
Global Heparin Market Outlook 2030
Region:Global
Author(s):Shivani Mehra
Product Code:KROD5677

Region:Global
Author(s):Shivani Mehra
Product Code:KROD5677
November 2024
94
By Product Type: The global heparin market is segmented by product type into unfractionated heparin (UFH), low molecular weight heparin (LMWH), and fondaparinux. Low molecular weight heparin has emerged as the dominant segment due to its enhanced efficacy, lower incidence of side effects, and convenience of administration. Healthcare providers increasingly favor LMWH over UFH because of its predictable pharmacokinetics, which allow for fixed dosing without routine monitoring. The growing preference for LMWH is underscored by clinical evidence supporting its effectiveness in various thromboembolic conditions.
By Region: The global heparin market is segmented by region, encompassing North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa. North America dominates the market, driven by its robust healthcare infrastructure, high healthcare expenditure, and the presence of key market players. The United States, in particular, leads due to a well-established reimbursement framework for anticoagulant therapies and a significant number of surgical procedures requiring heparin. The increasing awareness of thromboembolic disorders also contributes to the market growth in this region.
The global heparin market is characterized by the presence of a few major players, including Sanofi S.A., B. Braun Melsungen AG, Pfizer Inc., Novartis AG, and GlaxoSmithKline plc. These companies hold significant market share due to their established product portfolios, extensive distribution networks, and ongoing investments in research and development. This consolidation indicates the strong influence of these key players in shaping market trends and driving innovation.
|
Company |
Establishment Year |
Headquarters |
Market Focus |
R&D Investment |
Product Portfolio |
Global Reach |
|
Sanofi S.A. |
2004 |
Paris, France |
Pharmaceuticals |
|||
|
B. Braun Melsungen AG |
1839 |
Melsungen, Germany |
Medical Devices and Pharmaceuticals |
|||
|
Pfizer Inc. |
1849 |
New York, USA |
Pharmaceuticals |
|||
|
Novartis AG |
1996 |
Basel, Switzerland |
Pharmaceuticals |
|||
|
GlaxoSmithKline plc |
2000 |
Brentford, UK |
Pharmaceuticals |
Over the next five years, the global heparin market is expected to experience robust growth driven by continuous advancements in therapeutic formulations and an increasing emphasis on effective anticoagulation management. Key factors such as the rising incidence of thromboembolic disorders, expanding surgical procedures, and a growing geriatric population will contribute to the market's positive trajectory. Additionally, ongoing research initiatives aimed at developing novel heparin products are likely to enhance treatment outcomes and expand patient access to these critical therapies.
|
By Product Type |
|
|||||
|
By Route of Administration |
Intravenous Subcutaneous |
|||||
|
By Application |
Cardiovascular Diseases Orthopedic Disorders Surgery and Critical Care |
|||||
|
By End-User |
Hospitals Clinics Homecare Settings |
|||||
|
By Region |
North America Europe Asia-Pacific Latin America Middle East & Africa |
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
3.1. Growth Drivers
3.1.1. Increasing Prevalence of Thromboembolic Disorders
3.1.2. Rise in Surgical Procedures
3.1.3. Growing Demand for Anticoagulant Therapies
3.1.4. Expansion of the Elderly Population
3.2. Market Challenges
3.2.1. Risk of Heparin-Induced Thrombocytopenia
3.2.2. Competition from Novel Anticoagulants
3.2.3. Stringent Regulatory Requirements
3.3. Opportunities
3.3.1. Research and Development in Antithrombotic Therapies
3.3.2. Expansion into Emerging Markets
3.3.3. Technological Advancements in Drug Delivery Systems
3.4. Trends
3.4.1. Adoption of Low Molecular Weight Heparin (LMWH)
3.4.2. Increasing Use of Generic Heparin Products
3.4.3. Integration of Digital Health Technologies
3.5. Government Regulation
3.5.1. Drug Approval Processes
3.5.2. Market Surveillance and Safety Regulations
3.5.3. Reimbursement Policies
3.6. SWOT Analysis
3.7. Stake Ecosystem
3.8. Porters Five Forces
3.9. Competition Ecosystem
4.1. By Product Type (In Value %)
4.1.1. Unfractionated Heparin (UFH)
4.1.2. Low Molecular Weight Heparin (LMWH)
4.1.3. Fondaparinux
4.2. By Route of Administration (In Value %)
4.2.1. Intravenous
4.2.2. Subcutaneous
4.3. By Application (In Value %)
4.3.1. Cardiovascular Diseases
4.3.2. Orthopedic Disorders
4.3.3. Surgery and Critical Care
4.4. By End-User (In Value %)
4.4.1. Hospitals
4.4.2. Clinics
4.4.3. Homecare Settings
4.5. By Region (In Value %)
4.5.1. North America
4.5.2. Europe
4.5.3. Asia-Pacific
4.5.4. Latin America
4.5.5. Middle East & Africa
5.1. Detailed Profiles of Major Companies
5.1.1. Sanofi S.A.
5.1.2. B. Braun Melsungen AG
5.1.3. Pfizer Inc.
5.1.4. Novartis AG
5.1.5. GlaxoSmithKline plc
5.1.6. Leo Pharma A/S
5.1.7. Hikma Pharmaceuticals PLC
5.1.8. Fresenius Kabi AG
5.1.9. Teva Pharmaceutical Industries Ltd.
5.1.10. Baxter International Inc.
5.1.11. Boehringer Ingelheim GmbH
5.1.12. Merck & Co., Inc.
5.1.13. Amgen Inc.
5.1.14. HLS Therapeutics Inc.
5.1.15. Medtronic PLC
5.2. Cross Comparison Parameters (Number of Employees, Headquarters, Inception Year, Revenue)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers and Acquisitions
5.6. Investment Analysis
5.7. Venture Capital Funding
5.8. Government Grants
5.9. Private Equity Investments
6.1. Environmental Standards
6.2. Compliance Requirements
6.3. Certification Processes
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8.1. By Product Type (In Value %)
8.2. By Route of Administration (In Value %)
8.3. By Application (In Value %)
8.4. By End-User (In Value %)
8.5. By Region (In Value %)
9.1. TAM/SAM/SOM Analysis
9.2. Customer Cohort Analysis
9.3. Marketing Initiatives
9.4. White Space Opportunity Analysis
The initial phase involves constructing an ecosystem map encompassing all major stakeholders within the global heparin market. This step is underpinned by extensive desk research, utilizing a combination of secondary and proprietary databases to gather comprehensive industry-level information. The primary objective is to identify and define the critical variables that influence market dynamics.
In this phase, we will compile and analyze historical data pertaining to the global heparin market. This includes assessing market penetration, the ratio of marketplaces to service providers, and the resultant revenue generation. Furthermore, an evaluation of service quality statistics will be conducted to ensure the reliability and accuracy of the revenue estimates.
Market hypotheses will be developed and subsequently validated through computer-assisted telephone interviews (CATI) with industry experts representing a diverse array of companies. These consultations will provide valuable operational and financial insights directly from industry practitioners, which will be instrumental in refining and corroborating the market data.
The final phase involves direct engagement with multiple heparin manufacturers to acquire detailed insights into product segments, sales performance, consumer preferences, and other pertinent factors. This interaction will serve to verify and complement the statistics derived from the bottom-up approach, thereby ensuring a comprehensive, accurate, and validated analysis of the global heparin market.
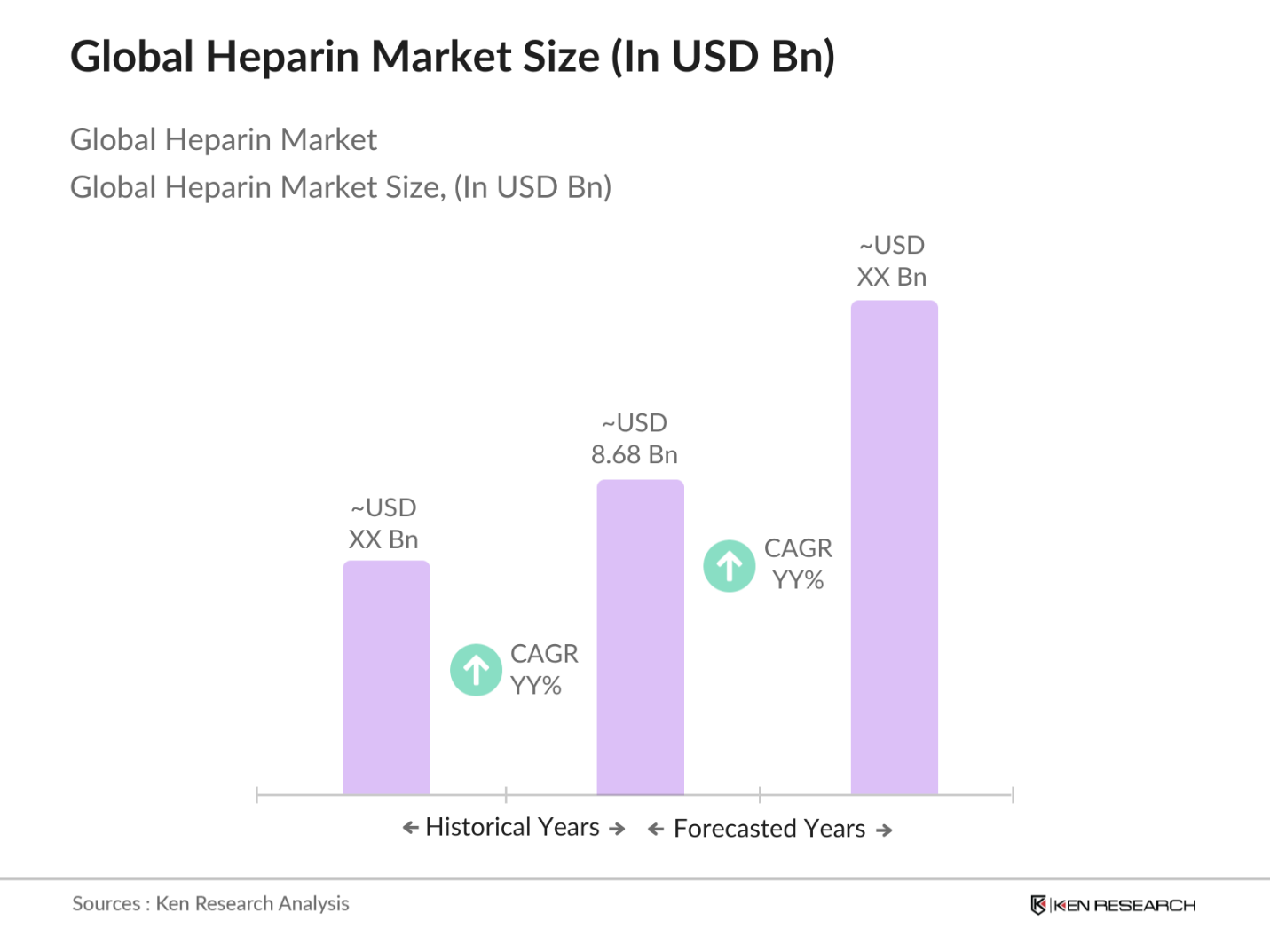
The global heparin market is valued at USD 8.68 billion, driven by a growing demand for anticoagulants due to the increasing prevalence of thromboembolic disorders and surgical procedures.
Key drivers include the rising incidence of cardiovascular diseases, an expanding elderly population, and increased adoption of anticoagulant therapies in healthcare settings, contributing to significant market growth.
Key players include Sanofi S.A., B. Braun Melsungen AG, Pfizer Inc., Novartis AG, and GlaxoSmithKline plc, which dominate the market due to their established product portfolios and robust distribution networks.
Challenges include the risk of heparin-induced thrombocytopenia, competition from newer anticoagulants, and stringent regulatory requirements that may impact product availability and pricing.

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.