
KSA Point-of-Care Molecular Diagnostics Market
Region:Middle East
Author(s):Sanjna Verma
Product Code:KROD789
July 2024
90
About the Report
KSA Point-of-Care Molecular Diagnostics Market Overview
- KSA Point-of-Care (POC) Molecular Diagnostics Market has experienced notable growth, this is reflected in the global Point-of-Care Diagnostics market valued at USD 2 billion in 2023. This growth is primarily driven by the increasing prevalence of infectious diseases, rising demand for rapid diagnostic techniques, and advancements in molecular diagnostic technologies.
- Key players in the KSA POC Molecular Diagnostics Market include Abbott Laboratories, Roche Diagnostics, Cepheid, BioMérieux and Thermo Fisher Scientific. These companies are leading the market with their innovative products and extensive distribution networks.
- In 2023, Roche Diagnostics launched its cobas® Liat® system in Saudi Arabia, a highly efficient and rapid point-of-care molecular testing device. The cobas® Liat® system fully automates the PCR process, allowing the rapid and accurate analysis of single samples.
KSA Point-of-Care Molecular Diagnostics Current Market Analysis
- Continuous innovations in molecular diagnostic technologies, such as next-generation sequencing (NGS) and CRISPR-based diagnostics, are significantly enhancing the accuracy, speed, and range of diagnostics available at the point of care.
- The impact of growth drivers is substantial, leading to quicker diagnosis, timely treatment, and better patient outcomes. Moreover, the integration of advanced diagnostics at the point of care reduces the burden on centralized laboratories and enhances the overall efficiency of healthcare services.
- The Central Region, including Riyadh, is dominating the market in particular. This dominance is due to the concentration of advanced healthcare facilities, higher investment in healthcare infrastructure, and the presence of major diagnostic companies.
KSA Point-of-Care Molecular Diagnostics Market Segmentation
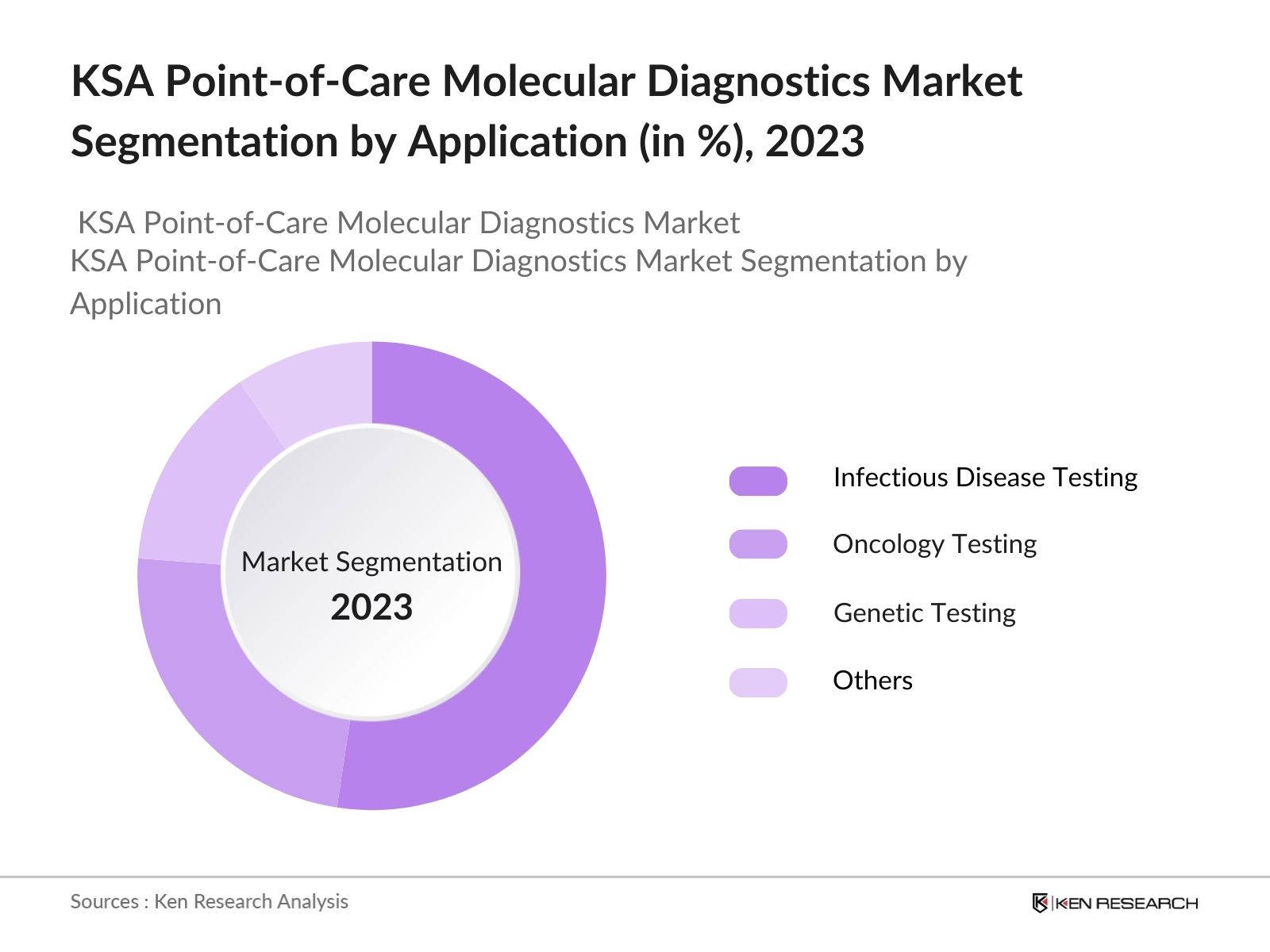
By Application: By application, the KSA point-of-care molecular diagnostics market is segmented into infectious disease testing, oncology testing, genetic testing & others. In 2023, Infectious disease testing dominates due to the high prevalence of infectious diseases and the critical need for rapid diagnosis to prevent outbreaks.
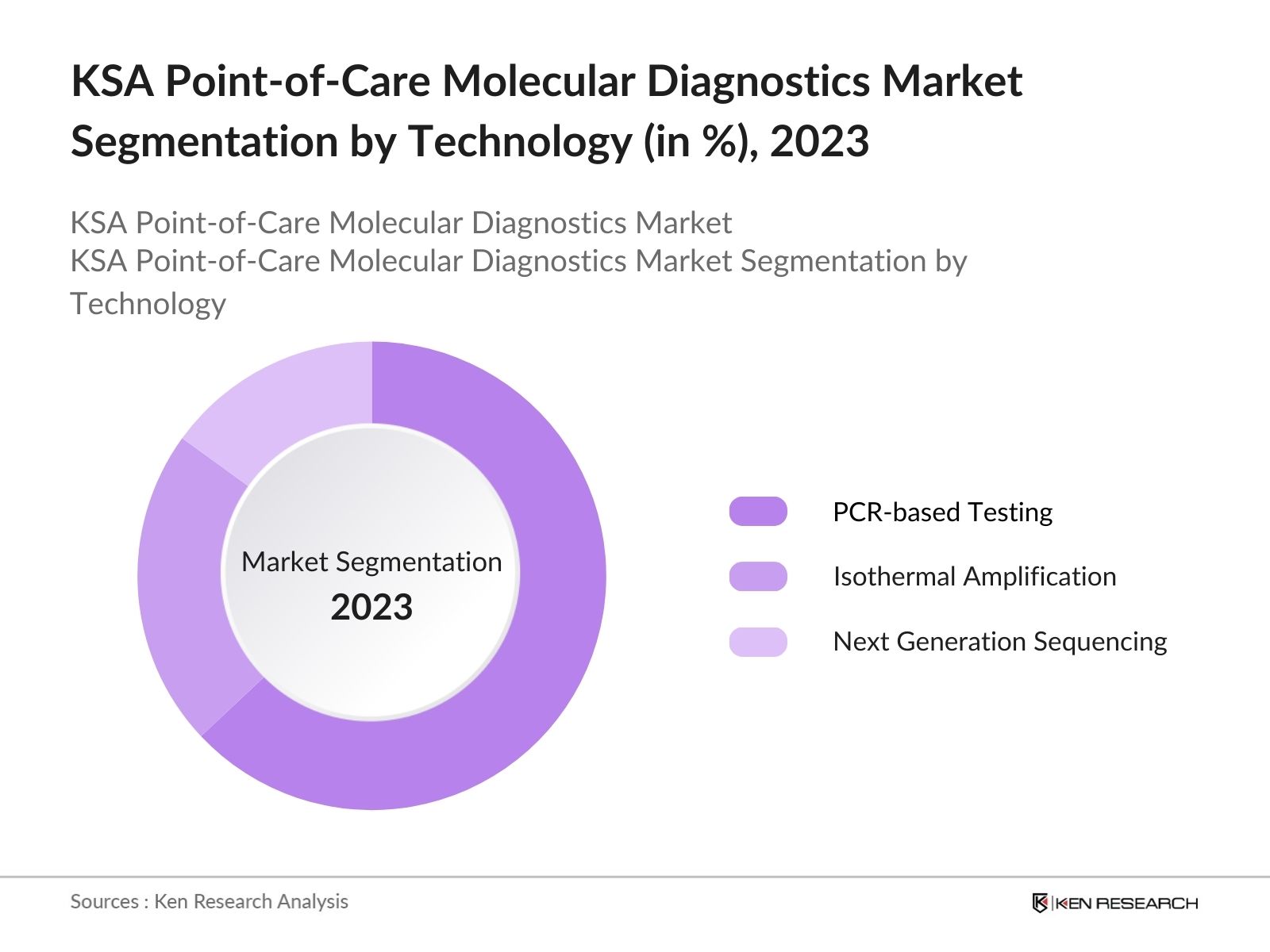
By Technology: By technology, the market is segmented into PCR-based testing, isothermal amplification and next generation sequencing. In 2023, PCR-based testing dominates because of its high accuracy, reliability, and wide application in detecting various pathogens and genetic anomalies.
By End-Users: The market is segmented into hospitals, diagnostic centres, research laboratories and others. In 2023, Hospitals is dominate as they are the primary centers for diagnosing and treating serious health conditions, ensuring a steady demand for advanced diagnostics.
KSA Point-of-Care Molecular Diagnostics Market Competitive Landscape
- Roche Diagnostics: In March 2024, Roche acquired the point-of-care (POC) testing technology from LumiraDX, a London-based company, for $295 million. This acquisition is expected to close in mid-2024 and will enable Roche to transform testing at the point of care.
- Thermo Fisher Scientific: In February 2023, Thermo Fisher Scientific announced the launch of its Applied Biosystems TaqPath PCR kits for infectious diseases like multi-drug-resistant tuberculosis (MTB MDR), M. tuberculosis complex (MTB), hepatitis B virus (HBV), hepatitis C virus (HCV), human immunodeficiency virus (HIV), and for genetic analysis (HLA B27).
- Cepheid (Danaher Corp): In 2022, Cepheid launched the Xpert® MTB/RIF Ultra test in Saudi Arabia, significantly enhancing the diagnosis and management of tuberculosis (TB). This test offers improved sensitivity and faster results, enabling healthcare providers to quickly and accurately diagnose TB and detect rifampicin resistance.
KSA Point-of-Care Molecular Diagnostics Industry Analysis
KSA Point-of-Care Molecular Diagnostics Growth Drivers
- Saudi Vision 2030 Initiative: In 2023, the government allocated USD 50.4 billion towards healthcare infrastructure development, enhancing the adoption of advanced diagnostic technologies, including POC molecular diagnostics. The initiative aims to increase private sector contribution, thereby promoting investments in healthcare innovations and technologies.
- Healthcare Infrastructure Development: The Saudi government has been aggressively investing in the expansion and modernization of healthcare facilities. In 2023, the number of hospitals was 497, with an emphasis on incorporating advanced diagnostic equipment. This expansion is supported by the government's budget allocation of USD 50.4 billion for healthcare and social development in 2023. Such investments directly enhance the capacity and capabilities of healthcare providers to implement POC molecular diagnostics.
- Increased Prevalence of Chronic and Infectious Diseases: The rise in chronic and infectious diseases within Saudi Arabia is a significant driver for the POC molecular diagnostics market. According to the World Health Organization (WHO), non-communicable diseases like cancer kill 41 million people, out of which 80% of premature deaths come from low- & middle-income countries like Saudi Arabia.
KSA Point-of-Care Molecular Diagnostics Market Challenges
- Stringent Approval Processes: The Saudi Food and Drug Authority (SFDA) has stringent regulatory requirements for the approval of new diagnostic tests. The approval process for POC molecular diagnostics can take up to 18 months, delaying market entry and innovation. This regulatory bottleneck affects the speed at which new and advanced diagnostic technologies can be adopted, potentially hindering market growth.
- Cost Constraints: The high cost of POC molecular diagnostic devices and tests can be a significant barrier, particularly for smaller healthcare facilities and clinics. The average cost of a PCR-based POC diagnostic test ranges from USD 50 to USD 100, which may not be affordable for all segments of the population.
- Regulatory and Compliance Issues: Regulatory and compliance issues also pose challenges to the market. The stringent regulatory framework for the approval of diagnostic tools can delay the introduction of new technologies. These regulatory hurdles can slow down the market's growth by delaying the availability of innovative diagnostic solutions.
KSA Point-of-Care Molecular Diagnostics Recent Developments
- Siemens Healthineers’ Launches Atellica IM Analyzer: In 2023, Siemens Healthineers expanded its molecular point-of-care testing capabilities in Saudi Arabia by introducing the Atellica IM Analyzer. This analyzer allows for rapid and accurate testing of various infectious diseases, including COVID-19 and hepatitis. It integrates multiple diagnostic technologies, providing comprehensive diagnostic capabilities in a single platform.
- BD Diagnostics Launch of BD Max System: In 2023, BD launched the BD MAXâ„¢ system in Saudi Arabia, an automated molecular diagnostic platform designed for a variety of infectious disease assays. The BD MAXâ„¢ system offers flexibility and high throughput, meeting the needs of modern laboratories.
- BioFire Launched by BioMerieux: In 2022, BioMérieux launched its BioFire® FilmArray® panels in Saudi Arabia, offering comprehensive syndromic testing for respiratory, gastrointestinal, and bloodstream infections. These panels provide rapid, accurate results, enabling healthcare professionals to make timely and informed treatment decisions, significantly improving patient outcomes.
KSA Point-of-Care Molecular Diagnostics Future Market Outlook
The KSA POC Molecular Diagnostics Market is expected to continue growing from 2023 to 2028. Key drivers include technological advancements, the rising prevalence of chronic and infectious diseases, and ongoing government initiatives to improve healthcare infrastructure.
Future Market Trends:
- Increasing Adoption of Personalized Medicine: The shift towards personalized medicine is driving the demand for POC molecular diagnostics that offer rapid and precise genetic profiling. This trend is supported by the global push towards precision healthcare, aiming to improve patient outcomes through customized treatment plans.
- Advancements in Genetic Testing: Technological advancements in genetic testing, such as CRISPR and next-generation sequencing (NGS), will become more accessible at the point of care. These advancements enhance the capabilities of POC molecular diagnostics, making them more integral to modern healthcare.
- Integration with Digital Health Platforms: The integration of POC molecular diagnostics with digital health platforms is enhancing data management and patient monitoring. This trend supports the efficient use of diagnostic data, improving overall healthcare delivery and patient outcomes.
Scope of the Report
|
By Application |
Infectious Disease Testing Oncology testing Genetic Testing Others |
|
By Technology |
PCR-based Testing Isothermal Amplification Next Generation Sequencing |
|
By End-Users |
Hospitals Diagnostic Centers Research Laboratories Others |
Products
Key Target Audience – Organizations and Entities Who Can Benefit by Subscribing This Report:
Manufacturers of Point-of-Care Molecular Diagnostic Devices and Assays
Health Insurance Companies
Banks & Financial Institutions
Diagnostics Companies
Physicians and Clinicians
Laboratory Professionals
Government and Regulatory Bodies (Saudi Food & Drug Authority, Saudi Health Council etc.)
Clinical Research Organizations (CROs)
Time Period Captured in the Report:
- Historical Period: 2018-2023
- Base Year: 2023
- Forecast Period: 2023-2028
Companies
Players Mentioned in the Report:Â
- Roche Diagnostics
- Abbott Laboratories
- bioMérieux
- Danaher Corporation
- Cepheid
- Thermo Fisher Scientific
- Siemens Healthineers
- Qiagen
- Bio-Rad Laboratories
- Hologic
- Grifols
- GenMark Diagnostics
- Luminex Corporation
- Quidel Corporation
- OraSure Technologies
- Meridian Bioscience
- Myriad Genetics
- Sysmex Corporation
- PerkinElmer
- BD (Becton, Dickinson and Company)
Table of Contents
1. KSA Point-of-Care Molecular Diagnostics Market Overview
1.1 KSA Point-of-Care Molecular Diagnostics Market Taxonomy
2. KSA Point-of-Care Molecular Diagnostics Market Size (in USD Bn), 2018-2023
3. KSA Point-of-Care Molecular Diagnostics Market Analysis
3.1 KSA Point-of-Care Molecular Diagnostics Market Growth Drivers
3.2 KSA Point-of-Care Molecular Diagnostics Market Challenges and Issues
3.3 KSA Point-of-Care Molecular Diagnostics Market Trends and Development
3.4 KSA Point-of-Care Molecular Diagnostics Market Government Regulation
3.5 KSA Point-of-Care Molecular Diagnostics Market SWOT Analysis
3.6 KSA Point-of-Care Molecular Diagnostics Market Stake Ecosystem
3.7 KSA Point-of-Care Molecular Diagnostics Market Competition Ecosystem
4. KSA Point-of-Care Molecular Diagnostics Market Segmentation, 2023
4.1 KSA Point-of-Care Molecular Diagnostics Market Segmentation by Application, By Value, (In %), 2023
4.2 KSA Point-of-Care Molecular Diagnostics Market Segmentation by Technology, By Value, (In %), 2023
4.3 KSA Point-of-Care Molecular Diagnostics Market Segmentation by End-User Industry, By Value, (In %), 2023
5. KSA Point-of-Care Molecular Diagnostics Market Competition Benchmarking
5.1 KSA Point-of-Care Molecular Diagnostics Market Cross-Comparison (no. of employees, company overview, business strategy, USP, recent development, operational parameters, financial parameters and advanced analytics)
6. KSA Point-of-Care Molecular Diagnostics Future Market Size (in USD Bn), 2023-2028
7. KSA Point-of-Care Molecular Diagnostics Future Market Segmentation, 2028
7.1 KSA Point-of-Care Molecular Diagnostics Market Segmentation by Application, By Value, (In %), 2028
7.2 KSA Point-of-Care Molecular Diagnostics Market Segmentation by Technology, By Value, (In %), 2028
7.3 KSA Point-of-Care Molecular Diagnostics Market Segmentation by End-User Industry, By Value, (In %), 2028
8. KSA Point-of-Care Molecular Diagnostics Market Analysts’ Recommendations
8.1 KSA Point-of-Care Molecular Diagnostics Market TAM/SAM/SOM Analysis
8.2 KSA Point-of-Care Molecular Diagnostics Market Customer Cohort Analysis
8.3 KSA Point-of-Care Molecular Diagnostics Market Marketing Initiatives
8.4 KSA Point-of-Care Molecular Diagnostics Market White Space Opportunity Analysis
9. Disclaimer
10. Contact Us
Research Methodology
Step: 1 Identifying Key Variables:
Ecosystem creation for all the major entities and referring to multiple secondary and proprietary databases to perform desk research around the market to collate industry-level information.Â
Step: 2 Market Building:
Collating statistics on the KSA point-of-care molecular diagnostics market over the years, penetration of marketplaces and service providers ratio to compute revenue generated for the KSA point-of-care molecular diagnostics market. We will also review service quality statistics to understand revenue generated which can ensure accuracy behind the data points shared.Â
Step: 3 Validating and Finalizing:
Building market hypotheses and conducting CATIs with industry experts belonging to different companies to validate statistics and seek operational and financial information from company representatives.Â
Step: 4 Research Output:
Our team will approach multiple point-of-care molecular diagnostics companies and understand the nature of product segments and sales, consumer preference and other parameters, which will support us validate statistics derived through bottom-to-top approaches from point-of-care molecular diagnostics companies.Â
Frequently Asked Questions
01 How big is the KSA Point-of-Care Molecular Diagnostics market?
The Global market size was valued at USD 2 billion in 2023, driven by the increasing prevalence of infectious diseases, rising demand for rapid diagnostic techniques, and advancements in molecular diagnostic technologies. This reflects the growth in the KSA Point-of-Care Molecular Diagnostic Market.
02 Who are the major players in the KSA Point-of-Care Molecular Diagnostics market?
The KSA Point-of-Care (POC) molecular diagnostics market major players include Roche Diagnostics, Abbott Laboratories, bioMérieux, Danaher Corporation, and Cepheid. These companies are leading the market with their innovative products and extensive distribution networks.
03 What factors drive the KSA Point-of-Care Molecular Diagnostics Market?
The KSA Point-of-Care (POC) molecular diagnostics market's key drivers include technological advancements, the rising prevalence of infectious diseases, increased healthcare expenditure, and government initiatives. The rise in chronic and infectious diseases within Saudi Arabia is a significant driver for the POC molecular diagnostics market.
04 What are the challenges in KSA Point-of-Care Molecular Diagnostics Market?
Cost constraints & stringent regulatory approvals are challenges hindering the KSA Point-of-Care (POC) molecular diagnostics market’s growth. The high cost of POC molecular diagnostic devices and tests can be a significant barrier, particularly for smaller healthcare facilities and clinics.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.