
USA Regenerative Medicine Market Outlook to 2030
Region:North America
Author(s):Yogita Sahu
Product Code:KROD2858
October 2024
89
About the Report
USA Regenerative Medicine Market Overview
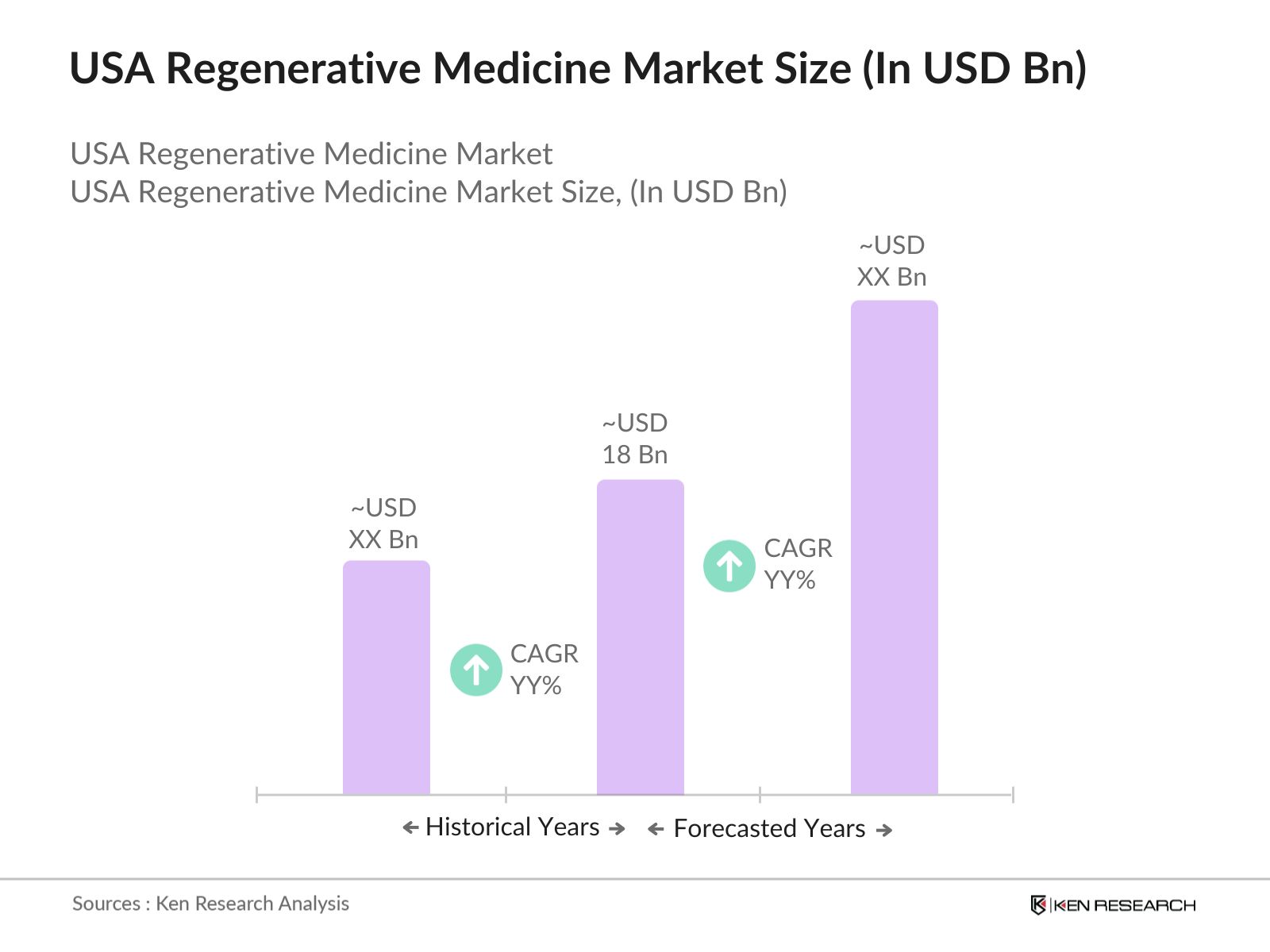
- The USA regenerative medicine market is valued at USD 18 billion, based on a five-year historical analysis. This market is driven by significant advancements in gene therapy, cell therapy, and tissue engineering technologies, along with the rising prevalence of chronic diseases and age-related conditions.
- The USA leads the global regenerative medicine market, with cities such as Boston, San Francisco, and New York acting as innovation hubs. These cities dominate due to their strong research infrastructure, access to funding, and the presence of leading academic institutions and biotech companies.
- The NIH's Regenerative Medicine Program (RMP) accelerated breakthroughs in stem cell-based therapies by developing clinical-grade induced pluripotent stem cell (iPSC) lines. By 2024, the program had generated multiple research-grade iPSC lines and initiated clinical trials for age-related macular degeneration treatments.
USA Regenerative Medicine Market Segmentation
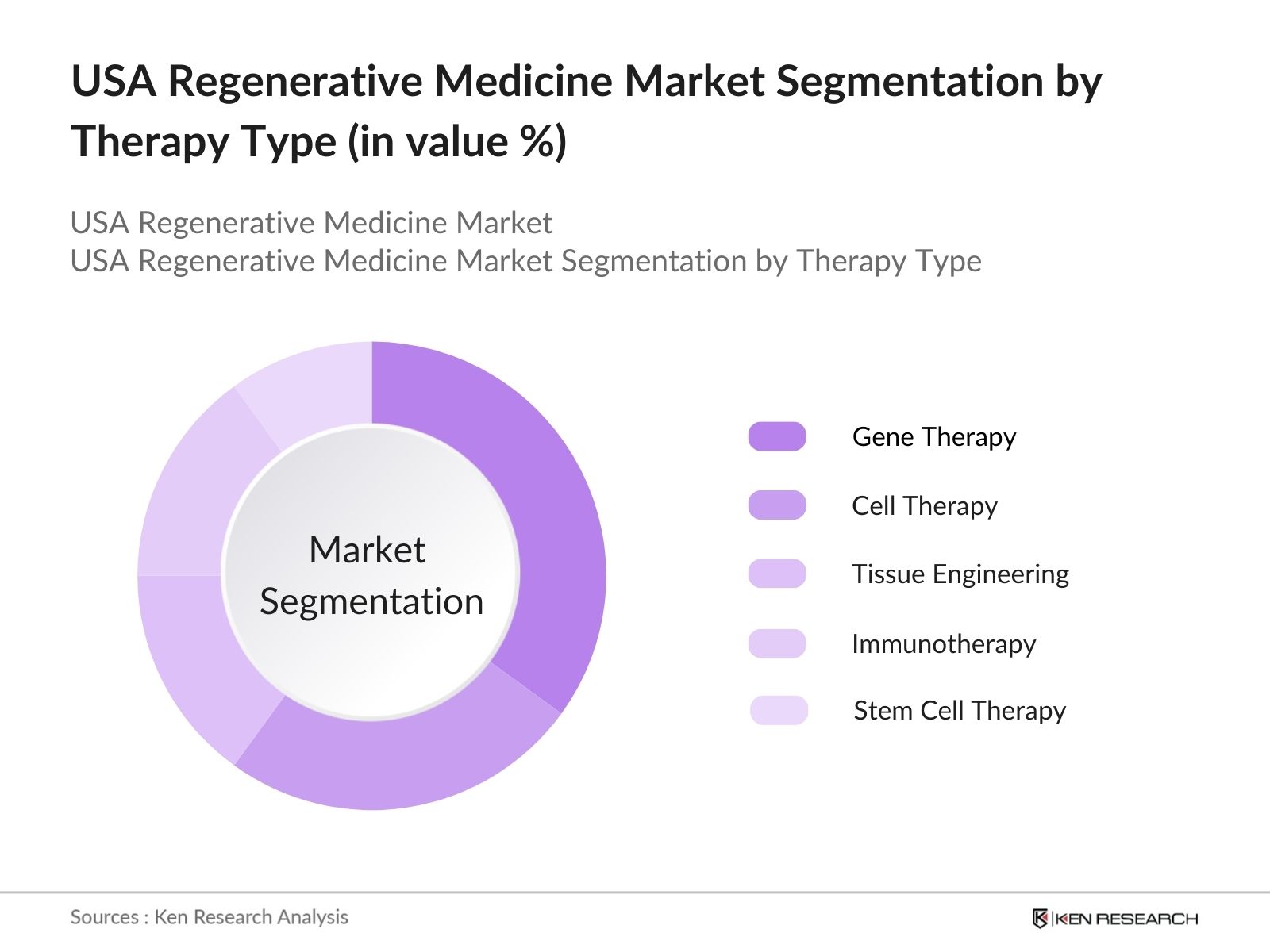
- By Therapy Type: The market is segmented by therapy type into gene therapy, cell therapy, tissue engineering, immunotherapy, and stem cell therapy. Cell therapy holds the dominant market share due to its increasing use in treating chronic diseases and the availability of a growing number of FDA-approved cell-based treatments.
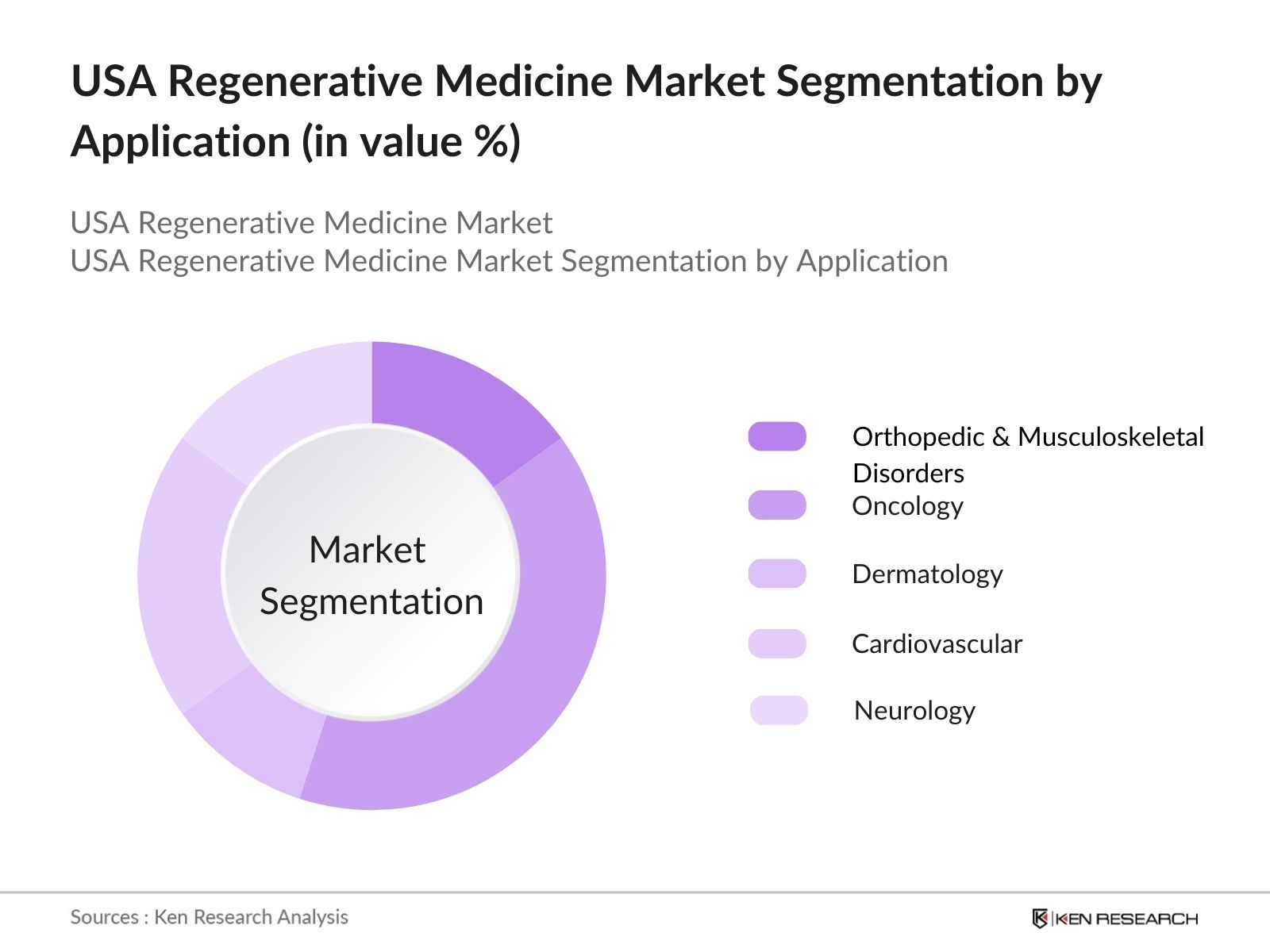
- By Application: The market is segmented by application into orthopedic & musculoskeletal disorders, oncology, dermatology, cardiovascular, and neurology. The oncology segment dominates the market due to the rising incidence of cancer and the development of regenerative therapies, such as CAR-T cell treatments and gene therapies, which offer new avenues for cancer treatment.
USA Regenerative Medicine Market Competitive Landscape
The market is dominated by a few major players, with contributions from global biotech and pharmaceutical companies. These companies are heavily involved in the development of advanced therapies, including gene editing, stem cell-based treatments, and tissue engineering solutions.
|
Company Name |
Establishment Year |
Headquarters |
No. of Patents |
Research Facilities |
R&D Investment (USD Bn) |
Strategic Partnerships |
FDA Approvals |
Clinical Trials Phases |
Product Portfolio |
|
Novartis AG |
1996 |
Basel, Switzerland |
|||||||
|
Bristol-Myers Squibb |
1887 |
New York, USA |
|||||||
|
Gilead Sciences |
1987 |
Foster City, USA |
|||||||
|
Kite Pharma |
2009 |
Santa Monica, USA |
|||||||
|
Spark Therapeutics |
2013 |
Philadelphia, USA |
USA Regenerative Medicine Market Analysis
Market Growth Drivers
- Increased Prevalence of Chronic Diseases: The increasing prevalence of chronic diseases, such as diabetes and cardiovascular conditions, is driving the demand for regenerative medicine in the USA. As of 2024, over 37 million Americans suffer from diabetes, while approximately 34 million have heart disease, according to the Centers for Disease Control and Prevention (CDC). These conditions require long-term treatments, and regenerative therapies such as stem cell and gene therapy are becoming key options for addressing these diseases.
- Aging Population: The U.S. is experiencing a rapidly aging population, which is expected to drive demand for regenerative treatments. According to U.S. Census Bureau data, over 54 million Americans were aged 65 or older in 2024, and this number is projected to reach 80 million by 2040. As the elderly are more prone to degenerative diseases such as osteoarthritis and Alzheimer's, regenerative treatments like cartilage regeneration and stem cell therapies are poised to play a critical role in addressing age-related conditions.
- Favorable Regulatory Environment: The U.S. Food and Drug Administration (FDA) has developed streamlined approval processes for regenerative medicines through the Regenerative Medicine Advanced Therapy (RMAT) designation. As of 2024, more than 70 RMAT designations had been granted to developers of regenerative treatments, fast-tracking these products through the regulatory process. This favorable regulatory environment is expected to drive the introduction of new regenerative therapies, encouraging investment in research and development while maintaining rigorous safety standards.
Market Restraints
- High Development and Treatment Costs: The cost of developing regenerative therapies remains prohibitively high, with the average cost for clinical trials in regenerative medicine surpassing $100 million by 2024. These costs significantly delay the commercialization of treatments and limit patient access. Additionally, stem cell therapies can cost anywhere between $10,000 and $50,000 per patient, making them inaccessible for many uninsured or underinsured individuals.
- Limited Skilled Workforce: The shortage of professionals skilled in regenerative medicine technologies, including tissue engineering and gene editing, is a critical barrier to market growth. In 2023, the U.S. biomedical engineering workforce reported a 20% deficit in the number of professionals needed to meet the growing demand for regenerative medicine expertise.
USA Regenerative Medicine Market Future Outlook
Over the next five years, the market is expected to show growth driven by advances in gene and cell therapies, an aging population, and increased investment from both public and private sectors. Continuous government support, in the form of funding and favorable regulatory pathways, will further encourage the development of new treatments.
Future Market Opportunities
- Increased Adoption of CRISPR for Gene Editing: Over the next five years, CRISPR technology is expected to become a key tool in developing personalized regenerative treatments. By 2029, gene editing therapies for genetic diseases such as cystic fibrosis and sickle cell anemia will likely see widespread clinical use.
- Widespread Adoption of Bioprinting: Over the next five years, the U.S. regenerative medicine market will witness the widespread adoption of bioprinting technology for tissue and organ regeneration. By 2029, bioprinting is expected to become a core technique in creating custom tissues for personalized medical treatments.
Scope of the Report
|
Therapy Type |
Gene Therapy Cell Therapy Tissue Engineering Immunotherapy Stem Cell Therapy |
|
Application |
Orthopedic & Musculoskeletal Disorders Oncology Dermatology Cardiovascular Neurology |
|
Source |
Autologous Allogeneic Xenogeneic |
|
End-User |
Hospitals Specialty Clinics Research Institutes Ambulatory Care Centers |
|
Region |
North East West South |
Products
Key Target Audience Organizations and Entities Who Can Benefit by Subscribing This Report:
Pharmaceutical Companies
Biotechnology Firms
Hospitals and Healthcare Providers
Government and Regulatory Bodies (FDA, NIH)
Research Institutes and Universities
Venture Capital and Investment Firms
Private Equity Firms
Companies
Players Mentioned in the Report:
Novartis AG
Bristol-Myers Squibb
Gilead Sciences
Kite Pharma
Spark Therapeutics
Organovo Holdings
Bluebird Bio
Astellas Pharma
Sangamo Therapeutics
Fate Therapeutics
Intellia Therapeutics
Editas Medicine
Regenxbio
CRISPR Therapeutics
Vericel Corporation
Table of Contents
USA Regenerative Medicine Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
USA Regenerative Medicine Market Size (In USD Bn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
USA Regenerative Medicine Market Analysis
3.1. Growth Drivers
3.1.1. Technological Advancements in Gene Therapy
3.1.2. Increasing Clinical Trials for Stem Cell Therapies
3.1.3. Rising Adoption of Tissue Engineering
3.1.4. Expanding Geriatric Population and Associated Conditions
3.2. Market Challenges
3.2.1. High R&D Costs (Regenerative Medicine R&D Spend, USD Bn)
3.2.2. Regulatory Hurdles (FDA Approvals and Review Timeframes)
3.2.3. Limited Commercialization Success Rates
3.2.4. Patient-Specific Therapy Customization
3.3. Opportunities
3.3.1. Expanding Applications of CRISPR and Gene Editing
3.3.2. Increased Government and Private Funding
3.3.3. Growing Demand for Cell-Based Immunotherapy (CAR-T, TCR Therapies)
3.3.4. Collaborations with Biotech Startups
3.4. Trends
3.4.1. Shift Toward Allogeneic Therapies (Market Shift towards Off-the-Shelf Therapies)
3.4.2. 3D Bioprinting for Organ Regeneration
3.4.3. Advancements in Biomaterial Technology
3.4.4. Use of AI in Regenerative Medicine R&D
3.5. Government Regulation
3.5.1. FDA’s RMAT Designation (Regenerative Medicine Advanced Therapy Designation)
3.5.2. Cell Therapy Guidelines and Regulatory Pathways
3.5.3. Accelerated Approvals and Fast-Track Designations
3.5.4. Ethical Considerations in Genetic Engineering
3.6. SWOT Analysis
3.7. Stakeholder Ecosystem (Regenerative Medicine Stakeholders: Researchers, Clinicians, Investors, Patients)
3.8. Porter’s Five Forces Analysis (Competitive Landscape, Market Entry Barriers, Buyer Power)
3.9. Competition Ecosystem
USA Regenerative Medicine Market Segmentation
4.1. By Therapy Type (In Value %)
4.1.1. Gene Therapy
4.1.2. Cell Therapy
4.1.3. Tissue Engineering
4.1.4. Immunotherapy
4.1.5. Stem Cell Therapy
4.2. By Application (In Value %)
4.2.1. Orthopedic & Musculoskeletal Disorders
4.2.2. Oncology
4.2.3. Dermatology
4.2.4. Cardiovascular
4.2.5. Neurology
4.3. By Source (In Value %)
4.3.1. Autologous
4.3.2. Allogeneic
4.3.3. Xenogeneic
4.4. By End-User (In Value %)
4.4.1. Hospitals
4.4.2. Specialty Clinics
4.4.3. Research Institutes
4.4.4. Ambulatory Care Centers
4.5. By Region (In Value %)
4.5.1. North-East
4.5.2. Mid-West
4.5.3. South
4.5.4. West
USA Regenerative Medicine Market Competitive Analysis
5.1 Detailed Profiles of Major Companies
5.1.1. Novartis AG
5.1.2. Bristol-Myers Squibb
5.1.3. Gilead Sciences
5.1.4. Sangamo Therapeutics
5.1.5. Kite Pharma
5.1.6. Organovo Holdings
5.1.7. Bluebird Bio
5.1.8. Astellas Pharma
5.1.9. Spark Therapeutics
5.1.10. Fate Therapeutics
5.1.11. Intellia Therapeutics
5.1.12. Editas Medicine
5.1.13. Regenxbio
5.1.14. CRISPR Therapeutics
5.1.15. Vericel Corporation
5.2 Cross Comparison Parameters (Number of Patents, Research Facilities, R&D Investment, Strategic Partnerships, Product Portfolio, FDA Approvals, Clinical Trial Phases, Geographic Footprint)
5.3. Market Share Analysis
5.4. Strategic Initiatives (Partnerships, Collaborations, New Launches)
5.5. Mergers and Acquisitions
5.6. Investment Analysis (Venture Capital Funding, Private Equity Investment)
5.7. Government Grants and Funding Opportunities
USA Regenerative Medicine Market Regulatory Framework
6.1. Compliance with FDA Regulations (IND Filings, BLA Submissions)
6.2. Ethical and Legal Framework for Gene Editing
6.3. Certification and Licensing Requirements
USA Regenerative Medicine Future Market Size (In USD Bn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
USA Regenerative Medicine Future Market Segmentation
8.1. By Therapy Type (in value %)
8.2. By Application (in value %)
8.3. By Source (in value %)
8.4. By End-User (in value %)
8.5. By Region (in value %)
USA Regenerative Medicine Market Analysts’ Recommendations
9.1. TAM/SAM/SOM Analysis
9.2. Competitive Strategy Recommendations
9.3. White Space Opportunity Analysis
9.4. Marketing and Product Innovation Recommendations
Disclaimer
Contact Us
Research Methodology
Step 1: Identification of Key Variables
The initial phase involves constructing an ecosystem map encompassing all major stakeholders within the USA Regenerative Medicine Market. This step is underpinned by extensive desk research, utilizing a combination of secondary and proprietary databases to gather comprehensive industry-level information. The primary objective is to identify and define the critical variables that influence market dynamics.
Step 2: Market Analysis and Construction
In this phase, we compile and analyze historical data pertaining to the USA Regenerative Medicine Market. This includes assessing market penetration, the ratio of marketplaces to service providers, and the resultant revenue generation. Furthermore, an evaluation of service quality statistics is conducted to ensure the reliability and accuracy of the revenue estimates.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are developed and subsequently validated through computer-assisted telephone interviews (CATIS) with industry experts representing a diverse array of companies. These consultations provide valuable operational and financial insights directly from industry practitioners, which are instrumental in refining and corroborating the market data.
Step 4: Research Synthesis and Final Output
The final phase involves direct engagement with multiple regenerative medicine manufacturers to acquire detailed insights into product segments, sales performance, consumer preferences, and other pertinent factors. This interaction serves to verify and complement the statistics derived from the bottom-up approach, thereby ensuring a comprehensive, accurate, and validated analysis of the USA Regenerative Medicine market.
Frequently Asked Questions
01. How big is the USA Regenerative Medicine Market?
The USA regenerative medicine market is valued at USD 18 billion, driven by advancements in gene therapy, cell therapy, and tissue engineering technologies.
02. What are the challenges in the USA Regenerative Medicine Market?
Challenges in the USA regenerative medicine market include high R&D costs, complex regulatory pathways, and the limited commercialization success of new therapies. Furthermore, patient-specific therapies require significant customization, adding to costs.
03. Who are the major players in the USA Regenerative Medicine Market?
Key players in the USA regenerative medicine market include Novartis AG, Bristol-Myers Squibb, Gilead Sciences, Kite Pharma, and Spark Therapeutics, which dominate through their extensive R&D capabilities and regulatory expertise.
04. What are the growth drivers of the USA Regenerative Medicine Market?
The USA regenerative medicine market is propelled by advancements in gene editing technologies, rising demand for personalized medicine, and increased investments from both the government and private sectors.
05. What are the future opportunities in the USA Regenerative Medicine Market?
Future opportunities in the USA regenerative medicine market lie in the expansion of gene editing, stem cell therapies, and the application of 3D bioprinting technologies to create functional tissue and organ replacements.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















