
Indian Clinical Trials Market Outlook to 2030
Region:Global
Author(s):Shivani Mehra
Product Code:KROD3938
December 2024
86
About the Report
Indian Clinical Trials Market Overview
- The Indian Clinical Trials market is valued at USD 2.27 billion, based on a five-year historical analysis. This growth is driven by an increase in disease prevalence across multiple therapeutic areas, including oncology, cardiovascular, and neurology, which is pushing demand for new treatment evaluations and drug approvals. Additionally, advancements in biotechnology and government initiatives to streamline the clinical trial approval process further support market expansion.
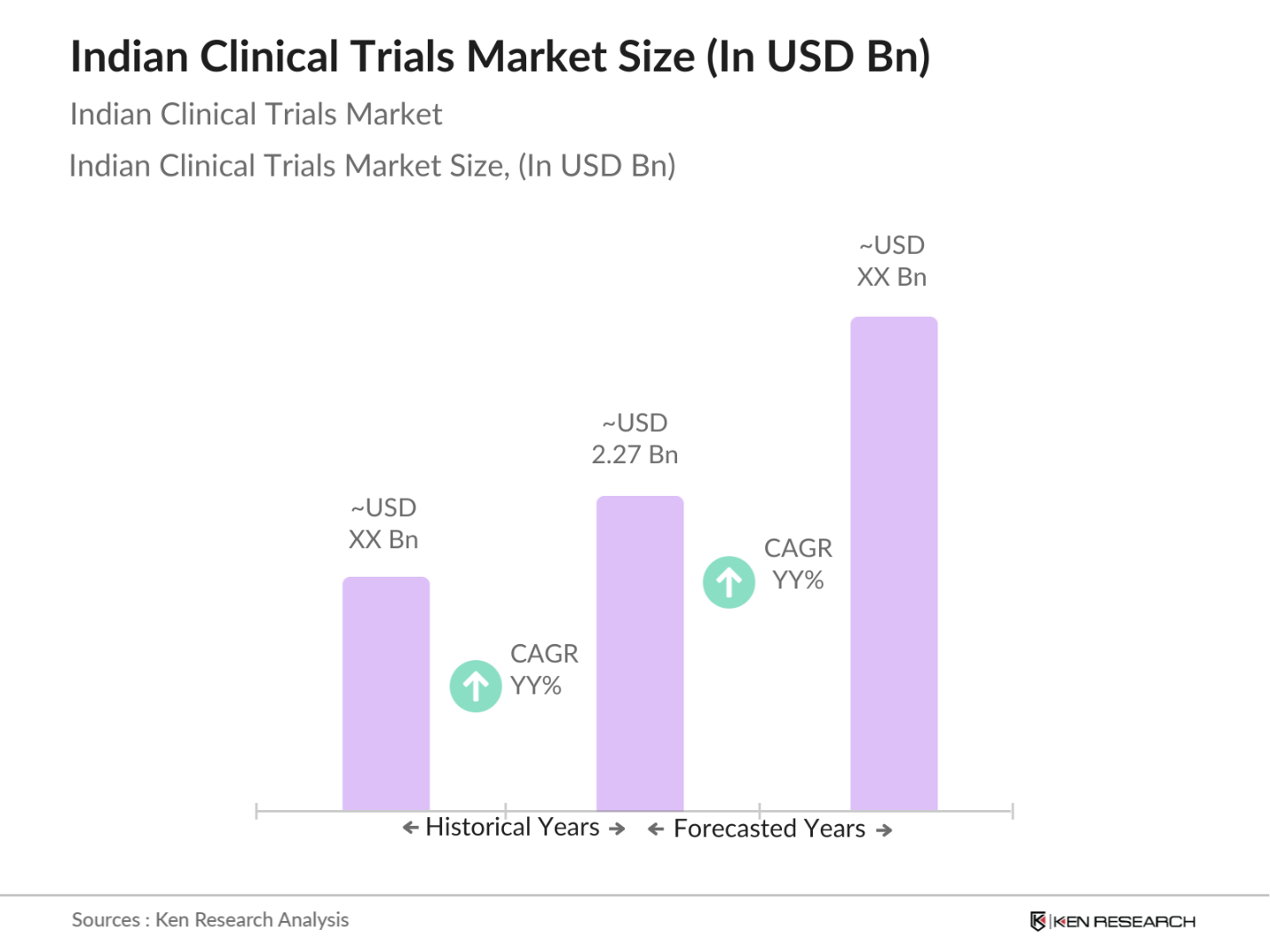
- The market is dominated by major metropolitan areas like Delhi, Mumbai, and Bangalore, along with regions in southern India due to their well-established healthcare infrastructure and availability of qualified investigators and healthcare professionals. These cities also have a higher concentration of top pharmaceutical and biotechnology companies, as well as established hospitals and research centers, making them prime locations for clinical trial operations.
- The New Drugs and Clinical Trials Rules, introduced, include provisions for accelerated drug approval, particularly for treatments targeting life-threatening conditions. This regulation enables faster approval of drugs showing significant therapeutic potential, especially for critical diseases such as cancer and rare disorders. By prioritizing drugs with a high potential to save lives, this initiative aims to address urgent healthcare needs while boosting clinical research activity.
Indian Clinical Trials Market Segmentation
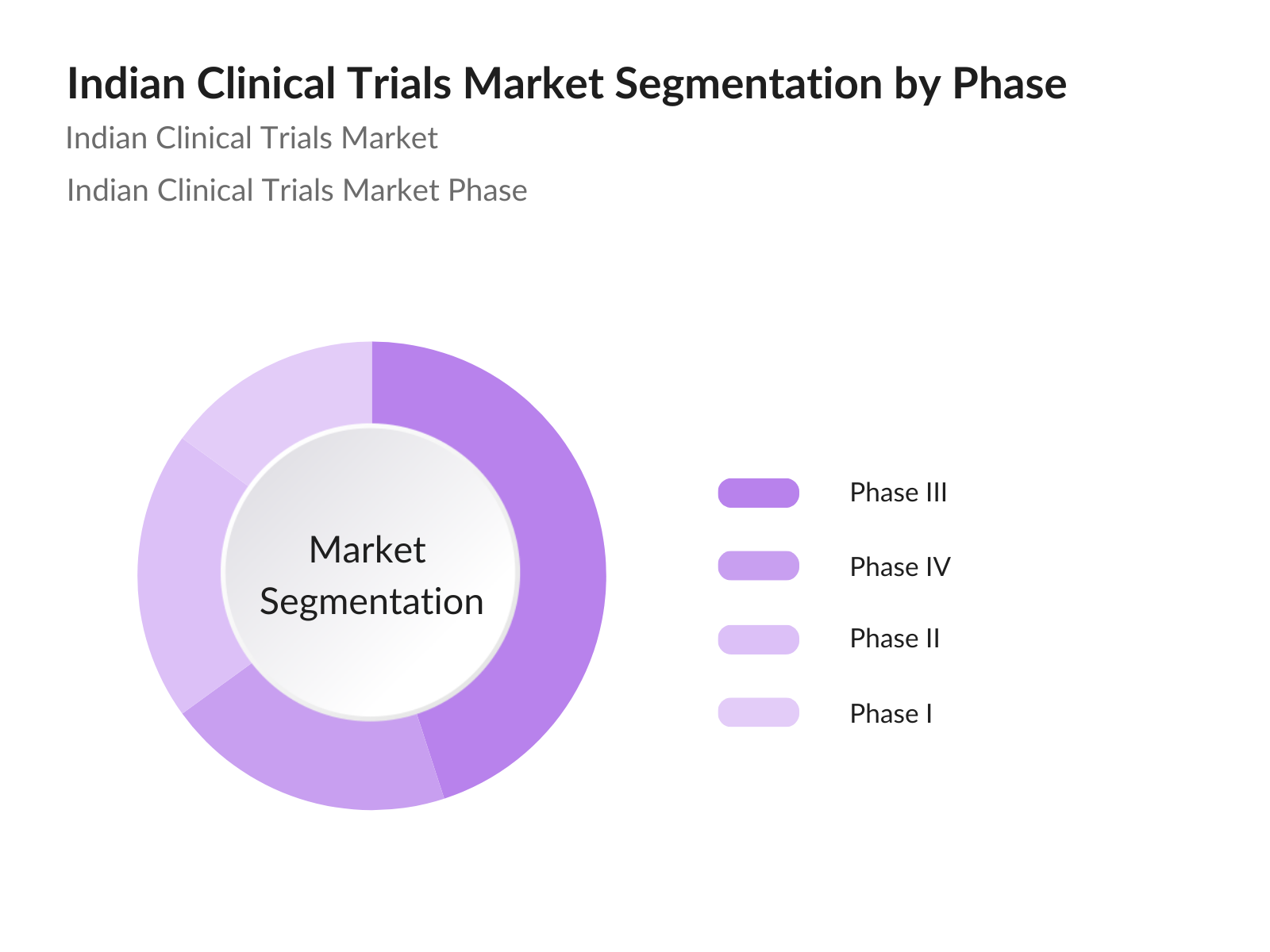
By Phase: The Indian Clinical Trials market is segmented by phase into Phase I, Phase II, Phase III, and Phase IV trials. Phase III trials currently hold the largest market share, as they involve large-scale testing to assess the efficacy of treatments before submission for regulatory approval. These trials often attract significant funding from pharmaceutical companies due to their critical role in the drug development process. The scale and complexity of Phase III trials also contribute to their dominance, as they offer comprehensive data crucial for final approval.
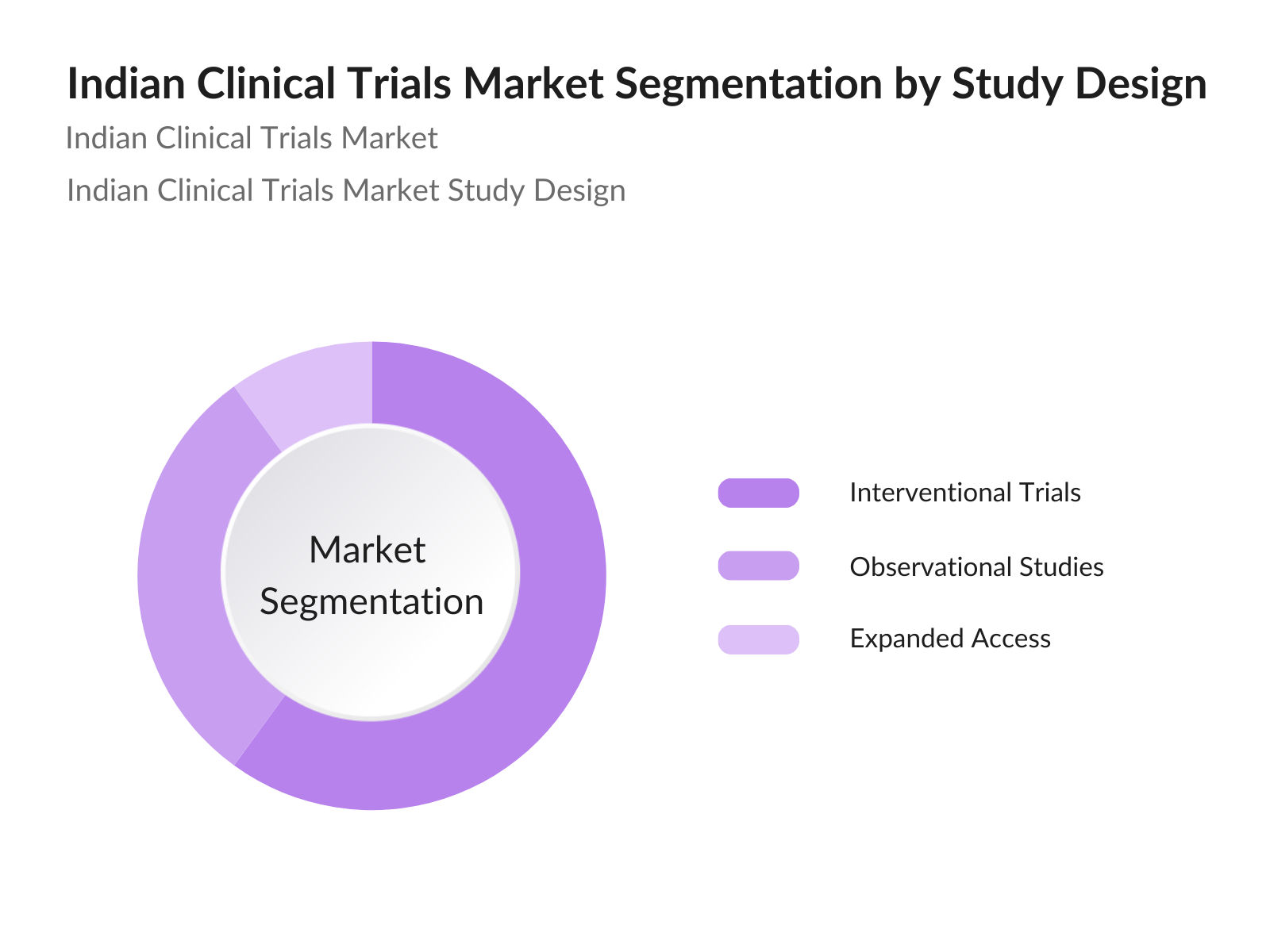
By Study Design: The Indian Clinical Trials market is segmented by study design into Interventional Trials, Observational Studies, and Expanded Access. Interventional trials lead the market, primarily due to their proactive approach in testing specific medical interventions, which is essential for obtaining FDA and CDSCO approvals. Pharmaceutical companies favor interventional trials as they provide clear data on the safety and efficacy of a treatment, a crucial requirement in gaining regulatory acceptance and market entry.
Indian Clinical Trials Market Competitive Landscape
The Indian Clinical Trials market is dominated by a few major players that drive significant market influence. These companies include local Contract Research Organizations (CROs) and international players, supported by collaborations with domestic pharmaceutical firms. This concentration highlights the prominent role of these companies in shaping the clinical trials landscape.
Indian Clinical Trials Market Analysis
Market Growth Drivers
- Increase in Disease Prevalence: India faces a significant burden of non-communicable diseases. In 2022, the country reported approximately 1.39 million new cancer cases, with breast, lung, and cervical cancers being the most prevalent. Cardiovascular diseases caused around 3 million deaths annually, underscoring the critical need for healthcare interventions. Neurological disorders, including stroke and epilepsy, affected millions, contributing significantly to the overall disease burden. Additionally, infectious diseases such as tuberculosis had over 2.6 million reported cases in 2022. This high prevalence of both communicable and non-communicable diseases emphasizes the importance of extensive clinical trials for developing effective treatments.
- Government Initiatives & Regulations: The Indian government has introduced measures to streamline the clinical trial process, focusing on reducing approval timelines and accelerating drug approval for critical treatments. Recent regulatory reforms, including provisions under the New Drugs and Clinical Trials Rules, have been established to create a more efficient and supportive environment for clinical research. These initiatives are designed to attract greater investment in clinical trials and foster innovation in developing treatments for serious health conditions.
- Rising Demand for Personalized Medicine: The global shift towards personalized medicine is evident in India, with a growing emphasis on genomics and targeted therapies. In 2023, over 200 clinical trials focused on personalized treatments were registered in India, covering areas like oncology and rare genetic disorders. The establishment of genomic databases and biobanks has further facilitated research in this domain, highlighting the country's commitment to advancing personalized healthcare.
Market Challenges:
- Regulatory Compliance Issues: Navigating India's regulatory landscape poses challenges for clinical trials. The CDSCO enforces stringent guidelines, and in 2023, numerous trial applications faced delays due to non-compliance. Ethical approvals, managed by Institutional Ethics Committees, add another layer of scrutiny, with a significant number of trials experiencing setbacks related to ethical considerations. These regulatory hurdles can impede the timely initiation and progression of clinical studies, impacting the overall efficiency of the clinical research process.
- Shortage of Skilled Workforce: India faces a deficit of trained professionals in clinical research. In 2023, the country had approximately 5,000 certified clinical research professionals, a number insufficient to meet the demands of over 3,000 ongoing trials. This shortage extends to qualified investigators and site staff, leading to challenges in trial management and data integrity. The limited availability of skilled personnel can compromise the quality and efficiency of clinical studies.
Indian Clinical Trials Market Future Outlook
Over the next five years, the Indian Clinical Trials market is expected to see robust growth, driven by increased government support, a surge in the outsourcing of clinical trials to India, and an emphasis on patient-centric trial designs. Additionally, advancements in digital health and telemedicine are expected to ease patient recruitment and improve data management processes. The growing interest in personalized medicine and genomics will also contribute to a favorable market trajectory.
Market Opportunities:
- Technological Advancements: The integration of technology in clinical trials presents significant opportunities. Artificial Intelligence (AI) is increasingly used for patient recruitment, with platforms analyzing electronic health records to identify suitable candidates, substantially reducing recruitment time. Advanced data management systems have been widely adopted by research organizations in India, enhancing data accuracy and compliance. These technological innovations are streamlining trial processes and significantly improving overall efficiency in clinical research.
- Global Collaborations: Indian research institutions have increasingly engaged in global collaborations. In 2023, over 300 multinational clinical studies involved Indian sites, covering therapeutic areas like oncology and infectious diseases. Partnerships with global pharmaceutical companies have led to joint ventures and knowledge exchange, enhancing the quality and scope of clinical research conducted in the country.
Scope of the Report
|
By Phase |
Phase I Phase II Phase III Phase IV |
|
By Study Design |
Interventional Trials Observational Studies Expanded Access |
|
By Indication |
Oncology Cardiovascular Diseases Neurological Disorders Infectious Diseases Endocrine Disorders |
|
By Sponsor Type |
Pharmaceutical Companies Biotech Companies Academic Institutions Government Agencies |
|
By Region |
North-East Midwest West Coast Southern States |
Products
Key Target Audience
Pharmaceutical Companies
Biotechnology Firms
Contract Research Organizations (CROs)
Clinical Trial Site Operators
Medical Device Manufacturers
Government and Regulatory Bodies (e.g., CDSCO, ICMR)
Investments and Venture Capitalist Firms
Hospitals and Healthcare Networks
Companies
Player Mention in the Report
IQVIA
Syneos Health
PAREXEL International
Medpace, Inc.
Veeda Clinical Research
Cliantha Research Limited
Neeman Medical International
Siro Clinpharm Pvt. Ltd.
Jubilant Biosys Ltd.
Lotus Labs Pvt Ltd.
Accutest Research Laboratories
Dr. Reddy's Laboratories Ltd.
Alkem Laboratories Ltd.
Fortis Clinical Research Ltd.
Sun Pharmaceutical Industries Ltd.
Table of Contents
Research Methodology
Step 1: Identification of Key Variables
The first stage involves mapping the entire ecosystem of stakeholders within the Indian Clinical Trials Market. Extensive desk research is conducted to gather relevant data from secondary sources, including government publications and proprietary databases. This step helps in identifying crucial variables like trial phases, patient demographics, and regulatory impacts.
Step 2: Market Analysis and Construction
In this phase, historical data on the Indian Clinical Trials Market is collated and analyzed. This includes assessing trial volume, regional participation, and service provider networks. Additionally, revenue estimates are derived by evaluating cost structures and pricing trends across trial phases.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are developed based on preliminary findings and validated through telephonic interviews with industry experts, including clinical researchers and pharmaceutical executives. These consultations provide firsthand insights into operational and financial parameters, ensuring data accuracy.
Step 4: Research Synthesis and Final Output
The final step involves synthesizing findings with direct input from CROs and pharmaceutical companies. This phase ensures that the insights cover a comprehensive view of market trends, consumer behavior, and industry-specific challenges in the Indian Clinical Trials Market.
Frequently Asked Questions
01. How big is the Indian Clinical Trials Market?
The Indian Clinical Trials Market, valued at USD 2.27 billion, is driven by increasing demand for drug development and growing pharmaceutical R&D investments.
02. What are the challenges in the Indian Clinical Trials Market?
Challenges include regulatory hurdles, high operational costs, and a shortage of skilled investigators. These factors can limit the speed and scale of clinical trials in India.
03. Who are the major players in the Indian Clinical Trials Market?
Key players include IQVIA, Syneos Health, PAREXEL International, and Medpace. Their dominance stems from strong networks, extensive expertise, and high funding levels.
04. What are the growth drivers of the Indian Clinical Trials Market?
Growth drivers include increased prevalence of chronic diseases, advancements in biotechnology, and supportive government initiatives aimed at accelerating clinical research.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















