About the Report
Base Year 2024Japan In Situ Gel Drug Delivery Market Overview
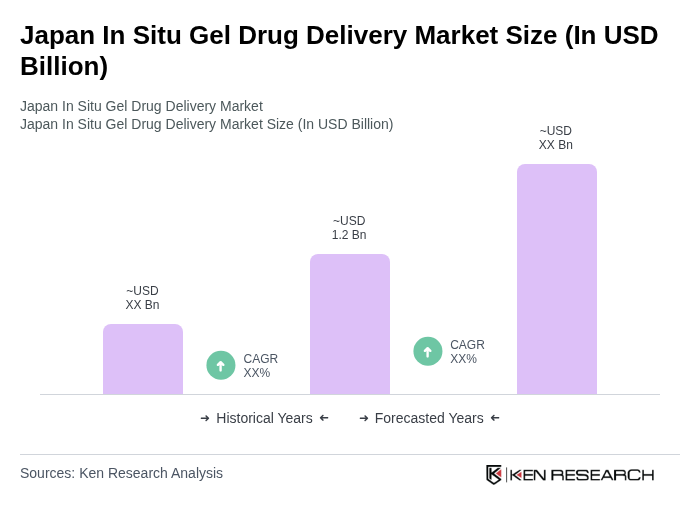
- The Japan In Situ Gel Drug Delivery Market is valued at USD 1.2 billion, based on a five-year historical analysis. This growth is primarily driven by advancements in drug formulation technologies, increasing prevalence of chronic diseases, and a rising demand for targeted drug delivery systems. The market is also supported by the growing geriatric population, which necessitates innovative healthcare solutions.
- Tokyo, Osaka, and Yokohama are the dominant cities in the Japan In Situ Gel Drug Delivery Market. Tokyo leads due to its status as a major healthcare hub with numerous research institutions and pharmaceutical companies. Osaka and Yokohama follow closely, benefiting from their strategic locations and robust healthcare infrastructure, which facilitate the development and distribution of advanced drug delivery systems.
- In 2023, the Japanese government implemented the "Pharmaceutical and Medical Device Act," which aims to streamline the approval process for innovative drug delivery systems, including in situ gels. This regulation encourages research and development in the pharmaceutical sector, promoting faster access to new therapies for patients while ensuring safety and efficacy standards are met.
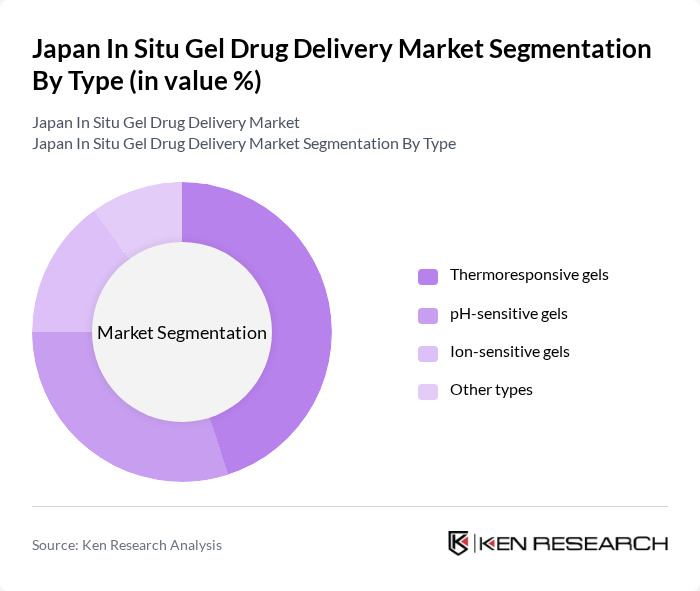
Japan In Situ Gel Drug Delivery Market Segmentation
By Type:The market is segmented into various types of in situ gel formulations, including thermoresponsive gels, pH-sensitive gels, ion-sensitive gels, and other types. Among these, thermoresponsive gels are gaining significant traction due to their ability to transition from a sol to a gel state in response to temperature changes, making them ideal for injectable drug delivery. The increasing demand for minimally invasive procedures and patient-friendly drug administration methods is driving the growth of this segment.
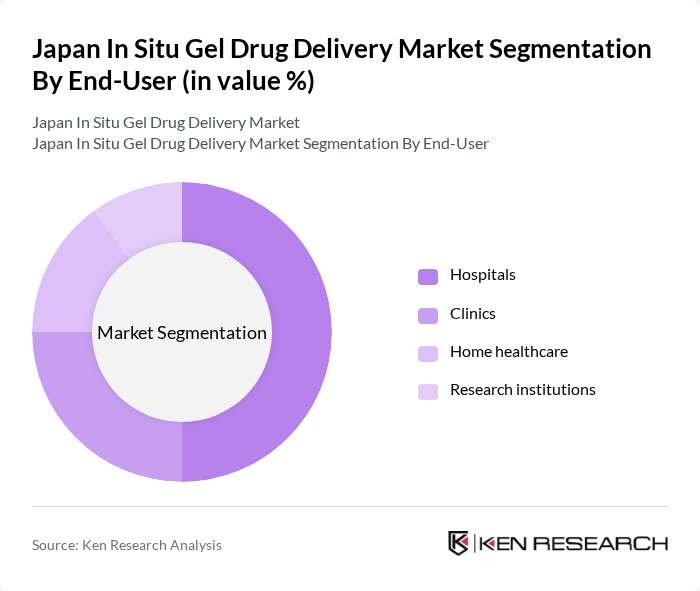
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, and research institutions. Hospitals are the leading end-users of in situ gel drug delivery systems, primarily due to their extensive patient base and the need for advanced therapeutic solutions. The increasing adoption of innovative drug delivery methods in hospital settings is driven by the need for effective pain management and treatment of chronic diseases.
Japan In Situ Gel Drug Delivery Market Competitive Landscape
The Japan In Situ Gel Drug Delivery Market is characterized by a dynamic mix of regional and international players. Leading participants such as Astellas Pharma Inc., Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company, Limited, Otsuka Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Kyowa Kirin Co., Ltd., Eisai Co., Ltd., Shionogi & Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Fujifilm Corporation, Nipro Corporation, Kaken Pharmaceutical Co., Ltd., Santen Pharmaceutical Co., Ltd., A2A Pharmaceuticals, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Japan In Situ Gel Drug Delivery Market Industry Analysis
Growth Drivers
- Increasing Demand for Targeted Drug Delivery Systems:The Japanese healthcare market is witnessing a significant shift towards targeted drug delivery systems, driven by a growing aging population, which is projected to reach 36.5 million in the future. This demographic shift is accompanied by a rise in chronic diseases, necessitating more effective treatment options. The demand for precision medicine is expected to increase, with the market for targeted therapies projected to reach ¥1.2 trillion in the future, highlighting the need for innovative drug delivery solutions.
- Advancements in Polymer Technology:The development of advanced polymer materials is revolutionizing the in situ gel drug delivery market in Japan. Innovations in biocompatible and biodegradable polymers are enhancing the efficacy and safety of drug delivery systems. For instance, the market for biodegradable polymers is expected to grow to ¥300 billion in the future, driven by their application in drug delivery. These advancements are crucial for creating gels that can provide sustained release of therapeutics, improving patient outcomes significantly.
- Rising Prevalence of Chronic Diseases:Chronic diseases such as diabetes and cardiovascular disorders are on the rise in Japan, with approximately 10 million people diagnosed with diabetes as of the future. This increasing prevalence is driving the demand for effective drug delivery systems that can manage these conditions more efficiently. The healthcare expenditure on chronic disease management is projected to exceed ¥50 trillion in the future, creating a substantial market for innovative drug delivery solutions like in situ gels.
Market Challenges
- High Manufacturing Costs:The production of in situ gel drug delivery systems involves complex processes and high-quality materials, leading to elevated manufacturing costs. For instance, the average cost of developing a new drug delivery system can exceed ¥1 billion, which poses a significant barrier for smaller companies. This financial burden can limit innovation and market entry, hindering the overall growth of the in situ gel market in Japan.
- Regulatory Hurdles:The regulatory landscape for drug delivery systems in Japan is stringent, with the Pharmaceuticals and Medical Devices Agency (PMDA) enforcing rigorous approval processes. The average time for drug approval can take up to 3-5 years, which delays market entry for new products. These regulatory challenges can deter investment in research and development, slowing the pace of innovation in the in situ gel drug delivery sector.
Japan In Situ Gel Drug Delivery Market Future Outlook
The future of the in situ gel drug delivery market in Japan appears promising, driven by technological advancements and increasing healthcare demands. The integration of smart technologies, such as IoT and AI, is expected to enhance drug delivery efficiency and patient monitoring. Additionally, the focus on personalized medicine will likely lead to tailored drug delivery solutions, improving therapeutic outcomes. As healthcare infrastructure expands, the market is poised for significant growth, addressing the evolving needs of patients and healthcare providers alike.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Japanese government is investing heavily in healthcare infrastructure, with a budget increase to ¥45 trillion in the future. This expansion will facilitate the adoption of advanced drug delivery systems, including in situ gels, providing a significant opportunity for market players to introduce innovative solutions that meet the growing healthcare demands.
- Collaborations with Pharmaceutical Companies:Strategic partnerships between drug delivery companies and pharmaceutical firms are on the rise, with over 50 collaborations reported in the future. These alliances can accelerate the development and commercialization of in situ gel systems, leveraging combined expertise and resources to enhance product offerings and market reach.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Thermoresponsive gels pH-sensitive gels Ion-sensitive gels Other types |
| By End-User | Hospitals Clinics Home healthcare Research institutions |
| By Application | Oncology Cardiovascular diseases Pain management Other applications |
| By Delivery Method | Injectable Implantable Transdermal Other methods |
| By Material | Natural polymers Synthetic polymers Hybrid materials Other materials |
| By Region | Kanto Kansai Chubu Other regions |
| By Others | Custom formulations Combination therapies Other innovations |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Pharmaceuticals and Medical Devices Agency, Ministry of Health, Labour and Welfare)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Biotechnology Firms
Pharmaceutical Companies
Insurance Companies and Payers
Players Mentioned in the Report:
Astellas Pharma Inc.
Takeda Pharmaceutical Company Limited
Daiichi Sankyo Company, Limited
Otsuka Pharmaceutical Co., Ltd.
Chugai Pharmaceutical Co., Ltd.
Mitsubishi Tanabe Pharma Corporation
Kyowa Kirin Co., Ltd.
Eisai Co., Ltd.
Shionogi & Co., Ltd.
Sumitomo Dainippon Pharma Co., Ltd.
Fujifilm Corporation
Nipro Corporation
Kaken Pharmaceutical Co., Ltd.
Santen Pharmaceutical Co., Ltd.
A2A Pharmaceuticals, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan In Situ Gel Drug Delivery Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan In Situ Gel Drug Delivery Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan In Situ Gel Drug Delivery Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for targeted drug delivery systems
3.1.2 Advancements in polymer technology
3.1.3 Rising prevalence of chronic diseases
3.1.4 Growing investment in R&D for drug delivery systems
3.2 Market Challenges
3.2.1 High manufacturing costs
3.2.2 Regulatory hurdles
3.2.3 Limited awareness among healthcare professionals
3.2.4 Competition from alternative drug delivery methods
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Collaborations with pharmaceutical companies
3.3.3 Development of personalized medicine
3.3.4 Increasing focus on patient-centric drug delivery solutions
3.4 Market Trends
3.4.1 Growth of biodegradable in situ gels
3.4.2 Integration of smart technology in drug delivery
3.4.3 Rise in demand for minimally invasive procedures
3.4.4 Focus on sustainable and eco-friendly materials
3.5 Government Regulation
3.5.1 Stricter guidelines for drug approval processes
3.5.2 Incentives for R&D in drug delivery technologies
3.5.3 Regulations on manufacturing practices
3.5.4 Compliance requirements for clinical trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan In Situ Gel Drug Delivery Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan In Situ Gel Drug Delivery Market Segmentation
8.1 By Type
8.1.1 Thermoresponsive gels
8.1.2 pH-sensitive gels
8.1.3 Ion-sensitive gels
8.1.4 Other types
8.2 By End-User
8.2.1 Hospitals
8.2.2 Clinics
8.2.3 Home healthcare
8.2.4 Research institutions
8.3 By Application
8.3.1 Oncology
8.3.2 Cardiovascular diseases
8.3.3 Pain management
8.3.4 Other applications
8.4 By Delivery Method
8.4.1 Injectable
8.4.2 Implantable
8.4.3 Transdermal
8.4.4 Other methods
8.5 By Material
8.5.1 Natural polymers
8.5.2 Synthetic polymers
8.5.3 Hybrid materials
8.5.4 Other materials
8.6 By Region
8.6.1 Kanto
8.6.2 Kansai
8.6.3 Chubu
8.6.4 Other regions
8.7 By Others
8.7.1 Custom formulations
8.7.2 Combination therapies
8.7.3 Other innovations
9. Japan In Situ Gel Drug Delivery Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Product Development Cycle Time
9.2.7 Pricing Strategy
9.2.8 Distribution Efficiency
9.2.9 Brand Recognition
9.2.10 Innovation Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Astellas Pharma Inc.
9.5.2 Takeda Pharmaceutical Company Limited
9.5.3 Daiichi Sankyo Company, Limited
9.5.4 Otsuka Pharmaceutical Co., Ltd.
9.5.5 Chugai Pharmaceutical Co., Ltd.
9.5.6 Mitsubishi Tanabe Pharma Corporation
9.5.7 Kyowa Kirin Co., Ltd.
9.5.8 Eisai Co., Ltd.
9.5.9 Shionogi & Co., Ltd.
9.5.10 Sumitomo Dainippon Pharma Co., Ltd.
9.5.11 Fujifilm Corporation
9.5.12 Nipro Corporation
9.5.13 Kaken Pharmaceutical Co., Ltd.
9.5.14 Santen Pharmaceutical Co., Ltd.
9.5.15 A2A Pharmaceuticals, Inc.
10. Japan In Situ Gel Drug Delivery Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health, Labour and Welfare
10.1.2 Ministry of Education, Culture, Sports, Science and Technology
10.1.3 Ministry of Economy, Trade and Industry
10.1.4 Other ministries
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare infrastructure
10.2.2 Funding for research and development
10.2.3 Expenditure on technology upgrades
10.2.4 Other spending patterns
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home healthcare providers
10.3.4 Research institutions
10.4 User Readiness for Adoption
10.4.1 Awareness of in situ gel technologies
10.4.2 Training and education needs
10.4.3 Infrastructure readiness
10.4.4 Other readiness factors
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of treatment outcomes
10.5.2 Cost-effectiveness analysis
10.5.3 User feedback and satisfaction
10.5.4 Other expansion opportunities
11. Japan In Situ Gel Drug Delivery Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from Japanese pharmaceutical and biotechnology associations
- Review of academic journals and publications focusing on in situ gel drug delivery technologies
- Examination of regulatory frameworks and guidelines from Japan's Ministry of Health, Labour and Welfare
Primary Research
- Interviews with R&D heads at leading pharmaceutical companies specializing in drug delivery systems
- Surveys with healthcare professionals, including oncologists and pain management specialists
- Field interviews with pharmacists and hospital administrators regarding drug delivery preferences
Validation & Triangulation
- Cross-validation of data through multiple sources, including industry reports and expert opinions
- Triangulation of findings from primary interviews with secondary data insights
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and pharmaceutical sales data
- Segmentation by therapeutic areas such as oncology, ophthalmology, and pain management
- Incorporation of demographic trends and aging population statistics in Japan
Bottom-up Modeling
- Volume estimates derived from clinical trial data and product launch timelines
- Cost analysis based on pricing strategies of existing in situ gel products
- Market penetration rates calculated from adoption trends in hospitals and clinics
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors like technological advancements and regulatory changes
- Scenario modeling based on potential market disruptions and healthcare policy shifts
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Drug Delivery Systems | 100 | Oncologists, Clinical Researchers |
| Ophthalmic Gel Applications | 80 | Ophthalmologists, Pharmacists |
| Pain Management Solutions | 70 | Pain Specialists, General Practitioners |
| Regulatory Compliance Insights | 60 | Regulatory Affairs Managers, Compliance Officers |
| Market Trends in Drug Delivery | 90 | Healthcare Analysts, Market Researchers |
Frequently Asked Questions
What is the current value of the Japan In Situ Gel Drug Delivery Market?
The Japan In Situ Gel Drug Delivery Market is valued at approximately USD 1.2 billion, reflecting significant growth driven by advancements in drug formulation technologies and an increasing prevalence of chronic diseases.