
USA Pharmaceutical Excipients Market Outlook to 2030
Region:North America
Author(s):Samanyu
Product Code:KROD5287
November 2024
95
About the Report
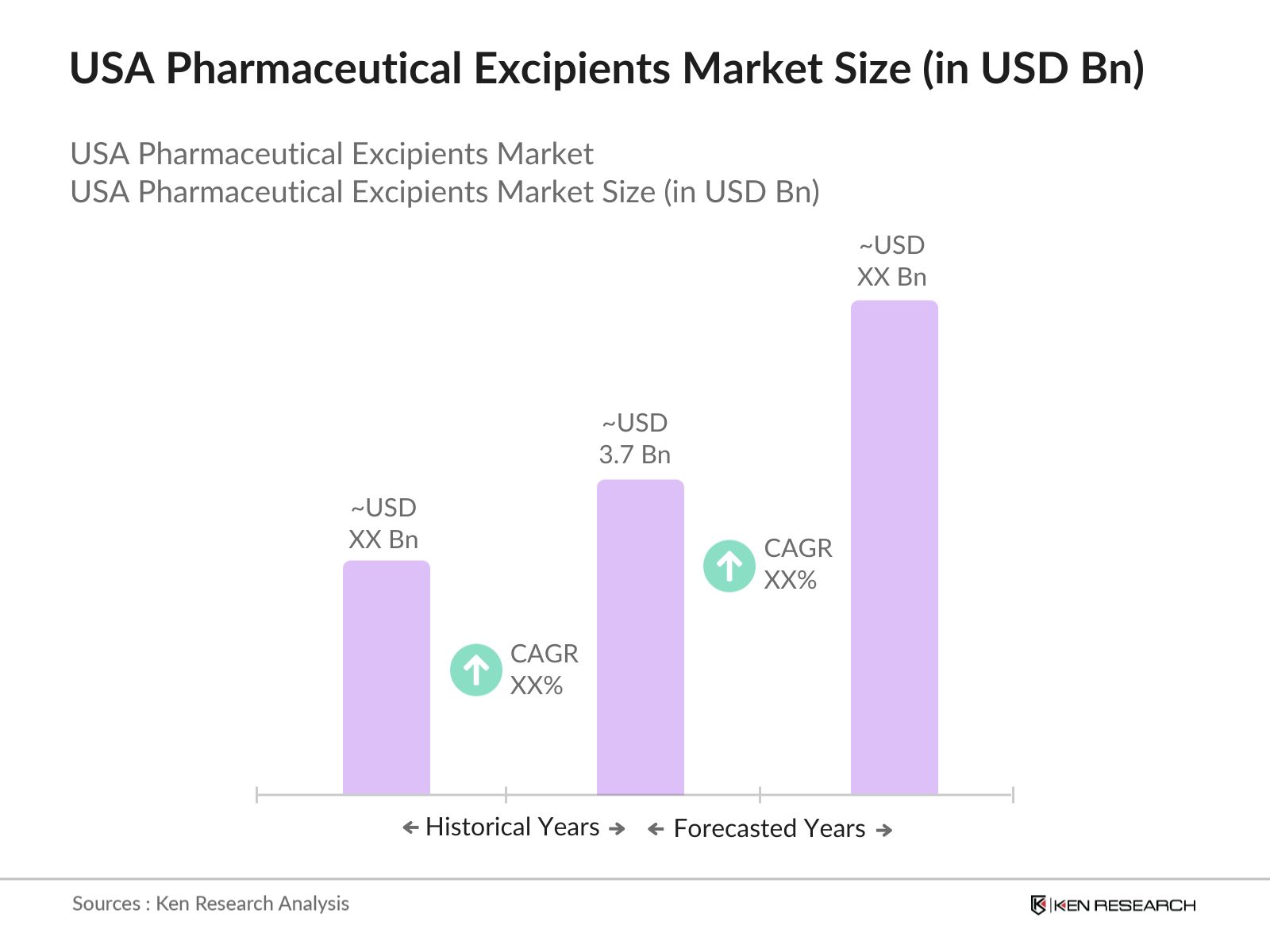
USA Pharmaceutical Excipients Market Overview
- The USA Pharmaceutical Excipients market is valued at USD 3.7 Bn, based on a five-year historical analysis. This market is primarily driven by the growing demand for pharmaceutical formulations in the U.S., propelled by the expansion of the biopharmaceutical industry, increasing prevalence of chronic diseases, and advancements in drug delivery systems. Excipients play a critical role in the stabilization, bioavailability, and delivery of active pharmaceutical ingredients, which is driving the markets sustained growth.
- The USA is a dominant force in the global pharmaceutical excipients market, with cities such as New York, Boston, and San Francisco leading the charge due to their strong concentration of pharmaceutical R&D, cutting-edge manufacturing technologies, and favorable regulatory environment. These cities are home to a large number of pharmaceutical companies and research institutions, which collaborate closely to innovate in drug formulations. Furthermore, regulatory frameworks such as the U.S. FDAs stringent guidelines ensure high standards, giving the U.S. a competitive edge in the market.
- Environmental sustainability is becoming a priority for the pharmaceutical industry. In 2024, biodegradable excipients, particularly those derived from renewable sources, have seen a 10% increase in demand. According to the Environmental Protection Agency (EPA), pharmaceutical waste accounts for 2-3% of hazardous waste in the U.S., driving the need for eco-friendly alternatives in drug formulations. Biodegradable excipients are also gaining popularity due to their reduced environmental impact during manufacturing and disposal, positioning them as a key trend in sustainable pharmaceutical development.
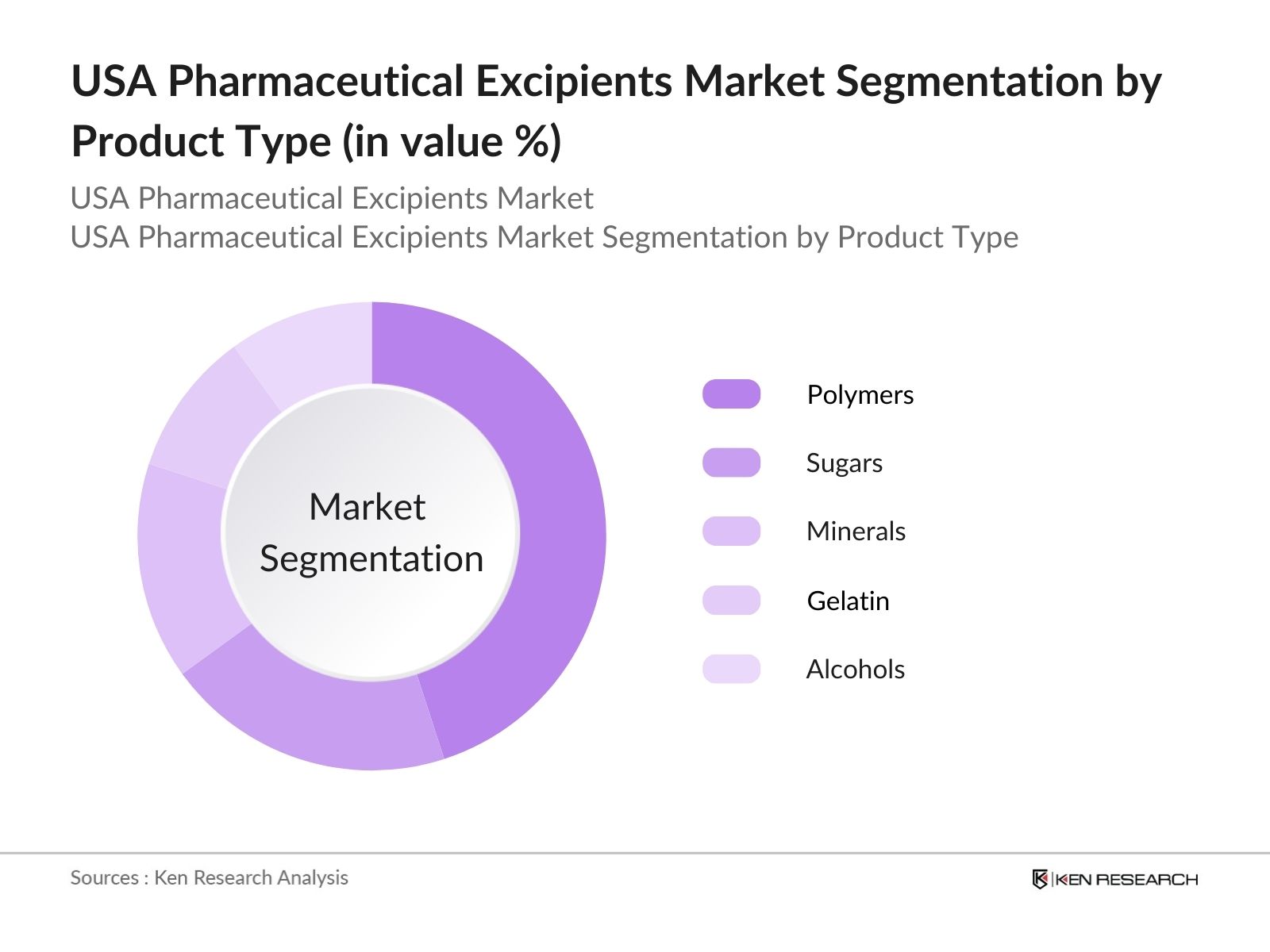
USA Pharmaceutical Excipients Market Segmentation
By Product Type: The market is segmented by product type into polymers, sugars, minerals, gelatin, and alcohols. Recently, polymers have dominated the product type segmentation due to their versatility in pharmaceutical formulations. Polymers are widely used in drug delivery systems because of their ability to control the release of active ingredients, ensuring the drug reaches its target more effectively. Polymers like polyethylene glycol (PEG) are frequently used in controlled-release formulations, contributing to their dominant position.
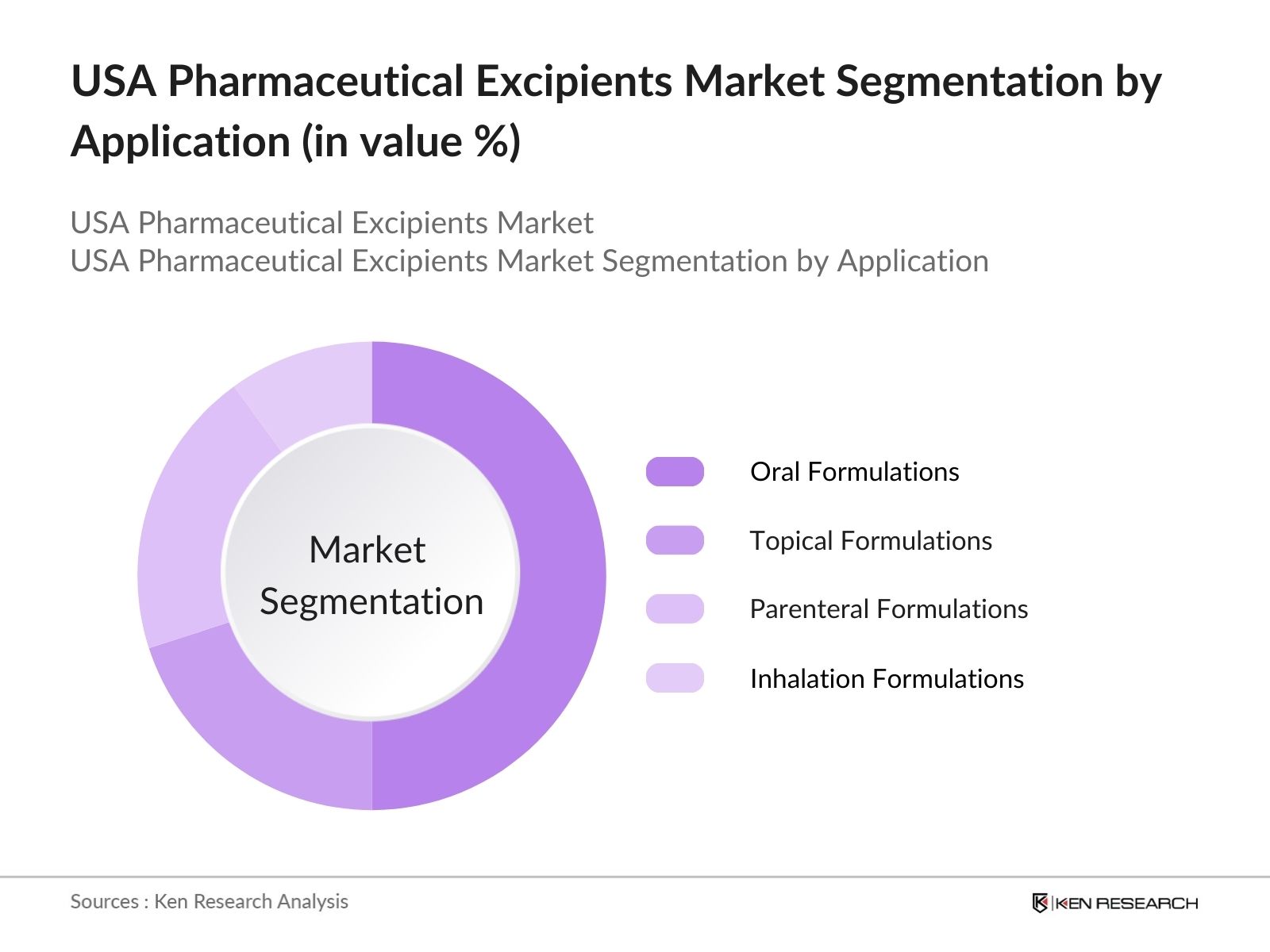
By Application: The market is also segmented by application into oral formulations, topical formulations, parenteral formulations, and inhalation formulations. Oral formulations hold the dominant market share due to their widespread use and patient preference for oral drug delivery methods. Tablets and capsules are the most common forms of oral formulations, and excipients are essential for improving their taste, stability, and bioavailability, making them an indispensable part of the pharmaceutical industry.
USA Pharmaceutical Excipients Market Competitive Landscape
The USA Pharmaceutical Excipients market is dominated by several key players, each contributing significantly to the market's development. This consolidation is marked by established players who have a strong foothold in the market due to their extensive R&D capabilities, strong product portfolios, and collaborations with pharmaceutical manufacturers. The key companies focus on innovation in excipient development to address the evolving needs of the pharmaceutical industry.
|
Company Name |
Establishment Year |
Headquarters |
Revenue (2023) |
Employees |
Key Product |
R&D Investment |
Key Clients |
Partnerships |
|
BASF SE |
1865 |
Ludwigshafen, Germany |
||||||
|
Evonik Industries AG |
2007 |
Essen, Germany |
||||||
|
Ashland Global Holdings Inc. |
1924 |
Wilmington, USA |
||||||
|
Roquette Frres |
1933 |
Lestrem, France |
||||||
|
The Dow Chemical Company |
1897 |
Midland, USA |
USA Pharmaceutical Excipients Industry Analysis
Growth Drivers
- Increasing Demand for Generic Drugs: The USA has been witnessing substantial growth in the demand for generic drugs, driven by cost-saving measures and the expiration of patents for several major drugs. In 2022, the FDA approved over 95 generic drugs, contributing to a competitive marketplace that requires large-scale production of excipients. According to the FDAs Office of Generic Drugs, nearly 90% of all prescriptions filled in the U.S. are for generics, amplifying the demand for pharmaceutical excipients in 2024 to maintain quality standards in drug formulation, stability, and delivery.
- Growing Biopharmaceutical Industry: The biopharmaceutical sector has expanded significantly in the USA, with biopharmaceuticals accounting for approximately 30% of all new drug approvals by the FDA in 2023. Excipients play a critical role in stabilizing these complex biologics. Macroeconomic data from the U.S. Department of Commerce shows that the biopharmaceutical sector saw employment of over 850,000 professionals in 2023, with research spending exceeding USD 100 billion. This rising demand for biopharmaceuticals has necessitated the use of innovative excipients to ensure product stability and efficacy in advanced therapeutic formulations.
- Rising Preference for Novel Drug Delivery Systems: There has been an increasing shift towards novel drug delivery systems such as transdermal, inhalation, and controlled-release formulations. In 2024, over 15% of all new drugs approved by the FDA were in non-oral delivery formats, requiring specialized excipients for targeted delivery. The shift in drug delivery systems is driven by patient preferences for non-invasive methods and improved bioavailability. According to data from the National Institutes of Health (NIH), novel delivery systems have seen increased investment in clinical trials, with over 1,500 ongoing studies in this area by early 2024.
Market Challenges
- Regulatory Complexity: The U.S. pharmaceutical industry operates under stringent regulatory guidelines, especially from the FDA, leading to a lengthy and costly approval process for new excipients. In 2023, the average time for new drug and excipient approvals was 14-18 months. The need to adhere to multiple layers of regulatory compliance, including the U.S. Pharmacopoeia standards and international harmonization, has posed significant challenges for manufacturers in 2024. The regulatory complexity also limits innovation, as companies often focus on existing, well-documented excipients rather than risk new, untested options.
- High Manufacturing Costs: The cost of producing pharmaceutical excipients has risen sharply due to inflationary pressures and supply chain disruptions. In 2023, energy prices surged by 11%, affecting manufacturing operations across the pharmaceutical supply chain. The U.S. Bureau of Labor Statistics reported that pharmaceutical manufacturing wages have increased by an average of 3.5% annually, pushing up operational costs. This increase in production expenses for excipients, combined with higher raw material costs, has created barriers to market entry for smaller manufacturers in 2024.
USA Pharmaceutical Excipients Market Future Outlook
Over the next five years, the USA Pharmaceutical Excipients market is expected to show significant growth driven by advancements in drug delivery technologies, the rise of biologics, and increasing demand for controlled-release formulations. The growth of the generics and biosimilars market, coupled with innovations in excipient functionality, will create new opportunities for both established and emerging players in the excipients market.
Future Market Opportunities
- Emerging Markets for Drug Formulation: With rising healthcare expenditures, especially in the Middle East and Africa, U.S. pharmaceutical companies are eyeing these emerging markets for drug formulations that require tailored excipients. The U.S. Census Bureau reports that exports of pharmaceuticals to emerging markets reached USD 54 billion in 2023. This creates a lucrative opportunity for excipient manufacturers to supply innovative products suited for the specific climatic and storage conditions in these regions. Moreover, increasing regulatory collaboration with emerging economies has reduced barriers for U.S. pharmaceutical exports, enabling further market penetration in 2024.
- Advancements in Nanotechnology for Drug Delivery: Nanotechnology has transformed the drug delivery landscape, particularly in targeting cancer and infectious diseases. In 2024, over 60 FDA-approved nanomedicines were being marketed, requiring specialized excipients to stabilize nanoparticles and ensure targeted drug delivery. Data from the National Nanotechnology Initiative (NNI) indicates that nanomedicine investments in the U.S. crossed USD 15 billion in 2023. This growing demand for precision medicine and targeted delivery is creating new opportunities for excipient manufacturers to develop products that improve solubility, bioavailability, and controlled release of nanomedicines.
Scope of the Report
|
By Product Type |
Polymers Sugars Minerals Gelatin Alcohols |
|
By Application |
Oral Formulations Topical Formulations Parenteral Formulations Inhalation Formulations |
|
By Function |
Binders Fillers and Diluents Lubricants Disintegrants Preservatives |
|
By Form |
Liquid Solid Semi-Solid |
|
By Region |
Northeast Midwest South West |
Products
Key Target Audience
Pharmaceutical Manufacturers
Drug Formulators
Healthcare Providers
Banks and Financial Institutes
Biotechnology Firms
Contract Research Organizations (CROs)
Regulatory Bodies (FDA, EMA)
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (U.S. Department of Health and Human Services)
Companies
Players Mentioned in the Report
BASF SE
Evonik Industries AG
Ashland Global Holdings Inc.
Roquette Frres
The Dow Chemical Company
Kerry Group Plc
Colorcon Inc.
JRS Pharma
Avantor, Inc.
DuPont de Nemours, Inc.
Table of Contents
1. USA Pharmaceutical Excipients Market Overview
1.1. Definition and Scope (Pharmaceutical Excipients Definition, Functional Categories, Market Boundaries)
1.2. Market Taxonomy (Functional Excipients, Formulation Excipients, Specialty Excipients)
1.3. Market Growth Rate (CAGR, Revenue Growth)
1.4. Market Segmentation Overview (By Product Type, Application, Function, Form, Region)
2. USA Pharmaceutical Excipients Market Size (In USD Bn)
2.1. Historical Market Size (Past Revenue and Volume)
2.2. Year-On-Year Growth Analysis (Annual Performance by Key Metrics)
2.3. Key Market Developments and Milestones (FDA Approvals, Innovation in Formulation, New Entrants)
3. USA Pharmaceutical Excipients Market Analysis
3.1. Growth Drivers
3.1.1. Increasing Demand for Generic Drugs
3.1.2. Growing Biopharmaceutical Industry
3.1.3. Rising Preference for Novel Drug Delivery Systems
3.1.4. Expansion in Functional Excipients
3.2. Restraints
3.2.1. Regulatory Complexity
3.2.2. High Manufacturing Costs
3.2.3. Raw Material Shortages
3.3. Opportunities
3.3.1. Emerging Markets for Drug Formulation
3.3.2. Advancements in Nanotechnology for Drug Delivery
3.3.3. Growth in Specialty Excipients
3.4. Trends
3.4.1. Shift Towards Biodegradable Excipients
3.4.2. Usage of Multifunctional Excipients
3.4.3. Growth of Personalized Medicine
3.5. Regulatory Impact
3.5.1. U.S. FDA Guidelines
3.5.2. cGMP Compliance
3.5.3. International Harmonization (ICH, USP)
3.6. SWOT Analysis (Strengths, Weaknesses, Opportunities, Threats)
3.7. Supply Chain Analysis (Raw Material Suppliers, Distributors, End-Use Manufacturers)
3.8. Porters Five Forces (Bargaining Power of Suppliers, Threat of Substitutes, Industry Rivalry, etc.)
4. USA Pharmaceutical Excipients Market Segmentation
4.1. By Product Type (In Value %)
4.1.1. Polymers
4.1.2. Sugars
4.1.3. Minerals
4.1.4. Gelatin
4.1.5. Alcohols
4.2. By Application (In Value %)
4.2.1. Oral Formulations
4.2.2. Topical Formulations
4.2.3. Parenteral Formulations
4.2.4. Inhalation Formulations
4.3. By Function (In Value %)
4.3.1. Binders
4.3.2. Fillers and Diluents
4.3.3. Lubricants
4.3.4. Disintegrants
4.3.5. Preservatives
4.4. By Form (In Value %)
4.4.1. Liquid
4.4.2. Solid
4.4.3. Semi-Solid
4.5. By Region (In Value %)
4.5.1. Northeast
4.5.2. Midwest
4.5.3. South
4.5.4. West
5. USA Pharmaceutical Excipients Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. BASF SE
5.1.2. Evonik Industries AG
5.1.3. Ashland Global Holdings Inc.
5.1.4. Roquette Frres
5.1.5. The Dow Chemical Company
5.1.6. Kerry Group Plc
5.1.7. Colorcon Inc.
5.1.8. JRS Pharma
5.1.9. Avantor, Inc.
5.1.10. DuPont de Nemours, Inc.
5.1.11. Archer Daniels Midland Company (ADM)
5.1.12. Croda International Plc
5.1.13. Lubrizol Corporation
5.1.14. Merck KGaA
5.1.15. Associated British Foods Plc
5.2. Cross Comparison Parameters (Revenue, Market Share, Production Capacity, Number of Employees, Product Portfolio)
5.3. Market Share Analysis (By Competitor)
5.4. Strategic Initiatives (R&D Investments, Partnerships, Product Launches)
5.5. Mergers and Acquisitions (Recent Transactions, Synergies)
5.6. Investment Analysis (Private Investments, PE and VC Funding)
5.7. Government Grants and Support (Subsidies, Research Support)
6. USA Pharmaceutical Excipients Market Regulatory Framework
6.1. cGMP Requirements (For Excipients)
6.2. FDA Regulations (Excipients Safety and Use in Formulations)
6.3. Certification Processes (ISO, ICH Guidelines)
6.4. Import/Export Compliance (Regulatory Bodies, International Standards)
7. USA Pharmaceutical Excipients Market Future Market Size (In USD Bn)
7.1. Future Market Size Projections (Revenue Growth Forecast)
7.2. Key Factors Driving Future Market Growth (Demand for Drug Formulation, Innovation in Excipients)
8. USA Pharmaceutical Excipients Future Market Segmentation
8.1. By Product Type (In Value %)
8.2. By Application (In Value %)
8.3. By Function (In Value %)
8.4. By Form (In Value %)
8.5. By Region (In Value %)
9. USA Pharmaceutical Excipients Market Analysts' Recommendations
9.1. Total Addressable Market (TAM) Analysis
9.2. Serviceable Available Market (SAM) and Serviceable Obtainable Market (SOM) Analysis
9.3. Customer Behavior Analysis (Trends in Drug Development, Buyer Personas)
9.4. White Space Opportunity Analysis
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
The initial phase involves constructing an ecosystem map encompassing all major stakeholders within the USA Pharmaceutical Excipients Market. This step is underpinned by extensive desk research, utilizing a combination of secondary and proprietary databases to gather comprehensive industry-level information. The primary objective is to identify and define the critical variables that influence market dynamics.
Step 2: Market Analysis and Construction
In this phase, we will compile and analyze historical data pertaining to the USA Pharmaceutical Excipients Market. This includes assessing market penetration, the ratio of marketplaces to service providers, and the resultant revenue generation. Furthermore, an evaluation of service quality statistics will be conducted to ensure the reliability and accuracy of the revenue estimates.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses will be developed and subsequently validated through computer-assisted telephone interviews (CATIS) with industry experts representing a diverse array of companies. These consultations will provide valuable operational and financial insights directly from industry practitioners, which will be instrumental in refining and corroborating the market data.
Step 4: Research Synthesis and Final Output
The final phase involves direct engagement with multiple pharmaceutical excipient manufacturers to acquire detailed insights into product segments, sales performance, consumer preferences, and other pertinent factors. This interaction will serve to verify and complement the statistics derived from the bottom-up approach, thereby ensuring a comprehensive, accurate, and validated analysis of the USA Pharmaceutical Excipients market.
Frequently Asked Questions
01. How big is the USA Pharmaceutical Excipients Market?
The USA Pharmaceutical Excipients Market is valued at USD 3.7 billion and continues to grow, driven by increased pharmaceutical production, expanding generics market, and innovations in excipient functionality.
02. What are the challenges in the USA Pharmaceutical Excipients Market?
Challenges in the USA Pharmaceutical Excipients Market include stringent regulatory compliance, rising raw material costs, and the complexities involved in developing multifunctional excipients that meet diverse pharmaceutical needs.
03. Who are the major players in the USA Pharmaceutical Excipients Market?
Key players in the USA Pharmaceutical Excipients Market include BASF SE, Evonik Industries AG, Ashland Global Holdings Inc., Roquette Frres, and The Dow Chemical Company, all of whom lead the market due to their strong R&D capabilities and diverse excipient portfolios.
04. What are the growth drivers of the USA Pharmaceutical Excipients Market?
Growth drivers in the USA Pharmaceutical Excipients Market include the increasing demand for pharmaceutical formulations, the expansion of biologics, and advancements in drug delivery systems, which necessitate innovative excipient solutions.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















