
USA Tardive Dyskinesia Therapeutics Market Outlook to 2030
Region:North America
Author(s):Mukul
Product Code:KROD9289
October 2024
94
About the Report
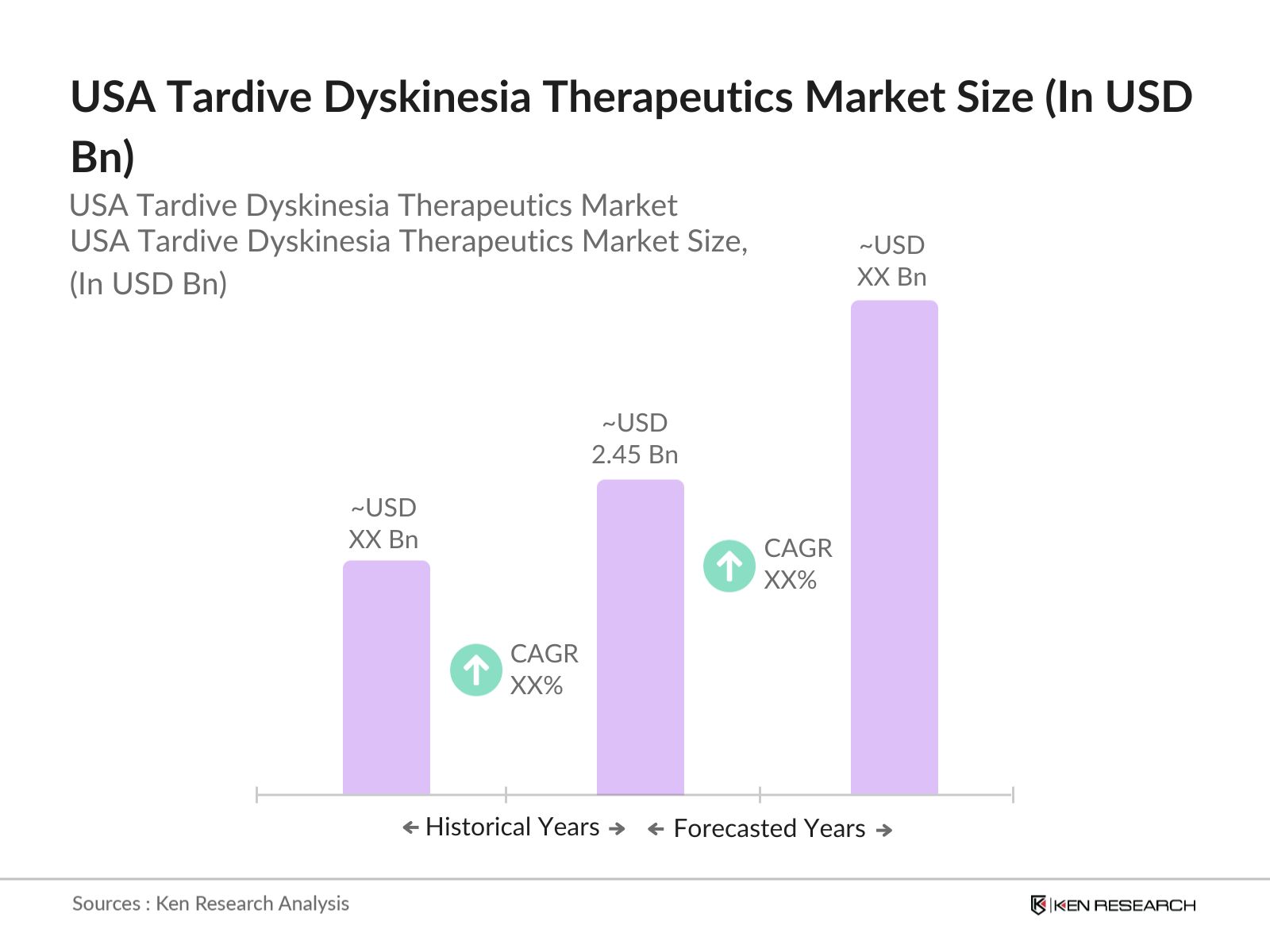
USA Tardive Dyskinesia Therapeutics Market Overview
- The USA Tardive Dyskinesia (TD) Therapeutics market is valued at USD 2.45 billion, driven by the increasing prevalence of TD due to the rise in antipsychotic drug usage for treating schizophrenia and bipolar disorders. Growth is supported by significant advancements in treatment options, such as VMAT2 inhibitors, which have shown efficacy in reducing symptoms. The presence of favorable reimbursement policies and strong regulatory support for orphan drugs further boosts the market.
- Geographically, the USA dominates the TD therapeutics market due to a combination of a large population base receiving antipsychotic treatments and a highly developed healthcare infrastructure. Major cities such as New York, Los Angeles, and Chicago are prominent, as they host a large number of psychiatric institutions and have a high concentration of patients requiring antipsychotic treatments. Additionally, the presence of key pharmaceutical players in these regions contributes to market dominance.
- Medicare reimbursement policies have expanded to include coverage for newer Tardive Dyskinesia treatments. In 2023, the U.S. Centers for Medicare and Medicaid Services (CMS) announced updates to their reimbursement schedule, covering telemedicine consultations and innovative therapeutics for neurological disorders. As of 2024, approximately 12 million Americans rely on Medicare for neurological treatments, and over $700 million was allocated to support neurological services under Medicare in 2023. This regulatory framework ensures better access to Tardive Dyskinesia treatments for senior citizens.
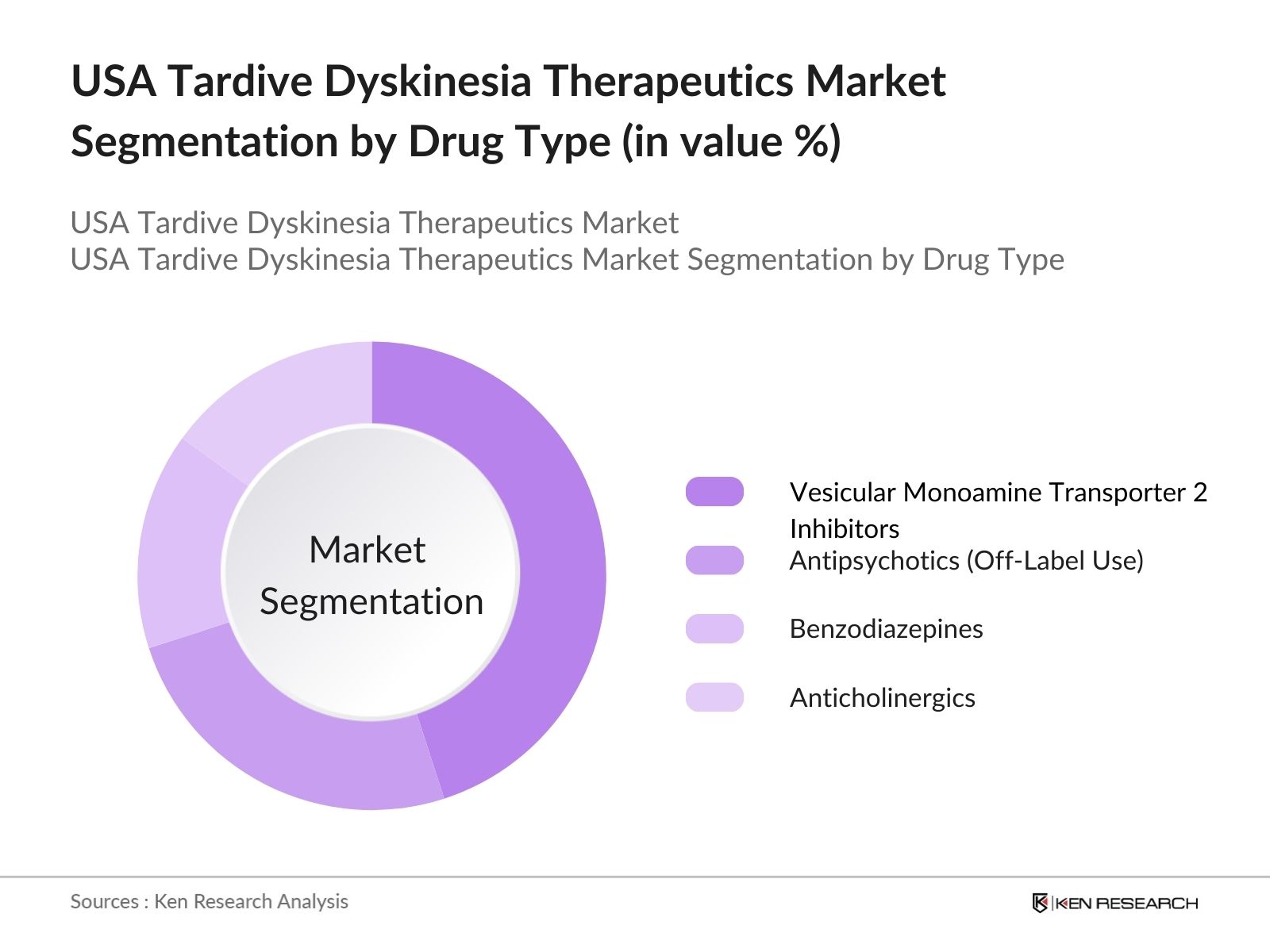
USA Tardive Dyskinesia Therapeutics Market Segmentation
By Drug Type: The USA Tardive Dyskinesia Therapeutics market is segmented by drug type into VMAT2 inhibitors, antipsychotics (off-label use), benzodiazepines, and anticholinergics. Recently, VMAT2 inhibitors have taken a dominant market share in the drug type segmentation due to their proven efficacy in managing TD symptoms with minimal side effects. Brands like Ingrezza and Austedo are leading the category, driven by increasing prescriptions from neurologists and psychiatrists across the USA, especially in urban regions with advanced medical facilities.
By Distribution Channel: The USA TD therapeutics market is also segmented by distribution channel into hospitals, retail pharmacies, and online pharmacies. Retail pharmacies have gained a dominant market share as the primary point of drug distribution, largely due to the accessibility of VMAT2 inhibitors and other drugs in major urban centers. Moreover, patients prefer retail pharmacies for their convenience, wide distribution network, and insurance coverage options available at these locations.
USA Tardive Dyskinesia Therapeutics Market Competitive Landscape
The USA Tardive Dyskinesia Therapeutics market is dominated by several key players who have established strong footholds through extensive research and product offerings. Companies like Neurocrine Biosciences and Teva Pharmaceuticals hold a significant market share due to their innovative drug pipelines, including market-leading products like Ingrezza and Austedo, respectively.
|
Company |
Establishment Year |
Headquarters |
R&D Spending (USD Bn) |
Revenue (USD Bn) |
Number of Employees |
Key Product |
FDA Approvals |
Market Expansion Strategy |
Recent Acquisition |
|
Neurocrine Biosciences |
1992 |
San Diego, CA |
|||||||
|
Teva Pharmaceuticals |
1901 |
Tel Aviv, Israel |
|||||||
|
Sunovion Pharmaceuticals |
1984 |
Marlborough, MA |
|||||||
|
AbbVie Inc. |
2013 |
North Chicago, IL |
|||||||
|
Eli Lilly and Company |
1876 |
Indianapolis, IN |
USA Tardive Dyskinesia Therapeutics Industry Analysis
Growth Drivers
- FDA Approvals for Tardive Dyskinesia Therapeutics: The growing number of FDA-approved therapeutics for Tardive Dyskinesia is a significant driver in the market. In recent years, the FDA approved medications like valbenazine and deutetrabenazine, which have shown promising results in managing Tardive Dyskinesia symptoms. The approval of these drugs has expanded treatment options for patients. As of 2023, over 500,000 patients in the U.S. are receiving treatment for neurological disorders, highlighting the rising demand for approved therapies. The U.S. pharmaceutical industry spent approximately $83 billion on R&D in 2022, demonstrating the resources directed towards developing and approving new drugs.
- Rise in Neuroleptic Drug Usage: The increasing usage of neuroleptic drugs, particularly antipsychotics, has been associated with a higher incidence of Tardive Dyskinesia. In the U.S., over 3.5 million people are currently on long-term antipsychotic treatments for conditions like schizophrenia and bipolar disorder, contributing to the prevalence of Tardive Dyskinesia. Neuroleptic drugs are a key factor in the development of this disorder, driving demand for therapeutic options. As of 2024, the U.S. government allocated over $13 billion in healthcare budgets to mental health services, indicating growing attention to neurological disorders.
- Improved Diagnostic Techniques: Advancements in diagnostic techniques have allowed for earlier and more accurate detection of Tardive Dyskinesia. Neurological assessments, imaging, and AI-powered diagnostic tools have become more sophisticated, helping clinicians identify symptoms earlier. The National Institutes of Health (NIH) reports that 50,000 new cases of Tardive Dyskinesia are diagnosed annually in the U.S. due to improved diagnostic capabilities. Investment in healthcare infrastructure reached $4.1 trillion in 2022, a portion of which is dedicated to enhancing diagnostic tools for neurological conditions.
Market Restraints
- Side Effects of Therapies: One of the major challenges in the Tardive Dyskinesia therapeutics market is the side effects associated with existing treatments. Medications like valbenazine and deutetrabenazine, while effective, can cause drowsiness, fatigue, and other neurological side effects. In 2023, approximately 30% of patients reported adverse reactions to their treatment, leading to non-compliance. This remains a significant barrier to market growth as it impacts patient outcomes. In addition, over $2.4 billion was spent on managing adverse drug reactions in the U.S. healthcare system in 2023, highlighting the economic burden of side effects.
- Lack of Awareness: Despite the increasing prevalence of Tardive Dyskinesia, awareness among both patients and healthcare providers remains limited. The National Institute of Mental Health (NIMH) estimated that nearly 50% of the population at risk for Tardive Dyskinesia remains undiagnosed or untreated due to a lack of knowledge. This gap has created a barrier in the adoption of available treatments. In 2022, the U.S. government allocated $240 million to public health campaigns, including those aimed at improving mental health and neurological disorder awareness, but gaps still persist.
USA Tardive Dyskinesia Therapeutics Market Future Outlook
The USA Tardive Dyskinesia Therapeutics market is expected to experience steady growth over the next few years, driven by increasing awareness among healthcare providers and patients about the available treatment options. Additionally, the development of newer, more effective drugs, coupled with advancements in genetic research, may provide personalized treatment plans, reducing the risk of side effects. The government's support for rare disease drug development through initiatives such as orphan drug designations further fuels optimism in this market.
Market Opportunities
- Emerging Novel Therapeutics: The development of novel therapeutics presents a significant opportunity in the Tardive Dyskinesia market. Biopharmaceutical companies are actively exploring gene therapy, biologics, and new pharmacological treatments that offer more targeted approaches to managing symptoms. Over 25 clinical trials for innovative therapies are ongoing as of 2024, with a focus on reducing the side effects and improving efficacy. The U.S. government has invested $2.3 billion in neurological research in 2023, providing a robust foundation for the development of next-generation therapies.
- Expanding Healthcare Coverage: Expanding healthcare coverage in the U.S. provides a growth opportunity for Tardive Dyskinesia treatments. With the Affordable Care Act (ACA) and Medicaid expansion, over 92% of the U.S. population had some form of health insurance in 2023, compared to 87% in 2019. This increase in coverage ensures better access to diagnostic and therapeutic services for individuals with Tardive Dyskinesia. Additionally, government healthcare spending increased to $1.9 trillion in 2022, further improving access to treatments.
Scope of the Report
|
Segment |
Sub-Segment |
|
By Drug Type |
Vesicular Monoamine Transporter 2 (VMAT2) Inhibitors |
|
Antipsychotics (Off-Label Use) |
|
|
Benzodiazepines |
|
|
Anticholinergics |
|
|
By Treatment Type |
Pharmacological Treatment |
|
Non-Pharmacological Therapies |
|
|
Supportive Care |
|
|
By Distribution Channel |
Hospitals |
|
Retail Pharmacies |
|
|
Online Pharmacies |
|
|
By Stage of Disease |
Early Stage Treatment |
|
Late Stage Treatment |
|
|
By Patient Type |
Schizophrenia Patients |
|
Bipolar Disorder Patients |
|
|
Major Depressive Disorder Patients |
Products
Key Target Audience
Pharmaceutical Companies
Healthcare Providers (Neurologists, Psychiatrists)
Government and Regulatory Bodies (FDA, NIH)
Patient Advocacy Groups
Hospitals and Specialty Clinics
Contract Research Organizations (CROs)
Investors and Venture Capitalist Firms
Biotechnology Firms
Companies
Players Mentioned in the Report
Neurocrine Biosciences
Teva Pharmaceuticals
Sunovion Pharmaceuticals
Johnson & Johnson
Pfizer Inc.
Mylan N.V.
AbbVie Inc.
Novartis AG
Bristol-Myers Squibb
Glenmark Pharmaceuticals
Eli Lilly and Company
Alkermes Plc
Takeda Pharmaceutical Company Limited
Sanofi S.A.
Merck & Co.
Table of Contents
1. USA Tardive Dyskinesia Therapeutics Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Disease Overview and Classification
1.4. Incidence and Prevalence of Tardive Dyskinesia
1.5. Diagnosis and Treatment Pathways
1.6. Market Growth Rate (FDA Approvals, Clinical Trials Progress)
2. USA Tardive Dyskinesia Therapeutics Market Size (In USD Bn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Market Value Chain
2.4. Key Market Developments and Milestones
3. USA Tardive Dyskinesia Therapeutics Market Analysis
3.1. Growth Drivers (FDA Approvals, Rise in Neuroleptic Drug Usage, Improved Diagnostic Techniques)
3.2. Market Challenges (Side Effects of Therapies, Lack of Awareness, High Treatment Costs)
3.3. Opportunities (Emerging Novel Therapeutics, Expanding Healthcare Coverage, Telemedicine in Neurology)
3.4. Trends (Gene Therapy, Personalized Medicine, AI in Diagnosis)
3.5. Government Regulation (FDA Guidelines, Orphan Drug Designation, Medicare Reimbursement Policies)
3.6. SWOT Analysis
3.7. Stakeholder Ecosystem (Patient Advocacy Groups, Neurology Societies)
3.8. Porters Five Forces
3.9. Competition Ecosystem (Pipeline Therapeutics, Marketed Products, Biotech Investments)
4. USA Tardive Dyskinesia Therapeutics Market Segmentation
4.1. By Drug Type (In Value %)
4.1.1. Vesicular Monoamine Transporter 2 (VMAT2) Inhibitors
4.1.2. Antipsychotics (Off-Label Use)
4.1.3. Benzodiazepines
4.1.4. Anticholinergics
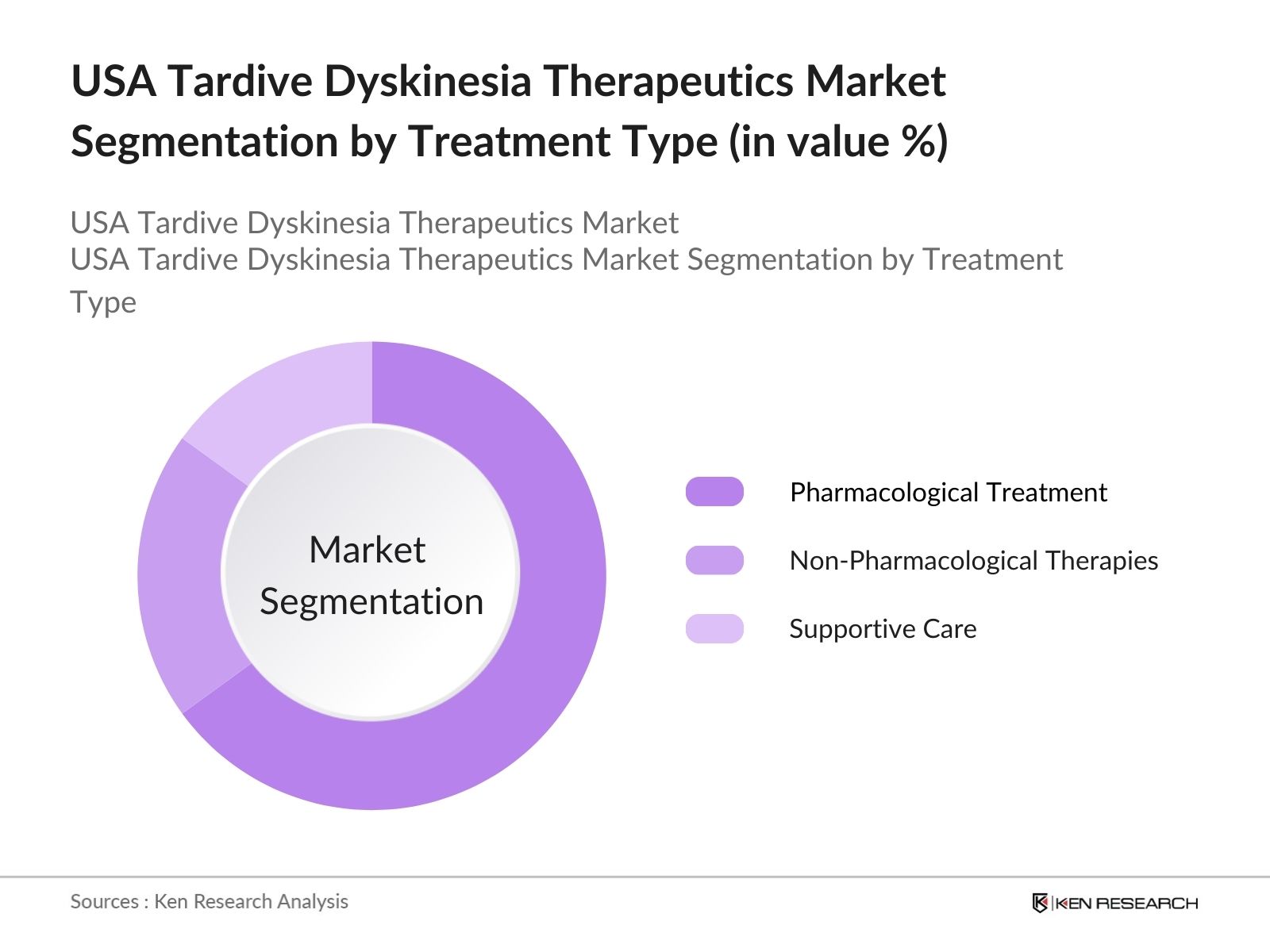
4.2. By Treatment Type (In Value %)
4.2.1. Pharmacological Treatment
4.2.2. Non-Pharmacological Therapies
4.2.3. Supportive Care
4.3. By Distribution Channel (In Value %)
4.3.1. Hospitals
4.3.2. Retail Pharmacies
4.3.3. Online Pharmacies
4.4. By Stage of Disease (In Value %)
4.4.1. Early Stage Treatment
4.4.2. Late Stage Treatment
4.5. By Patient Type (In Value %)
4.5.1. Schizophrenia Patients
4.5.2. Bipolar Disorder Patients
4.5.3. Major Depressive Disorder Patients
5. USA Tardive Dyskinesia Therapeutics Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. Neurocrine Biosciences
5.1.2. Teva Pharmaceuticals
5.1.3. Sunovion Pharmaceuticals
5.1.4. Mylan N.V.
5.1.5. Pfizer Inc.
5.1.6. Eli Lilly and Company
5.1.7. Johnson & Johnson
5.1.8. Novartis AG
5.1.9. AbbVie Inc.
5.1.10. Bristol-Myers Squibb
5.1.11. Alkermes Plc
5.1.12. Glenmark Pharmaceuticals
5.1.13. Sanofi S.A.
5.1.14. Merck & Co.
5.1.15. Takeda Pharmaceutical Company Limited
5.2. Cross Comparison Parameters (Market Share, Revenue, Product Pipeline, Strategic Partnerships, No. of Employees, R&D Spending, FDA Approvals, Recent Mergers & Acquisitions)
5.3. Market Share Analysis
5.4. Strategic Initiatives (Partnerships, Mergers & Acquisitions, Licensing Agreements)
5.5. Investment Analysis (Private Equity, Venture Capital, Government Grants)
5.6. Clinical Trial Landscape
5.7. Intellectual Property (Patent Expirations, New Patents Filed)
6. USA Tardive Dyskinesia Therapeutics Market Regulatory Framework
6.1. FDA Approval Process
6.2. Orphan Drug Designation
6.3. Reimbursement Policies
6.4. Patient Assistance Programs
6.5. Compliance Standards for Clinical Trials
7. USA Tardive Dyskinesia Therapeutics Future Market Size (In USD Bn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. USA Tardive Dyskinesia Therapeutics Future Market Segmentation
8.1. By Drug Type (In Value %)
8.2. By Treatment Type (In Value %)
8.3. By Distribution Channel (In Value %)
8.4. By Stage of Disease (In Value %)
8.5. By Patient Type (In Value %)
9. USA Tardive Dyskinesia Therapeutics Market Analysts Recommendations
9.1. TAM/SAM/SOM Analysis
9.2. Product Expansion Opportunities
9.3. Target Audience Identification
9.4. Go-To-Market Strategies
9.5. White Space Opportunity Analysis
Disclaimer Contact Us
Research Methodology
Step 1: Identification of Key Variables
The research began with a comprehensive mapping of all major stakeholders within the USA Tardive Dyskinesia Therapeutics Market. This included an extensive review of published research papers, government databases, and proprietary datasets to gather insights into key market drivers, challenges, and trends. Key variables such as drug efficacy, patient demographics, and treatment adoption rates were identified.
Step 2: Market Analysis and Construction
Using the data gathered in Step 1, a thorough analysis was conducted on historical market growth patterns, drug adoption rates, and revenue generation across different segments. This analysis allowed for an accurate construction of the market size for 2023, as well as projections for future growth. Special focus was given to emerging therapies and ongoing clinical trials.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses were validated through interviews with industry experts, including pharmaceutical executives, healthcare providers, and researchers. These consultations provided valuable insights into current trends and future market expectations, corroborating the findings of our analysis. Expert input was crucial in fine-tuning the projected growth rates and treatment adoption curves.
Step 4: Research Synthesis and Final Output
The final step involved synthesizing all the gathered data and expert insights into a cohesive and detailed report. This report was then reviewed by healthcare professionals and industry insiders to ensure its accuracy and relevance to current market conditions.
Frequently Asked Questions
01. How big is the USA Tardive Dyskinesia Therapeutics Market?
The USA Tardive Dyskinesia Therapeutics Market is valued at USD 2.45 billion, primarily driven by the rising prevalence of the disorder and the increasing use of VMAT2 inhibitors for effective symptom management.
02. What are the growth drivers of the USA Tardive Dyskinesia Therapeutics Market?
The market is driven by the approval of novel therapies, rising awareness about the disorder, and increasing usage of antipsychotics in mental health treatment, which results in a higher incidence of TD.
03. What are the challenges in the USA Tardive Dyskinesia Therapeutics Market?
Challenges include the high cost of novel treatments, potential side effects of therapies, and a lack of awareness in certain segments of the population, particularly in rural areas.
04. Who are the major players in the USA Tardive Dyskinesia Therapeutics Market?
Major players include Neurocrine Biosciences, Teva Pharmaceuticals, Pfizer, Johnson & Johnson, and AbbVie Inc. These companies lead the market due to their innovative product offerings and strong R&D capabilities.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















