
North America Ovulation Testing Kits Market Outlook to 2030
Region:North America
Author(s):Vijay Kumar
Product Code:KROD6383
November 2024
83
About the Report
North America Ovulation Testing Kits Market Overview
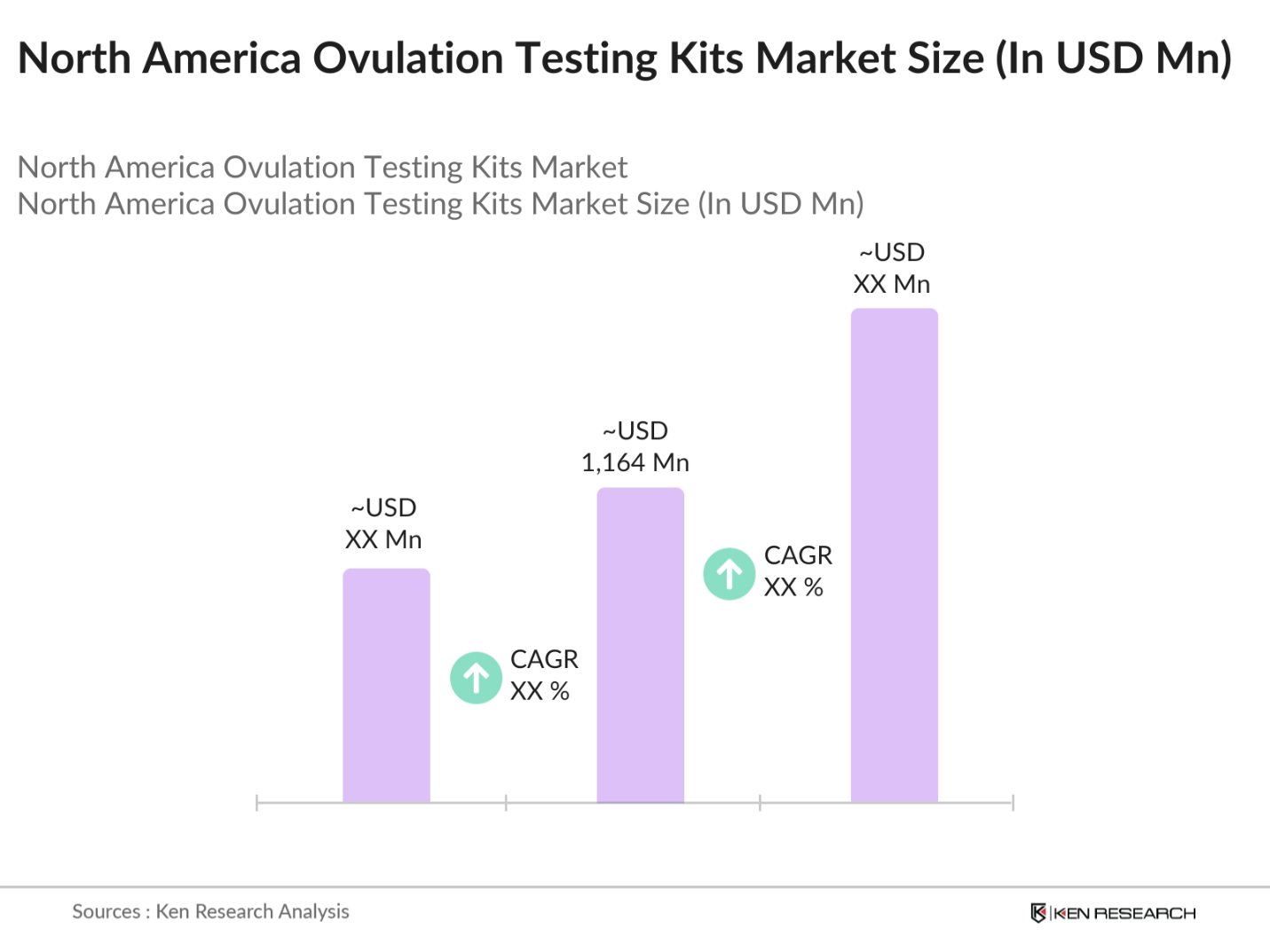
- The North America Ovulation Testing Kits market is valued at USD 1,164 million, based on a five-year historical analysis. The market's growth is driven by rising awareness around fertility and reproductive health, as well as advancements in at-home testing technologies. The convenience and accuracy offered by digital ovulation testing kits have made them increasingly popular among women seeking to monitor their fertility. In addition, increasing infertility rates and the trend toward delayed pregnancies have fueled demand for these kits, particularly in developed urban areas with higher healthcare access and awareness.
- In North America, the United States dominates the ovulation testing kits market, followed by Canada. The U.S. maintains its leadership due to a large population base, higher awareness regarding fertility products, and the presence of well-established healthcare infrastructure. Additionally, the U.S. has witnessed a rise in digital ovulation testing kits, which are preferred for their accuracy and convenience, further solidifying its dominance in the market.
- The U.S. FDA plays a critical role in regulating ovulation testing kits. In 2023, the FDA approved over 50 new ovulation testing products, both digital and non-digital, ensuring their safety and accuracy for consumer use. Regulatory oversight helps maintain high standards in the market, boosting consumer confidence in these products. With stringent approval processes, manufacturers must meet rigorous safety and efficacy standards, making FDA approval a significant factor in market entry.
North America Ovulation Testing Kits Market Segmentation
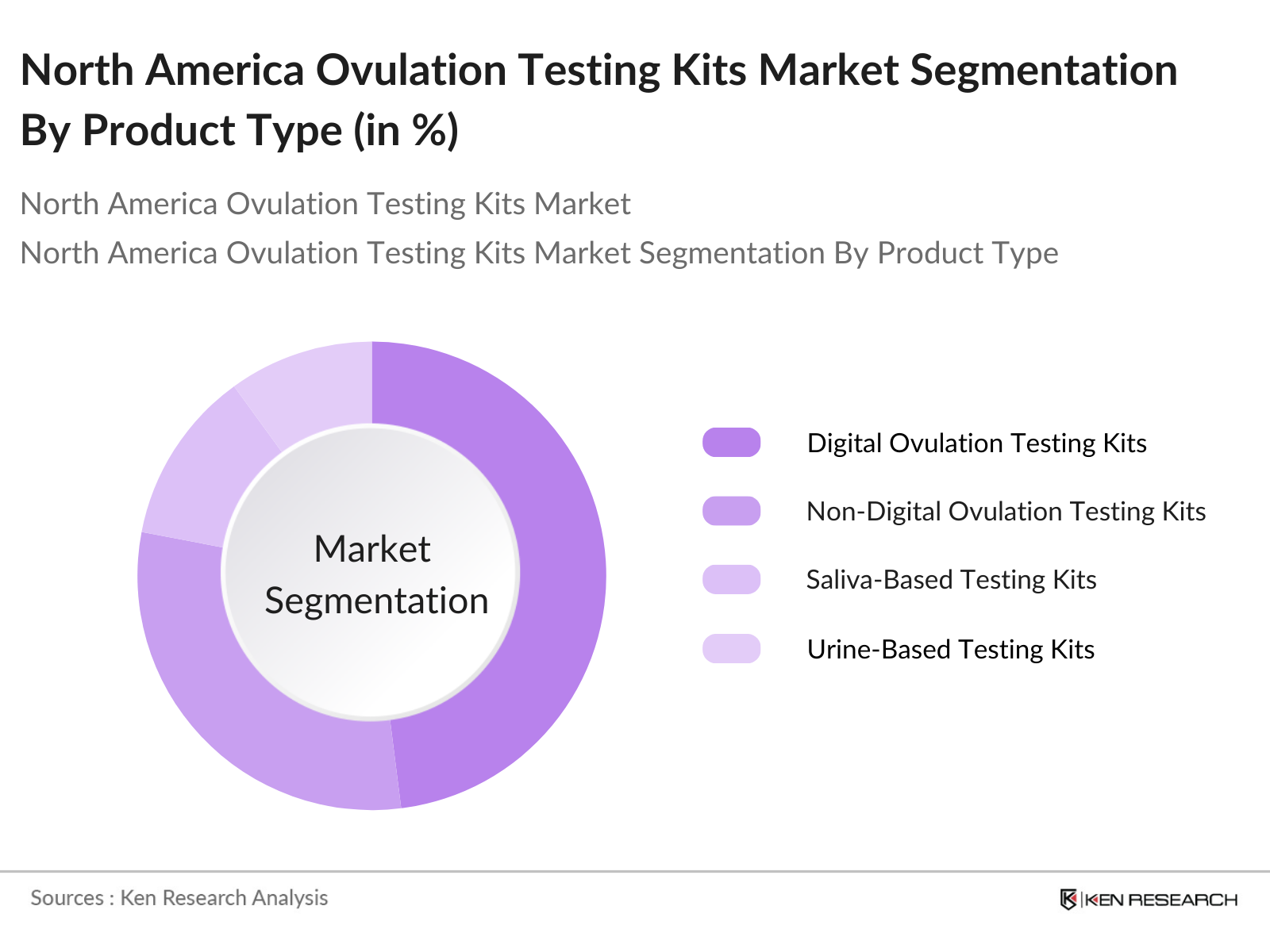
By Product Type: The North America Ovulation Testing Kits market is segmented by product type into digital ovulation testing kits, non-digital ovulation testing kits, saliva-based testing kits, and urine-based testing kits. Among these, digital ovulation testing kits hold the dominant market share in 2023, due to their precision and user-friendly nature. Consumers increasingly prefer these kits for their ability to provide clear results and reduce the chances of user error. Furthermore, these kits are widely available online and offline, making them accessible to a larger consumer base.
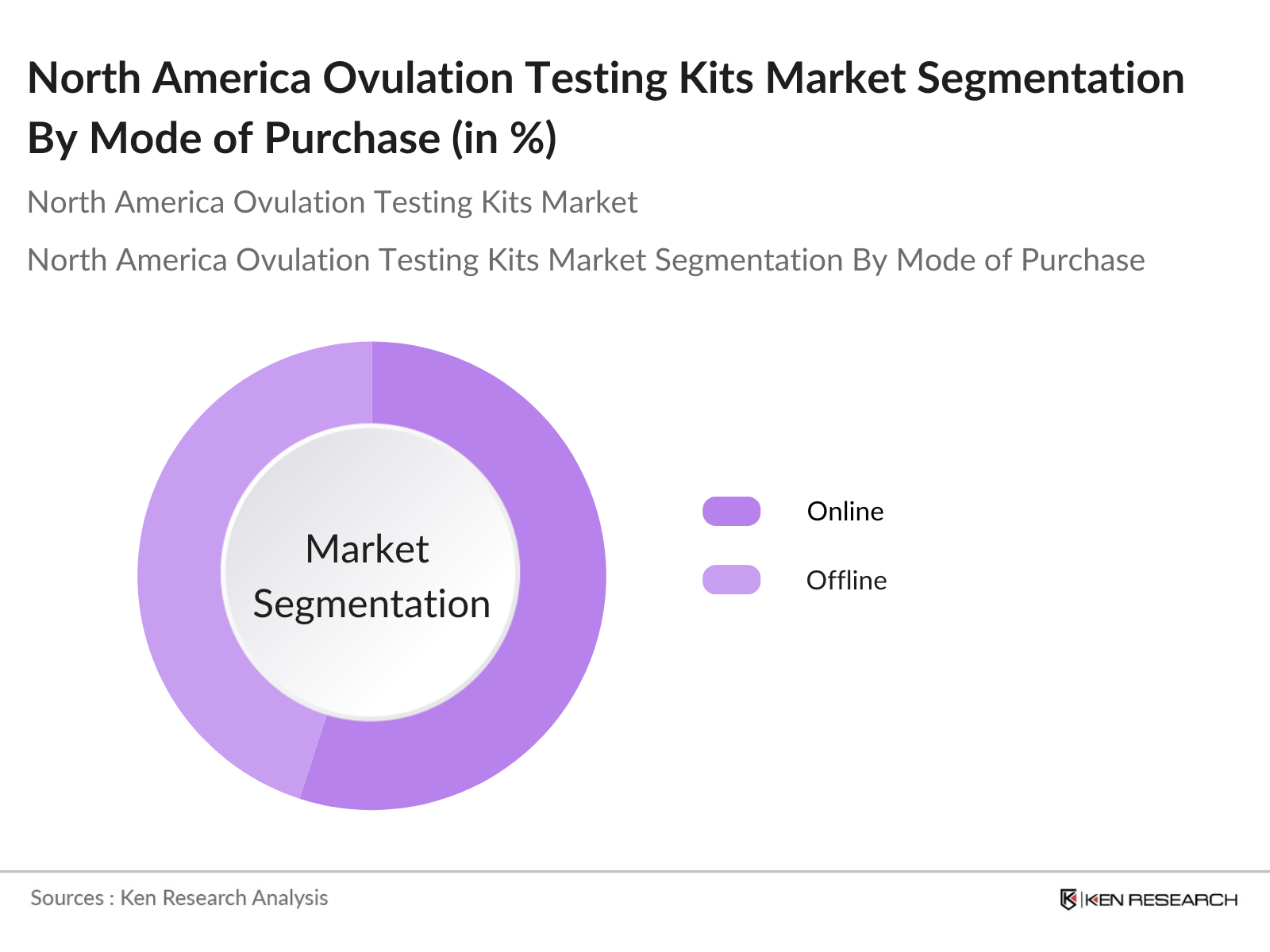
By Mode of Purchase: This market is also segmented by mode of purchase into online and offline channels, with online channels capturing a significant portion of the market share. The online segment has seen an upsurge, driven by the convenience it offers to consumers who prefer purchasing medical devices discreetly. Additionally, the expansion of e-commerce platforms, combined with competitive pricing and faster delivery options, has further boosted the online segment.
North America Ovulation Testing Kits Market Competitive Landscape
The North America Ovulation Testing Kits market is characterized by intense competition among a few key players, primarily multinational companies with an extensive product portfolio. The market is dominated by Swiss Precision Diagnostics (SPD) GmbH, which is known for its flagship product Clearblue, along with other notable players such as Church & Dwight and Fairhaven Health. These companies leverage their global reach, strong distribution networks, and constant innovation to maintain a competitive edge.
North America Ovulation Testing Kits Industry Analysis
Growth Drivers
- Rising Prevalence of Infertility (North America-specific): Infertility rates in North America have been steadily increasing, with approximately 15% of couples of reproductive age affected by infertility. According to the U.S. Centers for Disease Control and Prevention (CDC), nearly 7.5 million women aged 15 to 44 in the U.S. reported seeking infertility treatment in 2022. This growing prevalence is driving the demand for ovulation testing kits, particularly as awareness of infertility issues rises. Infertility treatments, such as assisted reproductive technology, often involve ovulation tracking, further supporting the market for these kits.
- Increasing Awareness of Ovulation Testing (Healthcare Outreach Programs): Healthcare outreach programs across North America have enhanced the awareness of fertility issues and the importance of ovulation testing. In Canada and the U.S., government and non-governmental organizations (NGOs) are promoting fertility education programs that encourage early testing. For example, Health Canadas Fertility Awareness Initiative aims to increase awareness and reduce the stigma around infertility, emphasizing the role of ovulation tracking. The Canadian government reported that in 2023, over 800,000 women participated in fertility-related outreach programs.
- Technological Advancements in Testing Kits (Digital and Non-Digital Kits): Technological advancements in both digital and non-digital ovulation testing kits have contributed significantly to market growth. With the integration of Bluetooth connectivity, smartphone compatibility, and enhanced accuracy, digital testing kits offer more precision in predicting ovulation. In 2024, the U.S. Food and Drug Administration (FDA) approved new digital testing kits that incorporate AI-driven predictions, increasing accuracy to over 99%. This level of precision is particularly important for couples using fertility treatments.
Market Challenges
- Lack of Awareness in Underdeveloped Regions (Rural Population Penetration): Despite growth in urban areas, rural regions of North America face challenges related to fertility awareness and access to ovulation testing kits. A 2023 report from the U.S. Department of Health and Human Services found that women in rural areas were 30% less likely to have access to fertility care, including ovulation testing, due to limited healthcare infrastructure and lower awareness. Addressing these disparities remains a challenge, as market penetration in rural regions lags behind urban centers.
- High Cost of Digital Testing Kits (Pricing Pressure): Digital ovulation testing kits, while technologically advanced, are often expensive, limiting accessibility for lower-income individuals. According to Health Canadas 2023 report, digital kits can cost up to three times more than traditional methods. This creates pricing pressure, as consumers weigh the benefits of digital accuracy against the higher costs. While some private insurance companies offer partial coverage, the high upfront costs deter widespread adoption, particularly in lower-income groups.
North America Ovulation Testing Kits Market Future Outlook
Over the next five years, the North America Ovulation Testing Kits market is projected to witness significant growth, driven by technological advancements in digital health products, the increasing preference for home-based fertility solutions, and the rising awareness surrounding fertility treatments. Continuous product innovations, along with strategic collaborations between healthcare providers and testing kit manufacturers, are expected to drive market expansion.
Market Opportunities
- Growth in E-Commerce Distribution Channels (Online Market Expansion): The shift towards e-commerce in North America presents a significant opportunity for ovulation testing kits. In 2023, over 67 million Americans purchased health and wellness products online, according to the U.S. Department of Commerce. This trend is expected to continue as consumers seek the convenience of home delivery and discreet purchasing. The rise of e-commerce platforms like Amazon and Walmart, which now offer subscription-based ovulation test deliveries, supports the market's growth by expanding access to these products, particularly in underserved areas.
- Integration of Ovulation Testing Kits with Smartphone Apps (Digital Health Integration): Ovulation testing kits integrated with smartphone apps have become a popular option for tech-savvy consumers. The CDC reported that in 2023, over 80 million smartphone users in North America downloaded fertility and ovulation tracking apps. These apps, when combined with testing kits, provide personalized insights into fertility patterns, increasing user engagement. This integration presents a growing market opportunity, as digital health solutions continue to be embraced by consumers seeking more control over their reproductive health.
Scope of the Report
|
Product Type |
Digital Ovulation Testing Kits |
|
Mode of Purchase |
Online |
|
Application |
Hospitals |
|
Distribution Channel |
E-Commerce |
|
Region |
United States |
Products
Key Target Audience
Manufacturers of Ovulation Testing Kits
Distributors and Suppliers of Fertility Devices
Healthcare Providers (Hospitals, Clinics)
Retail Pharmacies and E-Commerce Platforms
Investments and Venture Capital Firms
Government and Regulatory Bodies (FDA, Health Canada)
Telemedicine and Digital Health Platforms
Pharmaceutical Companies and Fertility Specialists
Companies
Players Mentioned in the Report
Swiss Precision Diagnostics (SPD) GmbH
Church & Dwight Co., Inc.
Fairhaven Health, LLC
bioZhena Corporation
HiLin Life Products, Inc.
Geratherm Medical AG
Piramal Enterprises Ltd.
Fertility Focus Ltd.
AccuQuik Test Kits
Inito Pte Ltd.
Table of Contents
1. North America Ovulation Testing Kits Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2. North America Ovulation Testing Kits Market Size (In USD Bn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
3. North America Ovulation Testing Kits Market Analysis
3.1. Growth Drivers
3.1.1. Rising Prevalence of Infertility (North America-specific)
3.1.2. Increasing Awareness of Ovulation Testing (Healthcare Outreach Programs)
3.1.3. Technological Advancements in Testing Kits (Digital and Non-Digital Kits)
3.1.4. Rise in Late Parenthood Trends (Demographic Changes)
3.2. Market Challenges
3.2.1. Lack of Awareness in Underdeveloped Regions (Rural Population Penetration)
3.2.2. High Cost of Digital Testing Kits (Pricing Pressure)
3.2.3. Market Saturation in Urban Areas (Competitive Landscape)
3.3. Opportunities
3.3.1. Growth in E-Commerce Distribution Channels (Online Market Expansion)
3.3.2. Integration of Ovulation Testing Kits with Smartphone Apps (Digital Health Integration)
3.3.3. Strategic Partnerships with Healthcare Providers (Partnership Opportunities)
3.4. Trends
3.4.1. Increasing Consumer Preference for At-Home Fertility Testing Kits
3.4.2. Growing Adoption of Digital Ovulation Testing Kits
3.4.3. Rising Focus on Personalized Healthcare Solutions
3.5. Government Regulation
3.5.1. U.S. FDA Approvals (Regulatory Landscape)
3.5.2. Health Canada Regulations for Medical Devices
3.5.3. Reimbursement Policies (Insurance Coverage)
3.6. SWOT Analysis
3.7. Stakeholder Ecosystem
3.8. Porters Five Forces Analysis (Market-Specific Competitive Dynamics)
3.9. Competitive Ecosystem
4. North America Ovulation Testing Kits Market Segmentation
4.1. By Product Type (In Value %)
4.1.1. Digital Ovulation Testing Kits
4.1.2. Non-Digital Ovulation Testing Kits
4.1.3. Saliva-Based Testing Kits
4.1.4. Urine-Based Testing Kits
4.2. By Mode of Purchase (In Value %)
4.2.1. Online
4.2.2. Offline (Retail Pharmacies, Drug Stores)
4.3. By Application (In Value %)
4.3.1. Hospitals
4.3.2. Clinics
4.3.3. Home Care
4.4. By Distribution Channel (In Value %)
4.4.1. E-Commerce
4.4.2. Pharmacy Stores
4.4.3. Supermarkets and Hypermarkets
4.5. By Country (In Value %)
4.5.1. United States
4.5.2. Canada
4.5.3. Mexico
5. North America Ovulation Testing Kits Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. Swiss Precision Diagnostics (SPD) GmbH
5.1.2. Church & Dwight Co., Inc.
5.1.3. Fairhaven Health, LLC
5.1.4. bioZhena Corporation
5.1.5. HiLin Life Products, Inc.
5.1.6. Geratherm Medical AG
5.1.7. Piramal Enterprises Ltd.
5.1.8. Fertility Focus Ltd.
5.1.9. AccuQuik Test Kits
5.1.10. Inito Pte Ltd.
5.1.11. AVA Women
5.1.12. Clearblue
5.1.13. Wondfo Biotech
5.1.14. MFB Fertility, Inc.
5.1.15. Ningbo David Medical Device Co., Ltd.
5.2. Cross Comparison Parameters
(Headquarters, Inception Year, Revenue, Product Portfolio, Digital Innovation, Sales Channels, Distribution Network, Market Reach)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers and Acquisitions
5.6. Investment Analysis
5.6.1. Venture Capital Funding
5.6.2. Government Grants
5.6.3. Private Equity Investments
6. North America Ovulation Testing Kits Market Regulatory Framework
6.1. FDA Regulations and Approvals
6.2. Compliance Requirements
6.3. Certification Processes
7. North America Ovulation Testing Kits Future Market Size (In USD Bn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. North America Ovulation Testing Kits Future Market Segmentation
8.1. By Product Type (In Value %)
8.2. By Application (In Value %)
8.3. By Mode of Purchase (In Value %)
8.4. By Distribution Channel (In Value %)
8.5. By Country (In Value %)
9. North America Ovulation Testing Kits Market Analysts' Recommendations
9.1. TAM/SAM/SOM Analysis
9.2. Customer Cohort Analysis
9.3. Marketing Initiatives
9.4. White Space Opportunity Analysis
Research Methodology
Step 1: Identification of Key Variables
The initial step involves creating an ecosystem map of all significant players in the North America Ovulation Testing Kits Market. This includes conducting desk research and gathering data from proprietary databases to identify key factors such as consumer behavior, distribution channels, and product innovations.
Step 2: Market Analysis and Construction
In this phase, historical data is analyzed to assess market penetration and growth trends in the ovulation testing kits sector. Various qualitative and quantitative parameters such as product demand, pricing analysis, and consumer feedback are evaluated to build a robust market forecast model.
Step 3: Hypothesis Validation and Expert Consultation
Hypotheses regarding market dynamics are formulated and validated by consulting industry experts through structured interviews. Insights from these professionals provide a deeper understanding of the competitive landscape and consumer preferences.
Step 4: Research Synthesis and Final Output
The final step involves synthesizing the collected data and validating the findings through further market interactions. The data is then structured into a comprehensive report, ensuring accurate and actionable insights for key stakeholders in the North America Ovulation Testing Kits market.
Frequently Asked Questions
01. How big is the North America Ovulation Testing Kits Market?
The North America Ovulation Testing Kits Market is valued at USD USD 1,164 million and is driven by rising awareness surrounding fertility and the increasing preference for home-based testing solutions.
02. What are the challenges in the North America Ovulation Testing Kits Market?
Challenges include the high cost of digital testing kits, market saturation in urban areas, and the lack of awareness in rural populations, which limits the market penetration of advanced ovulation testing solutions.
03. Who are the major players in the North America Ovulation Testing Kits Market?
Key players in this market include Swiss Precision Diagnostics (SPD) GmbH, Church & Dwight Co., Inc., Fairhaven Health, and bioZhena Corporation, known for their strong brand presence and product innovation.
04. What are the growth drivers of the North America Ovulation Testing Kits Market?
The market is propelled by increasing infertility rates, technological advancements in testing kits, and the rising consumer demand for easy-to-use, home-based fertility monitoring solutions.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















