Type (In Value %)
4.1.1. Thoracentesis
4.1.2. Paracentesis
4.1.3. Amniocentesis
4.1.4. Arthrocentesis
4.1.5. Other Procedures
4.2. By Product Type (In Value %)
4.2.1. Centesis Needles
4.2.2. Drainage Catheters
4.2.3. Procedure Trays
4.2.4. Other Accessories
4.3. By Application (In Value %)
4.3.1. Diagnostic
4.3.2. Therapeutic
4.4. By End-User (In Value %)
4.4.1. Hospitals
4.4.2. Ambulatory Surgical Centers (ASCs)
4.4.3. Diagnostic Centers
4.4.4. Specialty Clinics
4.5. By Region (In Value %)
4.5.1. North America
4.5.2. Europe
4.5.3. Asia-Pacific
4.5.4. Latin America
4.5.5. Middle East & Africa
5. Global Centesis Device Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. Cardinal Health
5.1.2. BD (Becton, Dickinson and Company)
5.1.3. Teleflex Incorporated
5.1.4. Medline Industries, Inc.
5.1.5. Merit Medical Systems
5.1.6. Smiths Medical
5.1.7. Argon Medical Devices, Inc.
5.1.8. Cook Medical
5.1.9. Stryker Corporation
5.1.10. Rocket Medical plc
5.1.11. Biometrix
5.1.12. Halyard Health
5.1.13. Medtronic
5.1.14. SOMATEX Medical Technologies GmbH
5.1.15. PFM Medical
5.2. Cross Comparison Parameters (Product Portfolio, Technology Integration, Market Presence, Regulatory Approvals, Innovation Pipeline, Manufacturing Capabilities, Distribution Networks, Strategic Collaborations)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers and Acquisitions
5.6. Investment Analysis
5.7. Venture Capital Funding
5.8. Government Grants
5.9. Private Equity Investments
6. Global Centesis Device Market Regulatory Framework
6.1. Medical Device Standards (ISO 13485)
6.2. FDA Guidelines for Device Manufacturing
6.3. CE Marking and European Medical Device Regulations
6.4. Risk Management and Device Classification
7. Global Centesis Device Future Market Size (In USD Mn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. Global Centesis Device Future Market Segmentation
8.1. By Procedure Type (In Value %)
8.2. By Product Type (In Value %)
8.3. By Application (In Value %)
8.4. By End-User (In Value %)
8.5. By Region (In Value %)
9. Global Centesis Device Market Analysts Recommendations
9.1. TAM/SAM/SOM Analysis
9.2. Customer Cohort Analysis
9.3. Marketing Initiatives
9.4. White Space Opportunity Analysis
Disclaimer
Contact Us
Research Methodology
Step 1: Identification of Key Variables
In the initial phase, an ecosystem map was constructed encompassing all major stakeholders within the Global Centesis Device Market. This involved extensive desk research and the utilization of proprietary databases to gather comprehensive industry-level information. The primary objective was to identify and define critical variables influencing market dynamics.
Step 2: Market Analysis and Construction
During this phase, historical data was compiled and analyzed, assessing market penetration, the ratio of healthcare providers using centesis devices, and revenue generation. Market estimates were further corroborated by analyzing service quality statistics to ensure the reliability and accuracy of the data.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses were validated through consultations with industry experts representing hospitals, diagnostic centers, and centesis device manufacturers. These interviews provided operational and financial insights, helping to refine market segmentation and revenue estimates.
Step 4: Research Synthesis and Final Output
The final phase involved engaging with centesis device manufacturers to gain insights into product segments, sales performance, and customer preferences. This helped verify data derived from the bottom-up approach, ensuring a comprehensive analysis of the Global Centesis Device Market.
Frequently Asked Questions
01. How big is the Global Centesis Device Market?
The Global Centesis Device Market is valued at USD 430 million, driven by the increasing demand for minimally invasive procedures and advancements in medical imaging technologies.
02. What are the challenges in the Global Centesis Device Market?
Challenges in the Global Centesis Device Market include high costs associated with acquiring advanced centesis devices, regulatory hurdles that slow down product approvals, and the need for skilled professionals to operate these devices.
03. Who are the major players in the Global Centesis Device Market?
Key players in the Global Centesis Device Market include Cardinal Health, BD (Becton Dickinson and Company), Teleflex Incorporated, Medline Industries, Inc., and Merit Medical Systems, which dominate the market due to their robust product portfolios and technological innovations.
04. What are the growth drivers of the Global Centesis Device Market?
The Global Centesis Device Market is driven by the rising prevalence of chronic diseases, advancements in imaging technology, and the increasing demand for minimally invasive diagnostic and therapeutic procedures.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















