About the Report
Base Year 2024Japan Emergency Contraceptive Pills Market Overview
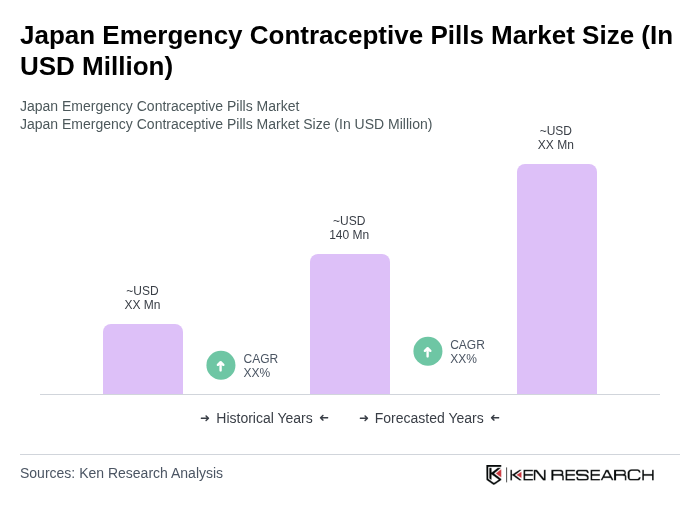
- The Japan Emergency Contraceptive Pills market is valued at USD 140 million, based on a five-year historical analysis and its share within the broader Japan contraceptive drugs market, which was valued at around USD 720 million. This growth is primarily driven by increasing awareness of reproductive health, rising demand for contraceptive options, and the growing acceptance of emergency contraceptive pills among women. Growing dual?income and single?person households, greater focus on delaying childbirth, and wider use of digital health and e?pharmacy channels are further supporting market expansion.
- Key cities such as Tokyo, Osaka, and Yokohama dominate the market due to their large populations, advanced healthcare infrastructure, and higher levels of education regarding reproductive health. The Kanto region, which includes Tokyo, represents the largest share of contraceptive drug consumption in Japan, underpinned by high purchasing power and dense pharmacy networks. These urban centers have a greater concentration of pharmacies, hospitals, clinics, and women’s health services, making emergency contraceptive pills more accessible to women in need.
- In 2023, Japan's Ministry of Health, Labour and Welfare launched a pilot program allowing over-the-counter access to emergency contraceptive pills (levonorgestrel products such as NorLevo and generics) without a prescription at selected pharmacies nationwide. This initiative, implemented under the ministry’s emergency contraceptive access framework and related ministerial notifications in 2023, requires participating pharmacies to have trained pharmacists, provide counseling, and maintain usage records, with the goal of improving timely access to ECPs and contributing to the reduction of unintended pregnancies.
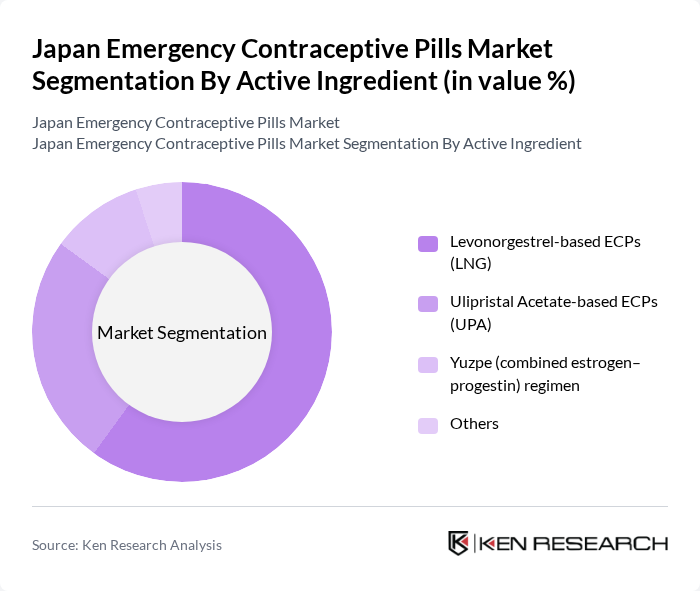
Japan Emergency Contraceptive Pills Market Segmentation
By Active Ingredient:The market is segmented into Levonorgestrel-based ECPs (LNG), Ulipristal Acetate-based ECPs (UPA), Yuzpe (combined estrogen–progestin) regimen, and Others. Levonorgestrel-based ECPs are the most widely used globally and in Japan due to their established efficacy, favorable safety profile, and broad regulatory acceptance as the standard first-line oral emergency contraceptive option. The preference for LNG is driven by its long-standing presence in the market, inclusion in national guidelines, and familiarity of healthcare providers and consumers with this option.
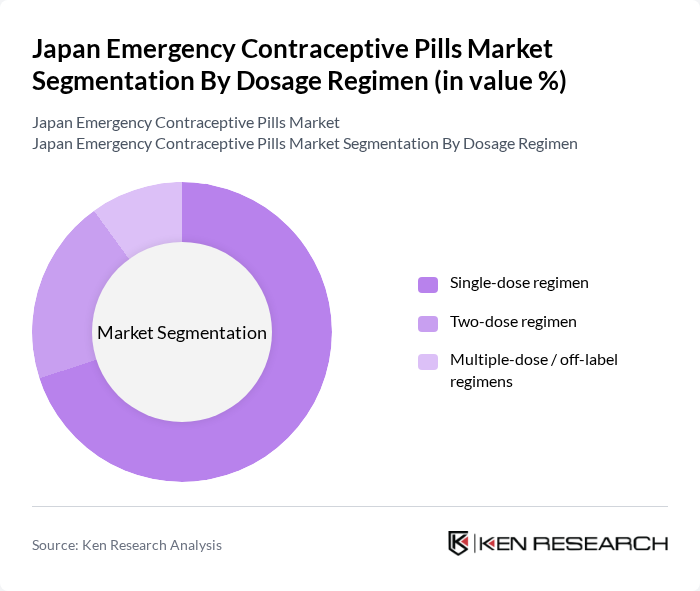
By Dosage Regimen:The market is categorized into Single-dose regimen, Two-dose regimen, and Multiple-dose / off-label regimens. The Single-dose regimen, particularly one?tablet levonorgestrel formulations, is the most popular choice among consumers due to its convenience, simpler counseling requirements, and alignment with global clinical practice recommendations for timely emergency contraception. This preference is largely influenced by the urgency associated with emergency contraception, where quick access to a single dose is critical for effectiveness.
Japan Emergency Contraceptive Pills Market Competitive Landscape
The Japan Emergency Contraceptive Pills Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Bayer AG, Pfizer Inc., HRA Pharma (Perrigo Company plc), Mylan N.V. (Viatris Inc.), Teva Pharmaceutical Industries Ltd., Merck & Co., Inc. (MSD), Gedeon Richter Plc., Aurobindo Pharma Limited, Glenmark Pharmaceuticals Ltd., Cipla Limited, Sun Pharmaceutical Industries Ltd., Sandoz (a Novartis division), Hetero Labs Limited, Alvogen contribute to innovation, geographic expansion, and service delivery in this space.
Japan Emergency Contraceptive Pills Market Industry Analysis
Growth Drivers
- Increasing Awareness of Reproductive Health:The Japanese government has reported a 30% increase in public awareness campaigns regarding reproductive health since 2020. This rise in awareness is crucial as it correlates with a growing understanding of emergency contraceptive pills (ECPs) among women aged 18-35. In future, approximately 60% of this demographic is expected to recognize ECPs as a viable option, driven by educational initiatives and social media outreach, enhancing market demand significantly.
- Government Initiatives Promoting Family Planning:In future, Japan's Ministry of Health allocated ¥5 billion (approximately $45 million) to family planning initiatives, including the promotion of emergency contraceptive pills. This funding aims to improve access and education surrounding reproductive health. The government’s commitment to reducing unintended pregnancies is expected to increase the utilization of ECPs, thereby expanding the market and supporting women's health initiatives across the nation.
- Accessibility Improvements in Urban Areas:Urban areas in Japan have seen a 25% increase in pharmacies offering emergency contraceptive pills since 2022. This improvement in accessibility is crucial, as urban populations are projected to grow by 1.2 million in future. Enhanced distribution channels, including extended pharmacy hours and online availability, are expected to facilitate easier access to ECPs, thereby driving market growth and meeting the needs of urban women effectively.
Market Challenges
- Cultural Stigma Surrounding Emergency Contraception:Despite increasing awareness, cultural stigma remains a significant barrier in Japan. A survey indicated that 40% of women still feel uncomfortable discussing emergency contraception openly. This stigma can deter women from seeking ECPs, limiting market growth. Addressing these cultural perceptions through targeted education and outreach is essential for overcoming this challenge and increasing acceptance of ECPs in society.
- Regulatory Hurdles in Product Approval:The approval process for new contraceptive products in Japan can take up to three years, significantly delaying market entry. In future, the Japan Pharmaceuticals and Medical Devices Agency (PMDA) is expected to review over 20 applications for new ECP formulations. Lengthy regulatory timelines can hinder innovation and limit the availability of diverse options for consumers, posing a challenge to market expansion and competitiveness.
Japan Emergency Contraceptive Pills Market Future Outlook
The future of the Japan emergency contraceptive pills market appears promising, driven by ongoing government support and increasing public awareness. As urbanization continues, the demand for accessible reproductive health solutions is expected to rise. Additionally, the shift towards over-the-counter availability and the integration of telemedicine services will likely enhance consumer access. These trends indicate a growing acceptance of emergency contraception, paving the way for innovative products and improved healthcare outcomes for women in Japan.
Market Opportunities
- Expansion of Online Pharmacies:The rise of e-commerce in Japan presents a significant opportunity for emergency contraceptive pills. With online pharmacy sales projected to reach ¥1 trillion (approximately $9 billion) in future, companies can leverage this trend to enhance accessibility and convenience for consumers, particularly in urban areas where demand is high.
- Collaborations with Healthcare Providers:Partnerships between pharmaceutical companies and healthcare providers can enhance education and distribution of emergency contraceptive pills. By integrating ECP education into routine healthcare services in future, initiatives are expected to increase awareness and acceptance, ultimately driving sales and improving reproductive health outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Active Ingredient | Levonorgestrel-based ECPs (LNG) Ulipristal Acetate-based ECPs (UPA) Yuzpe (combined estrogen–progestin) regimen Others |
| By Dosage Regimen | Single-dose regimen Two-dose regimen Multiple-dose / off?label regimens |
| By Access/Prescription Status | Prescription-only Pharmacy supply with pharmacist consultation Over-the-counter (OTC) / trial OTC schemes Telemedicine / e-prescription |
| By Distribution Channel | Community / retail pharmacies Hospital pharmacies Online pharmacies & e-commerce platforms Clinics, NGOs & Public Health Facilities |
| By End-User Profile | Adolescents (15–19 years) Young adults (20–29 years) Adults (30–44 years) + & peri?menopausal users |
| By Purchase Motivation / Use Case | Contraceptive failure (condom breakage, missed pills) Unprotected intercourse Sexual assault care pathways Backup to ongoing contraception |
| By Region | Kanto Kansai / Kinki Chubu (Central) Hokkaido & Tohoku Chugoku & Shikoku Kyushu–Okinawa |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Labour and Welfare)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Clinics
Industry Associations (e.g., Japan Pharmaceutical Manufacturers Association)
Health Insurance Companies
Players Mentioned in the Report:
Takeda Pharmaceutical Company Limited
Bayer AG
Pfizer Inc.
HRA Pharma (Perrigo Company plc)
Mylan N.V. (Viatris Inc.)
Teva Pharmaceutical Industries Ltd.
Merck & Co., Inc. (MSD)
Gedeon Richter Plc.
Aurobindo Pharma Limited
Glenmark Pharmaceuticals Ltd.
Cipla Limited
Sun Pharmaceutical Industries Ltd.
Sandoz (a Novartis division)
Hetero Labs Limited
Alvogen
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan Emergency Contraceptive Pills Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan Emergency Contraceptive Pills Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan Emergency Contraceptive Pills Market Analysis
3.1 Growth Drivers
3.1.1 Increasing awareness of reproductive health
3.1.3 Government initiatives promoting family planning
3.1.4 Accessibility improvements in urban areas
3.2 Market Challenges
3.2.1 Cultural stigma surrounding emergency contraception
3.2.2 Regulatory hurdles in product approval
3.2.3 Limited availability in rural regions
3.2.4 Competition from alternative contraceptive methods
3.3 Market Opportunities
3.3.1 Expansion of online pharmacies
3.3.2 Collaborations with healthcare providers
3.3.3 Increasing focus on women's health products
3.3.4 Development of new formulations
3.4 Market Trends
3.4.1 Shift towards over-the-counter availability
3.4.2 Growth in telemedicine consultations
3.4.3 Rising popularity of personalized healthcare
3.4.4 Increased marketing efforts targeting younger demographics
3.5 Government Regulation
3.5.1 Approval processes for new contraceptive products
3.5.2 Guidelines for advertising and promotion
3.5.3 Regulations on distribution channels
3.5.4 Policies supporting reproductive health education
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan Emergency Contraceptive Pills Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan Emergency Contraceptive Pills Market Segmentation
8.1 By Active Ingredient
8.1.1 Levonorgestrel-based ECPs (LNG)
8.1.2 Ulipristal Acetate-based ECPs (UPA)
8.1.3 Yuzpe (combined estrogen–progestin) regimen
8.1.4 Others
8.2 By Dosage Regimen
8.2.1 Single-dose regimen
8.2.2 Two-dose regimen
8.2.3 Multiple-dose / off?label regimens
8.3 By Access/Prescription Status
8.3.1 Prescription-only
8.3.2 Pharmacy supply with pharmacist consultation
8.3.3 Over-the-counter (OTC) / trial OTC schemes
8.3.4 Telemedicine / e-prescription
8.4 By Distribution Channel
8.4.1 Community / retail pharmacies
8.4.2 Hospital pharmacies
8.4.3 Online pharmacies & e-commerce platforms
8.4.4 Clinics, NGOs & Public Health Facilities
8.5 By End-User Profile
8.5.1 Adolescents (15–19 years)
8.5.2 Young adults (20–29 years)
8.5.3 Adults (30–44 years)
8.5.4 45+ & peri?menopausal users
8.6 By Purchase Motivation / Use Case
8.6.1 Contraceptive failure (condom breakage, missed pills)
8.6.2 Unprotected intercourse
8.6.3 Sexual assault care pathways
8.6.4 Backup to ongoing contraception
8.7 By Region
8.7.1 Kanto
8.7.2 Kansai / Kinki
8.7.3 Chubu (Central)
8.7.4 Hokkaido & Tohoku
8.7.5 Chugoku & Shikoku
8.7.6 Kyushu–Okinawa
9. Japan Emergency Contraceptive Pills Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Japan ECP Revenue (latest FY, US$ Mn)
9.2.3 Japan ECP Revenue CAGR (3–5 Year)
9.2.4 Japan ECP Market Share (%)
9.2.5 Volume Sold (Units of Packs)
9.2.6 Average Realized Price per Pack (US$)
9.2.7 Gross Margin (%) – ECP Portfolio
9.2.8 Marketing & Promotion Spend (% of ECP Sales)
9.2.9 Distribution Reach (No. of Pharmacies / Channels Served)
9.2.10 New Product / Line-Extension Launches (Last 3 Years)
9.2.11 R&D Intensity (% of Revenue, Women’s Health / ECP)
9.2.12 Regulatory / OTC Access Status by Product
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Takeda Pharmaceutical Company Limited
9.5.2 Bayer AG
9.5.3 Pfizer Inc.
9.5.4 HRA Pharma (Perrigo Company plc)
9.5.5 Mylan N.V. (Viatris Inc.)
9.5.6 Teva Pharmaceutical Industries Ltd.
9.5.7 Merck & Co., Inc. (MSD)
9.5.8 Gedeon Richter Plc.
9.5.9 Aurobindo Pharma Limited
9.5.10 Glenmark Pharmaceuticals Ltd.
9.5.11 Cipla Limited
9.5.12 Sun Pharmaceutical Industries Ltd.
9.5.13 Sandoz (a Novartis division)
9.5.14 Hetero Labs Limited
9.5.15 Alvogen
10. Japan Emergency Contraceptive Pills Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government purchasing policies
10.1.2 Budget allocation for reproductive health
10.1.3 Collaboration with NGOs
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare facilities
10.2.2 Funding for educational programs
10.2.3 Partnerships with pharmaceutical companies
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Accessibility issues
10.3.2 Affordability concerns
10.3.3 Awareness gaps
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Attitudes towards emergency contraception
10.4.2 Knowledge of available options
10.4.3 Willingness to seek medical advice
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of effectiveness
10.5.2 User feedback collection
10.5.3 Potential for product line expansion
10.5.4 Others
11. Japan Emergency Contraceptive Pills Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Operational framework design
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience definition
2.4 Communication channels selection
2.5 Campaign planning
2.6 Performance metrics establishment
2.7 Feedback mechanisms
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 E-commerce integration
3.4 Logistics optimization
3.5 Inventory management
3.6 Partnership with healthcare providers
3.7 Distribution channel evaluation
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing comparison
4.4 Consumer willingness to pay
4.5 Discount strategies
4.6 Value-based pricing considerations
4.7 Pricing strategy recommendations
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Product development opportunities
5.4 Market entry barriers
5.5 Customer feedback incorporation
5.6 Future trends anticipation
5.7 Strategic recommendations
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer engagement strategies
6.4 Feedback collection methods
6.5 Relationship management tools
6.6 Community building initiatives
6.7 Customer satisfaction metrics
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling points
7.4 Customer-centric approach
7.5 Competitive advantages
7.6 Market differentiation strategies
7.7 Value delivery mechanisms
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Marketing campaigns
8.5 Training and development
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries selection
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
10.6 Strategic fit evaluation
10.7 Long-term sustainability considerations
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Financial projections
11.4 Funding sources identification
11.5 Budget allocation strategies
11.6 Cost management plans
11.7 Risk mitigation strategies
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnership benefits
12.3 Risk assessment frameworks
12.4 Control mechanisms
12.5 Strategic alignment
12.6 Long-term implications
12.7 Decision-making processes
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin projections
13.4 Revenue growth strategies
13.5 Cost reduction initiatives
13.6 Financial health indicators
13.7 Investment return expectations
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Collaborative opportunities
14.6 Partnership evaluation criteria
14.7 Long-term partnership strategies
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for key activities
15.2.2 Milestone tracking
15.2.3 Performance evaluation
15.2.4 Adjustment strategies
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of government health statistics and reports on contraceptive usage in Japan
- Review of academic journals and publications focusing on emergency contraceptive pills (ECPs) and reproductive health
- Examination of market reports from health organizations and NGOs regarding access and awareness of ECPs
Primary Research
- Interviews with healthcare professionals, including gynecologists and pharmacists, to understand prescribing patterns
- Surveys conducted with women of reproductive age to gauge awareness and attitudes towards emergency contraceptive pills
- Focus groups with stakeholders in reproductive health advocacy to discuss barriers to access and education
Validation & Triangulation
- Cross-validation of findings through comparison with international studies on ECP usage
- Triangulation of data from healthcare providers, patient surveys, and market reports
- Sanity checks through expert panel reviews involving reproductive health specialists
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total market size based on national contraceptive sales data and population demographics
- Segmentation of the market by age group, urban vs. rural distribution, and socio-economic status
- Incorporation of trends in contraceptive use and public health initiatives promoting ECPs
Bottom-up Modeling
- Collection of sales data from pharmacies and healthcare providers regarding ECP distribution
- Estimation of average pricing and volume sold per transaction across various retail channels
- Analysis of prescription vs. over-the-counter sales to determine market dynamics
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as public health campaigns and changes in legislation
- Scenario modeling based on potential shifts in consumer behavior and healthcare policies
- Development of baseline, optimistic, and pessimistic forecasts for the ECP market through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Provider Insights | 100 | Gynecologists, Family Physicians |
| Pharmacy Distribution Analysis | 80 | Pharmacists, Pharmacy Managers |
| Consumer Awareness Survey | 120 | Women aged 18-45, Urban and Rural Residents |
| Advocacy Group Perspectives | 60 | Reproductive Health Advocates, NGO Representatives |
| Market Trend Analysis | 70 | Market Analysts, Health Economists |
Frequently Asked Questions
What is the current value of the Japan Emergency Contraceptive Pills market?
The Japan Emergency Contraceptive Pills market is valued at approximately USD 140 million, representing a significant segment of the broader Japan contraceptive drugs market, which is valued at around USD 720 million.