About the Report
Base Year 2024Japan Diabetes Drugs Market Overview
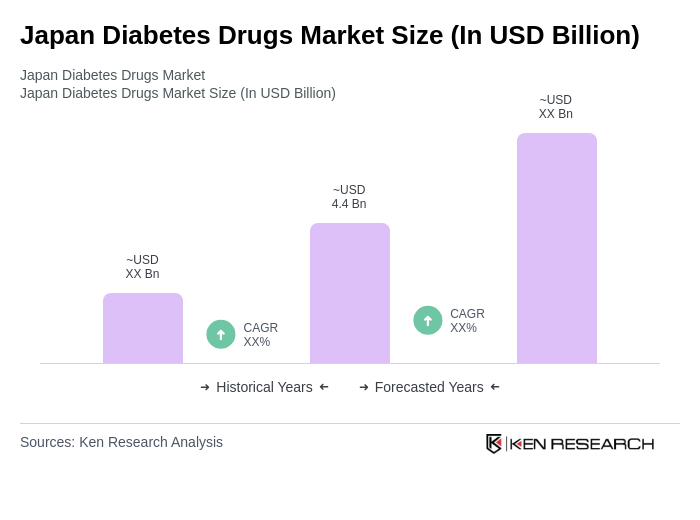
- The Japan Diabetes Drugs Market is valued at USD 4.4 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of diabetes, advances in drug formulations such as GLP-1 receptor agonists, and heightened awareness of diabetes management among the population. Rising healthcare expenditure, the introduction of innovative therapies, and strategic alliances between pharmaceutical companies have further accelerated market expansion. For example, recent collaborations have made next-generation drugs like tirzepatide and TWYMEEG more widely available, improving treatment options for patients .
- Key cities such as Tokyo, Osaka, and Yokohama continue to dominate the market due to their high population density, advanced healthcare infrastructure, and the presence of leading pharmaceutical companies. These urban centers also report a higher incidence of diabetes, driving demand for effective treatment options and comprehensive healthcare services .
- In 2023, the Japanese government strengthened regulations to enhance accessibility of diabetes medications by mandating their inclusion in the national health insurance system. This ensures that patients have affordable access to essential treatments, supporting improved compliance and overall health outcomes. The policy has contributed to increased uptake of both traditional and novel diabetes drugs .
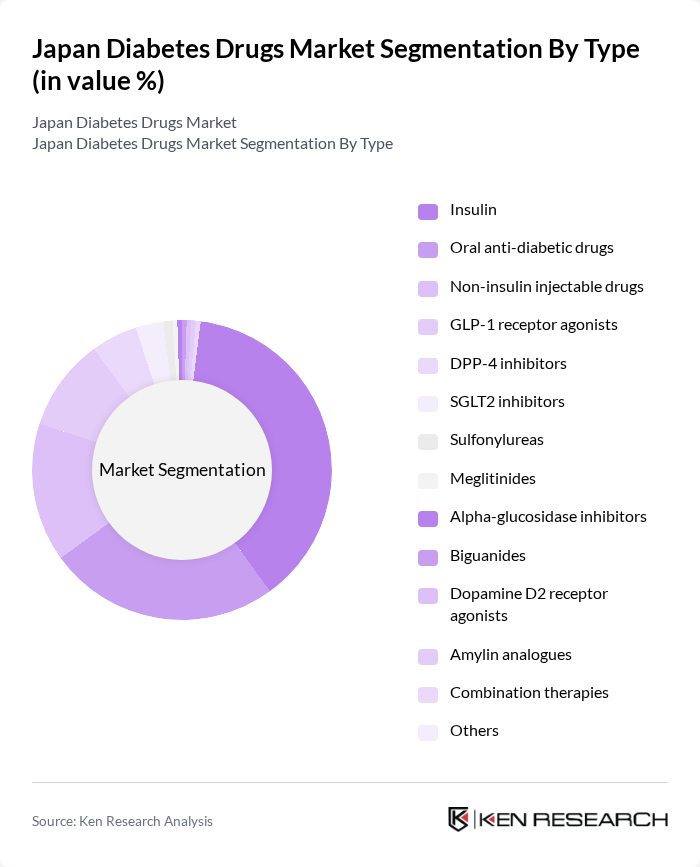
Japan Diabetes Drugs Market Segmentation
By Type:The market is segmented into various types of diabetes drugs, including insulin, oral anti-diabetic drugs, and non-insulin injectable drugs. Insulin remains a dominant segment due to its critical role in managing both Type 1 and Type 2 diabetes. The adoption of advanced insulin delivery systems and the rising prevalence of diabetes continue to drive sustained demand for insulin products. Notably, GLP-1 receptor agonists have emerged as the fastest-growing segment, reflecting a shift toward novel therapies for improved glycemic control and weight management .
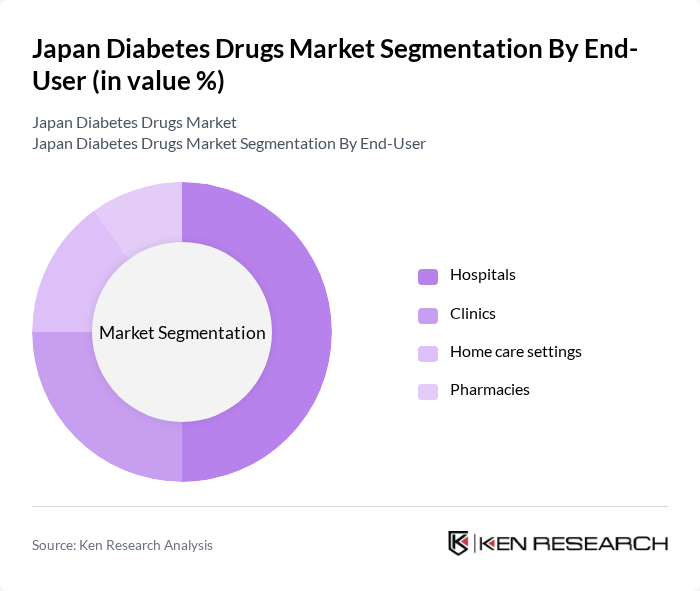
By End-User:The end-user segmentation includes hospitals, clinics, home care settings, and pharmacies. Hospitals are the leading end-user segment, primarily due to their capacity to provide comprehensive diabetes management services and access to advanced treatment options. The increasing number of diabetes patients requiring hospitalization for complications, along with the expansion of specialty diabetes centers, further drives this segment's growth. Clinics and home care settings are also expanding as outpatient management and remote monitoring become more prevalent .
Japan Diabetes Drugs Market Competitive Landscape
The Japan Diabetes Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Sanofi S.A., Novo Nordisk A/S, Eli Lilly and Company, AstraZeneca PLC, Boehringer Ingelheim GmbH, Merck & Co., Inc., Bayer AG, Johnson & Johnson, Bristol-Myers Squibb Company, Daiichi Sankyo Company, Limited, Otsuka Pharmaceutical Co., Ltd., Astellas Pharma Inc., Sumitomo Pharma Co., Ltd., Chugai Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Kyowa Kirin Co., Ltd., Pfizer Inc., Novartis AG contribute to innovation, geographic expansion, and service delivery in this space.
Japan Diabetes Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Diabetes:Japan has seen a significant rise in diabetes cases, with approximately 7.8 million adults diagnosed in future, according to the Ministry of Health, Labour and Welfare. This increase is attributed to an aging population and lifestyle changes, leading to a projected annual healthcare cost of ¥1.3 trillion (around $12 billion) for diabetes management. The growing patient base drives demand for diabetes medications, creating a robust market environment for pharmaceutical companies.
- Advancements in Drug Formulations:The Japanese pharmaceutical industry is witnessing rapid advancements in diabetes drug formulations, with over 25 new medications introduced in the last five years. Innovations such as GLP-1 receptor agonists and SGLT2 inhibitors have shown improved efficacy and safety profiles. The total expenditure on diabetes drugs in Japan reached ¥650 billion (approximately $6 billion) in future, reflecting the market's response to these advancements and the increasing demand for effective treatments.
- Rising Healthcare Expenditure:Japan's healthcare expenditure is projected to reach ¥55 trillion (around $500 billion) in future, driven by government initiatives to improve healthcare access and quality. This increase in spending is expected to enhance the availability of diabetes medications and support innovative treatment options. The government’s commitment to healthcare funding is crucial for addressing the diabetes epidemic, thereby fostering a favorable market environment for drug manufacturers and healthcare providers.
Market Challenges
- High Cost of Diabetes Medications:The cost of diabetes medications in Japan remains a significant barrier, with average annual expenses per patient exceeding ¥110,000 (approximately $1,010). This high cost can limit access for many patients, particularly those in lower-income brackets. As a result, healthcare providers face challenges in ensuring that patients receive necessary treatments, which can hinder overall market growth and patient outcomes in diabetes management.
- Stringent Regulatory Requirements:The Japanese pharmaceutical market is characterized by rigorous regulatory standards, with the Pharmaceuticals and Medical Devices Agency (PMDA) overseeing drug approvals. The average approval time for new diabetes medications can exceed 20 months, which delays market entry and increases development costs. These stringent regulations can deter smaller companies from entering the market, limiting competition and innovation in diabetes treatment options.
Japan Diabetes Drugs Market Future Outlook
The Japan diabetes drugs market is poised for significant evolution, driven by technological integration and a focus on preventive healthcare. The rise of telemedicine and digital health solutions is expected to enhance patient engagement and adherence to treatment plans. Additionally, the development of personalized medicine tailored to individual patient profiles will likely improve treatment outcomes. As awareness and education on diabetes management increase, the market will continue to adapt, fostering innovation and improving patient care in the coming years.
Market Opportunities
- Growth in Telemedicine and Digital Health:The telemedicine sector in Japan is projected to grow to ¥1.2 trillion (approximately $11 billion) in future. This growth presents opportunities for diabetes management solutions that leverage technology to improve patient monitoring and access to care, ultimately enhancing treatment adherence and outcomes.
- Development of Personalized Medicine:The personalized medicine market in Japan is expected to reach ¥350 billion (around $3.2 billion) in future. This trend offers significant opportunities for diabetes drug manufacturers to create tailored therapies that address the unique needs of individual patients, improving efficacy and patient satisfaction in diabetes management.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Insulin Oral anti-diabetic drugs Non-insulin injectable drugs GLP-1 receptor agonists DPP-4 inhibitors SGLT2 inhibitors Sulfonylureas Meglitinides Alpha-glucosidase inhibitors Biguanides Dopamine D2 receptor agonists Amylin analogues Combination therapies Others |
| By End-User | Hospitals Clinics Home care settings Pharmacies |
| By Distribution Channel | Direct sales Retail pharmacies Online pharmacies Wholesalers |
| By Patient Type | Type 1 diabetes Type 2 diabetes Gestational diabetes |
| By Age Group | Pediatric Adult Elderly |
| By Treatment Stage | Newly diagnosed Ongoing management Complications management |
| By Region | Kanto Kansai Chubu Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Labour and Welfare; Pharmaceuticals and Medical Devices Agency)
Pharmaceutical Manufacturers
Healthcare Providers and Hospitals
Insurance Companies and Payers
Biotechnology Firms
Market Access and Pricing Strategy Firms
Patient Advocacy Groups
Players Mentioned in the Report:
Takeda Pharmaceutical Company Limited
Sanofi S.A.
Novo Nordisk A/S
Eli Lilly and Company
AstraZeneca PLC
Boehringer Ingelheim GmbH
Merck & Co., Inc.
Bayer AG
Johnson & Johnson
Bristol-Myers Squibb Company
Daiichi Sankyo Company, Limited
Otsuka Pharmaceutical Co., Ltd.
Astellas Pharma Inc.
Sumitomo Pharma Co., Ltd.
Chugai Pharmaceutical Co., Ltd.
Mitsubishi Tanabe Pharma Corporation
Kyowa Kirin Co., Ltd.
Pfizer Inc.
Novartis AG
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan Diabetes Drugs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan Diabetes Drugs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan Diabetes Drugs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of diabetes
3.1.2 Advancements in drug formulations
3.1.3 Rising healthcare expenditure
3.1.4 Government initiatives for diabetes management
3.2 Market Challenges
3.2.1 High cost of diabetes medications
3.2.2 Stringent regulatory requirements
3.2.3 Competition from generic drugs
3.2.4 Limited patient access in rural areas
3.3 Market Opportunities
3.3.1 Growth in telemedicine and digital health
3.3.2 Development of personalized medicine
3.3.3 Expansion of distribution channels
3.3.4 Increasing awareness and education on diabetes
3.4 Market Trends
3.4.1 Shift towards injectable diabetes medications
3.4.2 Integration of technology in diabetes management
3.4.3 Focus on preventive healthcare
3.4.4 Rise of biosimilars in the market
3.5 Government Regulation
3.5.1 Price control regulations
3.5.2 Approval processes for new drugs
3.5.3 Reimbursement policies for diabetes medications
3.5.4 Guidelines for diabetes management
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan Diabetes Drugs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan Diabetes Drugs Market Segmentation
8.1 By Type
8.1.1 Insulin
8.1.2 Oral anti-diabetic drugs
8.1.3 Non-insulin injectable drugs
8.1.4 GLP-1 receptor agonists
8.1.5 DPP-4 inhibitors
8.1.6 SGLT2 inhibitors
8.1.7 Sulfonylureas
8.1.8 Meglitinides
8.1.9 Alpha-glucosidase inhibitors
8.1.10 Biguanides
8.1.11 Dopamine D2 receptor agonists
8.1.12 Amylin analogues
8.1.13 Combination therapies
8.1.14 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Clinics
8.2.3 Home care settings
8.2.4 Pharmacies
8.3 By Distribution Channel
8.3.1 Direct sales
8.3.2 Retail pharmacies
8.3.3 Online pharmacies
8.3.4 Wholesalers
8.4 By Patient Type
8.4.1 Type 1 diabetes
8.4.2 Type 2 diabetes
8.4.3 Gestational diabetes
8.5 By Age Group
8.5.1 Pediatric
8.5.2 Adult
8.5.3 Elderly
8.6 By Treatment Stage
8.6.1 Newly diagnosed
8.6.2 Ongoing management
8.6.3 Complications management
8.7 By Region
8.7.1 Kanto
8.7.2 Kansai
8.7.3 Chubu
8.7.4 Others
9. Japan Diabetes Drugs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Japan diabetes drugs segment)
9.2.4 Market Share (Japan diabetes drugs market)
9.2.5 Product Portfolio Breadth (number of diabetes drugs marketed in Japan)
9.2.6 Regulatory Approval Success Rate (Japan PMDA)
9.2.7 R&D Investment as % of Segment Revenue
9.2.8 Sales per Representative (Japan diabetes drugs)
9.2.9 Distribution Coverage (number of hospitals, clinics, pharmacies reached)
9.2.10 Patient Access Programs (existence and scale)
9.2.11 Pricing Strategy (premium, value, reimbursement status)
9.2.12 Customer Retention Rate (Japan diabetes drugs)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 List of Major Companies
9.5.1 Takeda Pharmaceutical Company Limited
9.5.2 Sanofi S.A.
9.5.3 Novo Nordisk A/S
9.5.4 Eli Lilly and Company
9.5.5 AstraZeneca PLC
9.5.6 Boehringer Ingelheim GmbH
9.5.7 Merck & Co., Inc.
9.5.8 Bayer AG
9.5.9 Johnson & Johnson
9.5.10 Bristol-Myers Squibb Company
9.5.11 Daiichi Sankyo Company, Limited
9.5.12 Otsuka Pharmaceutical Co., Ltd.
9.5.13 Astellas Pharma Inc.
9.5.14 Sumitomo Pharma Co., Ltd.
9.5.15 Chugai Pharmaceutical Co., Ltd.
9.5.16 Mitsubishi Tanabe Pharma Corporation
9.5.17 Kyowa Kirin Co., Ltd.
9.5.18 Pfizer Inc.
9.5.19 Novartis AG
10. Japan Diabetes Drugs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for diabetes care
10.1.2 Procurement processes and timelines
10.1.3 Preferred suppliers and contracts
10.1.4 Evaluation criteria for drug selection
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in diabetes care facilities
10.2.2 Funding for diabetes research and development
10.2.3 Partnerships with healthcare providers
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to affordable medications
10.3.2 Availability of diabetes management resources
10.3.3 Support for lifestyle changes
10.4 User Readiness for Adoption
10.4.1 Awareness of diabetes treatment options
10.4.2 Willingness to adopt new therapies
10.4.3 Training and education needs
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of treatment outcomes
10.5.2 Cost-effectiveness of diabetes drugs
10.5.3 Opportunities for therapy expansion
11. Japan Diabetes Drugs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps and opportunities
1.2 Business model options
1.3 Value proposition development
1.4 Revenue streams identification
1.5 Cost structure analysis
1.6 Key partnerships and resources
1.7 Customer segments and relationships
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication channels
2.5 Promotional tactics
2.6 Market positioning
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Partnerships with healthcare providers
3.5 Logistics and supply chain management
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Value-based pricing models
5. Unmet Demand & Latent Needs
5.1 Category gaps in diabetes care
5.2 Consumer segments with unmet needs
5.3 Innovations in diabetes management
5.4 Future trends in diabetes treatment
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer feedback mechanisms
6.4 Community engagement initiatives
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Health outcomes improvement
7.4 Cost savings for end-users
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Training and support for stakeholders
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band strategies
9.1.3 Packaging options
9.2 Export Entry Strategy
9.2.1 Target countries analysis
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity timelines
15.2.2 Milestone tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from health organizations and pharmaceutical associations in Japan
- Review of published articles and journals focusing on diabetes treatment and drug efficacy
- Examination of government health statistics and diabetes prevalence data from the Ministry of Health, Labour and Welfare
Primary Research
- Interviews with endocrinologists and diabetes specialists to gather insights on drug preferences and patient outcomes
- Surveys with pharmacists to understand prescription trends and patient demographics
- Focus groups with diabetes patients to assess treatment satisfaction and drug accessibility
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and patient surveys
- Triangulation of data from clinical studies, market reports, and expert opinions
- Sanity checks through feedback from a panel of healthcare professionals and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total diabetes drug market size based on national healthcare expenditure and diabetes prevalence rates
- Segmentation of market by drug class, including insulin, oral hypoglycemics, and GLP-1 receptor agonists
- Incorporation of growth rates from historical data and projected trends in diabetes management
Bottom-up Modeling
- Collection of sales data from major pharmaceutical companies operating in Japan
- Estimation of market share based on prescription volume and revenue from diabetes medications
- Analysis of pricing strategies and reimbursement policies affecting drug accessibility
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating demographic shifts, healthcare policies, and technological advancements
- Scenario modeling based on potential changes in diabetes prevalence and treatment guidelines
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Endocrinologist Insights | 60 | Endocrinologists, Diabetes Specialists |
| Pharmacist Perspectives | 50 | Community Pharmacists, Hospital Pharmacists |
| Patient Experience Feedback | 100 | Diabetes Patients, Caregivers |
| Healthcare Policy Analysis | 40 | Health Economists, Policy Makers |
| Market Access Insights | 40 | Market Access Managers, Reimbursement Specialists |
Frequently Asked Questions
What is the current value of the Japan Diabetes Drugs Market?
The Japan Diabetes Drugs Market is valued at approximately USD 4.4 billion, reflecting a significant growth driven by the increasing prevalence of diabetes and advancements in drug formulations, particularly GLP-1 receptor agonists and innovative therapies.