
United States Aseptic Sampling Market Outlook to 2030
Region:North America
Author(s):Sanjeev
Product Code:KROD10350
December 2024
89
About the Report
United States Aseptic Sampling Market Overview
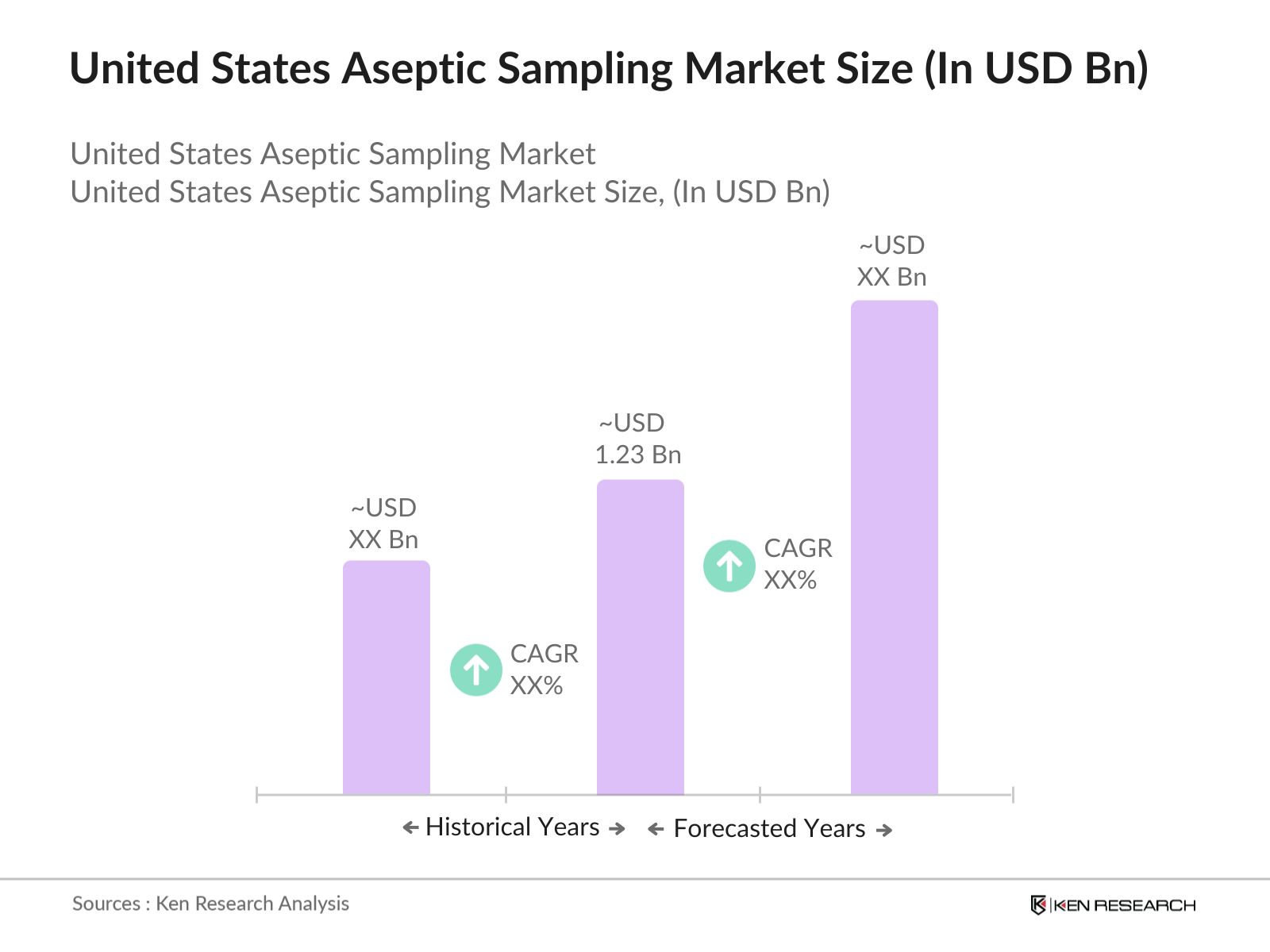
- The United States aseptic sampling market is valued at USD 1.23 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for biopharmaceuticals and the adoption of single-use aseptic sampling systems, which enhance process efficiency and reduce contamination risks.
- Major cities such as Boston, San Francisco, and San Diego dominate the market due to their robust biotechnology and pharmaceutical industries. These regions are hubs for research and development, housing numerous biotech firms and research institutions that contribute significantly to the aseptic sampling market.
- The FDA continues to update its guidelines on aseptic processing to enhance product safety. In 2023, the FDA released a comprehensive report emphasizing the importance of maintaining aseptic conditions during manufacturing. The guidelines outline the necessity for rigorous training and adherence to standard operating procedures (SOPs) to prevent contamination. In response to past compliance failures, the FDA has increased oversight, resulting in over 400 inspections specifically focused on aseptic processes in 2022. This regulatory emphasis underscores the critical need for robust aseptic sampling systems to meet stringent industry standards.
United States Aseptic Sampling Market Segmentation
The United States aseptic sampling market is segmented by type and by application.
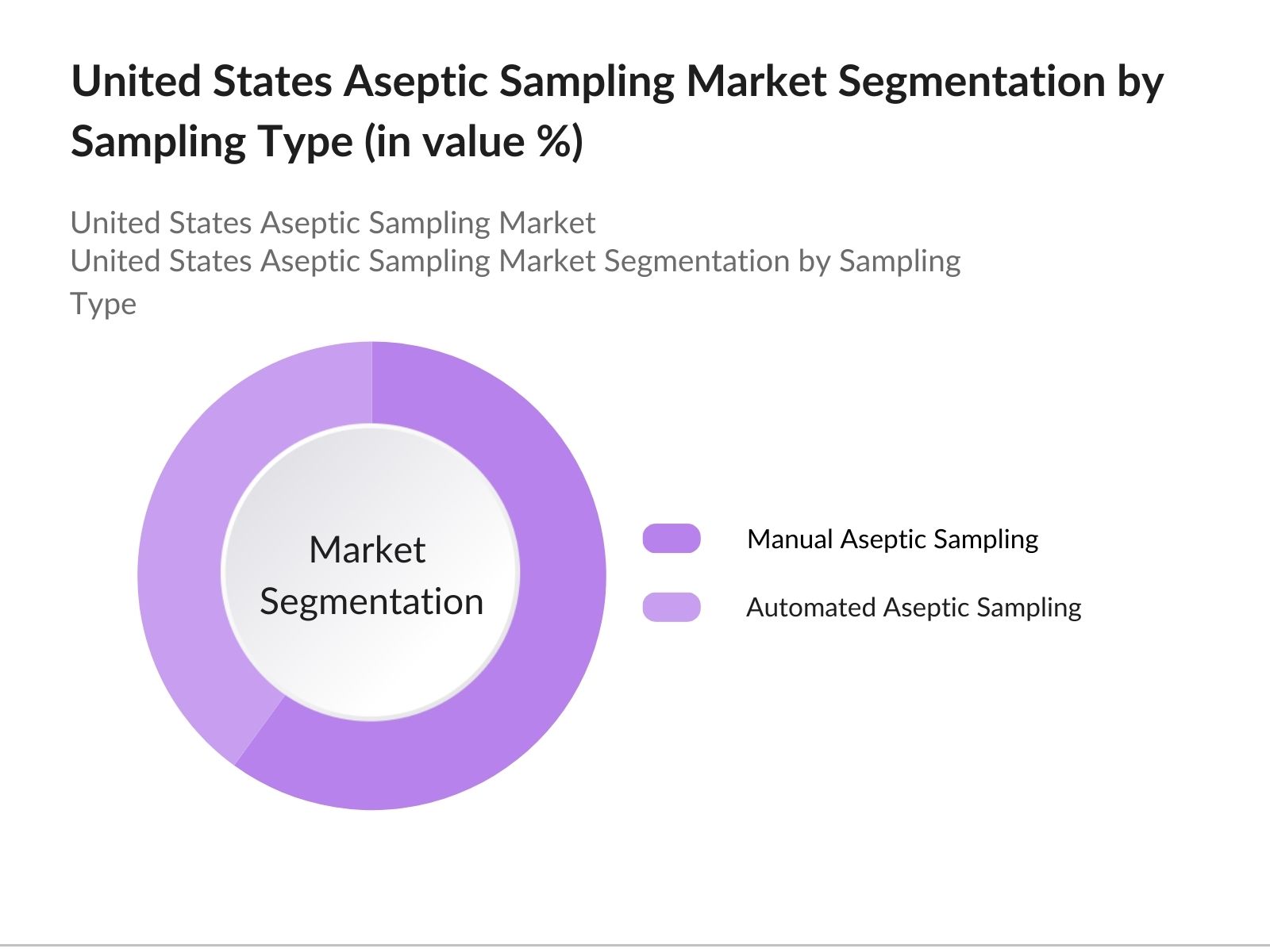
- By Sampling Type: The United States aseptic sampling market is segmented by type into manual aseptic sampling and automated aseptic sampling. Manual aseptic sampling currently holds a dominant market share due to its simplicity and lower equipment costs. Disposable sampling bags, bottles, and tubes offer convenience in collecting samples, making manual methods preferable in various applications.
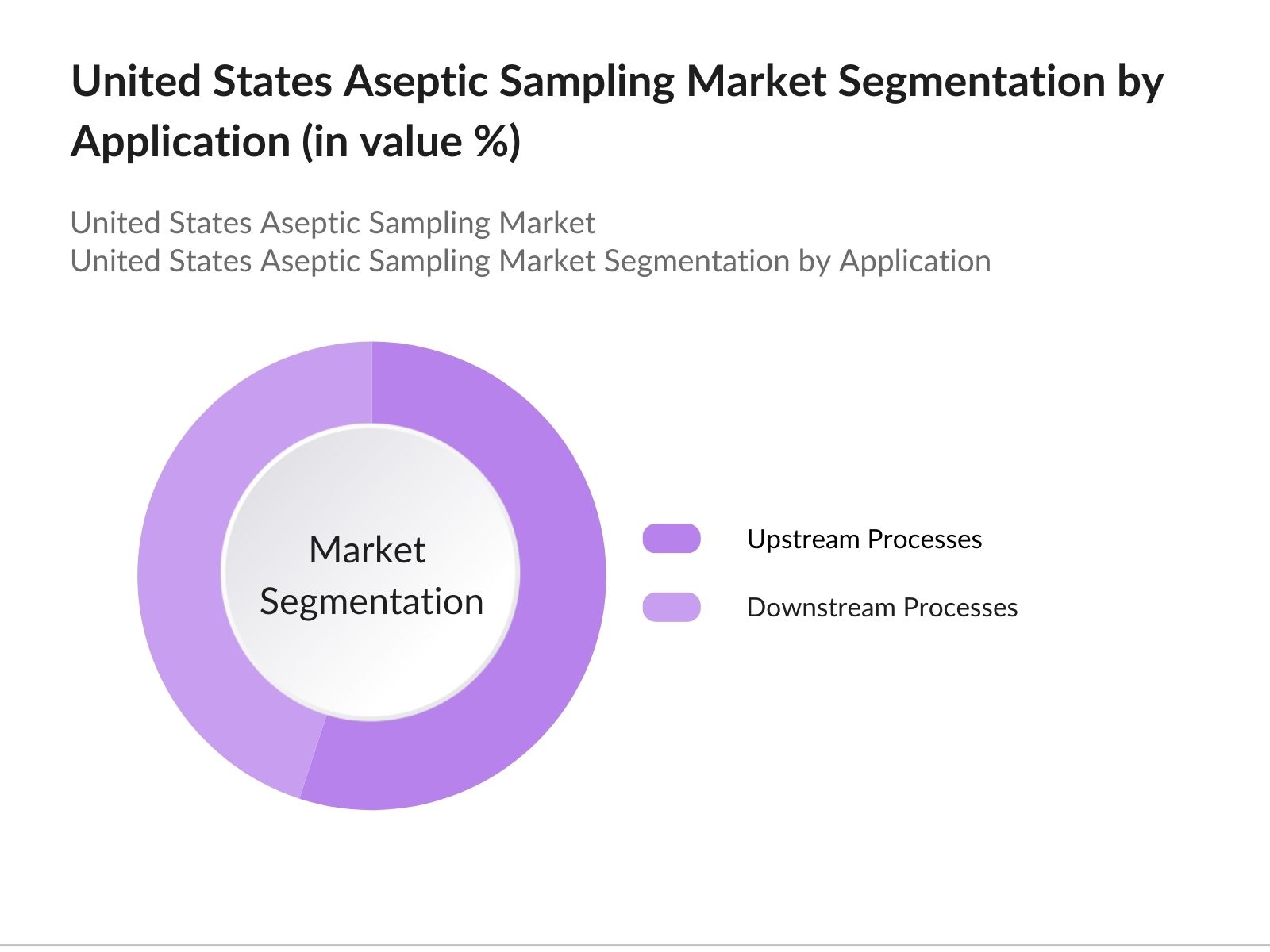
- By Application: The market is also segmented by application into upstream processes and downstream processes. Upstream processes account for the largest market share, driven by the increasing demand for biologics and pharmaceutical agents. Proper storage of microorganisms, cell isolation, and cultivation are critical in upstream processes, necessitating reliable aseptic sampling methods to ensure product quality and safety.
United States Aseptic Sampling Market Competitive Landscape
The United States aseptic sampling market is characterized by the presence of several key players who contribute significantly to market dynamics. These companies are engaged in continuous research and development to enhance product offerings and maintain a competitive edge.
United States Aseptic Sampling Industry Analysis
Growth Drivers
- Increasing Demand for Biopharmaceuticals: The global biopharmaceutical market reached a valuation of approximately $307.7 billion in 2022, reflecting robust growth driven by an aging population and rising chronic disease prevalence. In the U.S., the National Institutes of Health (NIH) reported that around 6 in 10 adults have a chronic disease, prompting a shift towards biopharmaceuticals that require aseptic sampling for quality assurance. Furthermore, the projected increase in biologic drugs is estimated to drive demand for aseptic techniques, essential for preventing contamination in critical manufacturing processes. This growing trend emphasizes the need for reliable aseptic sampling systems across the biopharmaceutical sector.
- Rising Incidence of Contaminated Products: The FDA reported that 35% of all recalls in 2022 were due to contamination, highlighting the critical need for stringent sampling methods. With the biopharmaceutical sectors shift towards more complex manufacturing processes, the incidence of contaminated products necessitates the adoption of robust aseptic sampling protocols. Additionally, the Centers for Disease Control and Prevention (CDC) indicates that hospital-acquired infections affect 1 in 31 hospital patients on any given day, further underscoring the urgency for improved aseptic techniques. This increase in contamination incidents stresses the importance of effective aseptic sampling to ensure product safety.
- Strict Regulatory Compliance: In 2023, the FDA introduced more rigorous guidelines aimed at improving aseptic processing, mandating that all pharmaceutical manufacturers adhere to strict compliance standards. This includes adherence to current Good Manufacturing Practices (cGMP), which are enforced to ensure product quality and safety. The 2022 FDA report indicated that non-compliance resulted in over 1,000 warning letters, emphasizing the critical need for effective aseptic sampling. Regulatory bodies globally are enhancing oversight, necessitating manufacturers to invest in advanced aseptic techniques, thereby driving market growth. Compliance with these regulations is essential for maintaining market access and consumer trust.
Market Challenges
- High Cost of Aseptic Sampling Systems: The initial investment for advanced aseptic sampling systems can exceed $250,000, which poses a barrier for many small and medium-sized enterprises. This significant expenditure includes costs associated with training, equipment maintenance, and compliance with regulatory standards. According to the Small Business Administration, about 60% of small businesses report that access to capital is a challenge, which impacts their ability to invest in necessary aseptic sampling technologies. Consequently, this challenge could hinder the overall growth of the aseptic sampling market, particularly among smaller players seeking to compete in the biopharmaceutical industry. Source: SBA
- Limited Awareness Among Small Manufacturers: Research indicates that approximately 40% of small-scale manufacturers lack awareness of the importance of aseptic sampling, which compromises product safety and regulatory compliance. The FDA's outreach efforts have highlighted this gap, with many small manufacturers unaware of the latest guidelines and technologies available for aseptic sampling. This lack of knowledge not only increases the risk of contamination but also limits market participation, as companies that fail to implement proper sampling techniques may face regulatory action or product recalls. Raising awareness and providing education are crucial for enabling small manufacturers to improve their practices.
United States Aseptic Sampling Market Future Outlook
Over the next five years, the United States aseptic sampling market is expected to experience significant growth. This expansion will be driven by continuous advancements in aseptic sampling technologies, increasing investments in biopharmaceutical research, and the growing emphasis on maintaining product quality and safety standards. Additionally, the adoption of automated sampling systems and the integration of process analytical technology (PAT) are anticipated to further propel market growth.
Market Opportunities
- Growth of the Biotechnology Sector: The biotechnology sector in the U.S. is projected to contribute approximately $1.5 trillion to the economy by 2025, driven by innovations in drug development and personalized medicine. As the sector expands, the demand for reliable aseptic sampling methods will increase to ensure the safety and efficacy of biopharmaceutical products. Additionally, the National Biotechnology Information Center reports that the number of biotechnology firms has grown by 20% from 2022 to 2023, indicating a vibrant market landscape. This growth presents a significant opportunity for manufacturers of aseptic sampling systems to cater to the burgeoning needs of the biotechnology sector.
- Expansion of Research & Development Activities: Federal funding for research and development (R&D) in the biopharmaceutical industry reached approximately $47 billion in 2023, reflecting a strong commitment to innovation. This influx of investment is expected to boost the demand for aseptic sampling solutions, as R&D facilities require stringent quality controls to maintain product integrity. Furthermore, the National Institutes of Health (NIH) reported that over 50% of new therapeutic entities are derived from R&D efforts, emphasizing the critical role of aseptic sampling in the development process. As R&D activities expand, the demand for advanced sampling techniques will continue to grow, presenting lucrative opportunities for market players.
Scope of the Report
|
Manual Aseptic Sampling Automated Aseptic Sampling |
|
|
By Technique |
Off-line Sampling Technique |
|
By Application |
Downstream Processing |
|
By End-Use |
Pharmaceutical & Biotechnology Companies |
|
By Region |
North East West South |
Products
Key Target Audience
Pharmaceutical & Biotechnology Companies
Contract Manufacturing Organizations (CMOs) & Contract Research Organizations (CROs)
Medical Device Manufacturers
Quality Assurance and Control Departments
Regulatory Affairs Professionals
Government and Regulatory Bodies (e.g., FDA, EPA)
Investments and Venture Capitalist Firms
Academic and Research Institutions
Companies
Major Players in the Market
Merck KGaA
Sartorius Stedim Biotech
Thermo Fisher Scientific
QualiTru Sampling Systems
Keofitt A/S
Emerson Electric Co.
Advanced Microdevices Pvt. Ltd.
Lonza Group
Sampling Systems Ltd.
Parasol Medical LLC
Bbi-biotech GmbH
KIESELMANN GmbH
Centec GmbH
3M Company
GEA Group
Table of Contents
1. United States Aseptic Sampling Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2. United States Aseptic Sampling Market Size (In USD Bn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
3. United States Aseptic Sampling Market Analysis
3.1. Growth Drivers
3.1.1. Increasing Demand for Biopharmaceuticals
3.1.2. Stringent Government Regulations
3.1.3. Advancements in Single-Use Technologies
3.1.4. Rising R&D Investments
3.2. Market Challenges
3.2.1. Concerns Over Extractables and Leachables
3.2.2. High Implementation Costs
3.2.3. Limited Skilled Workforce
3.3. Opportunities
3.3.1. Technological Innovations
3.3.2. Expansion into Emerging Markets
3.3.3. Collaborations and Partnerships
3.4. Trends
3.4.1. Adoption of Automated Sampling Systems
3.4.2. Integration with Process Analytical Technology (PAT)
3.4.3. Emphasis on Quality Assurance and Control
3.5. Government Regulation
3.5.1. FDA Guidelines on Aseptic Processing
3.5.2. cGMP Compliance Requirements
3.5.3. Environmental Monitoring Standards
3.5.4. Biologics License Application (BLA) Procedures
3.6. SWOT Analysis
3.7. Stakeholder Ecosystem
3.8. Porters Five Forces
3.9. Competitive Landscape
4. United States Aseptic Sampling Market Segmentation
4.1. By Type (In Value %)
4.1.1. Manual Aseptic Sampling
4.1.2. Automated Aseptic Sampling
4.2. By Technique (In Value %)
4.2.1. Off-line Sampling Technique
4.2.2. On-line Sampling Technique
4.2.3. In-line Sampling Technique
4.3. By Application (In Value %)
4.3.1. Downstream Processing
4.3.2. Upstream Processing
4.4. By End-Use (In Value %)
4.4.1. Pharmaceutical & Biotechnology Companies
4.4.2. Contract Manufacturing Organizations (CMOs) & Contract Research Organizations (CROs)
4.4.3. Others
4.5. By Region (In Value %)
4.5.1. Northeast
4.5.2. Midwest
4.5.3. South
4.5.4. West
5. United States Aseptic Sampling Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. Merck KGaA
5.1.2. Sartorius Stedim Biotech
5.1.3. Thermo Fisher Scientific
5.1.4. QualiTru Sampling Systems
5.1.5. Keofitt A/S
5.1.6. Emerson Electric Co.
5.1.7. Advanced Microdevices Pvt. Ltd.
5.1.8. Lonza Group
5.1.9. Sampling Systems Ltd.
5.1.10. Parasol Medical LLC
5.1.11. Bbi-biotech GmbH
5.1.12. KIESELMANN GmbH
5.1.13. Centec GmbH
5.1.14. 3M Company
5.1.15. GEA Group
5.2. Cross Comparison Parameters (Revenue, Market Share, Product Portfolio, R&D Investment, Regional Presence, Strategic Initiatives, Employee Strength, Year Established)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers and Acquisitions
5.6. Investment Analysis
5.7. Venture Capital Funding
5.8. Government Grants
5.9. Private Equity Investments
6. United States Aseptic Sampling Market Regulatory Framework
6.1. FDA Regulations
6.2. cGMP Standards
6.3. Environmental Protection Agency (EPA) Guidelines
6.4. Occupational Safety and Health Administration (OSHA) Standards
7. United States Aseptic Sampling Future Market Size (In USD Bn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. United States Aseptic Sampling Future Market Segmentation
8.1. By Type (In Value %)
8.2. By Technique (In Value %)
8.3. By Application (In Value %)
8.4. By End-Use (In Value %)
8.5. By Region (In Value %)
9. United States Aseptic Sampling Market Analysts Recommendations
9.1. Total Addressable Market (TAM), Serviceable Available Market (SAM), Serviceable Obtainable Market (SOM) Analysis
9.2. Customer Cohort Analysis
9.3. Marketing Initiatives
9.4. White Space Opportunity Analysis
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
The initial phase involves constructing an ecosystem map encompassing all major stakeholders within the United States aseptic sampling market. This step is underpinned by extensive desk research, utilizing a combination of secondary and proprietary databases to gather comprehensive industry-level information. The primary objective is to identify and define the critical variables that influence market dynamics.
Step 2: Market Analysis and Construction
In this phase, we compile and analyze historical data pertaining to the United States aseptic sampling market. This includes assessing market penetration, the ratio of marketplaces to service providers, and the resultant revenue generation. Furthermore, an evaluation of service quality statistics is conducted to ensure the reliability and accuracy of the revenue estimates.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are developed and subsequently validated through computer-assisted telephone interviews (CATIs) with industry experts representing a diverse array of companies. These consultations provide valuable operational and financial insights directly from industry practitioners, which are instrumental in refining and corroborating the market data.
Step 4: Research Synthesis and Final Output
The final phase involves direct engagement with multiple aseptic sampling equipment manufacturers to acquire detailed insights into product segments, sales performance, consumer preferences, and other pertinent factors. This interaction serves to verify and complement the statistics derived from the bottom-up approach, thereby ensuring a comprehensive, accurate, and validated analysis of the United States aseptic sampling market.
Frequently Asked Questions
01. How big is the United States Aseptic Sampling Market?
The United States aseptic sampling market is valued at USD 1.23 billion, driven by the growing demand for contamination-free sampling in biopharmaceutical and food industries.
02. What are the main drivers of the United States Aseptic Sampling Market?
United States aseptic sampling market Key drivers include increasing demand for biopharmaceutical products, advancements in single-use technologies, and stringent regulatory requirements focused on product safety and contamination control.
03. Which segment holds the largest share in the United States Aseptic Sampling Market?
Manual aseptic sampling dominates the United States aseptic sampling market due to its lower equipment costs and ease of use, especially in smaller-scale operations.
04. Who are the major players in the United States Aseptic Sampling Market?
United States aseptic sampling market Key players include Merck KGaA, Sartorius Stedim Biotech, Thermo Fisher Scientific, QualiTru Sampling Systems, and Keofitt A/S, who led through continuous innovation and extensive distribution networks.
05. What challenges does the United States Aseptic Sampling Market face?
United States aseptic sampling market Challenges include the high implementation costs of automated systems and concerns over extractables and leachables in single-use systems, which may affect product integrity.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















