About the Report
Base Year 2024Japan Smart Cold Chain for Pharmaceuticals Market Overview
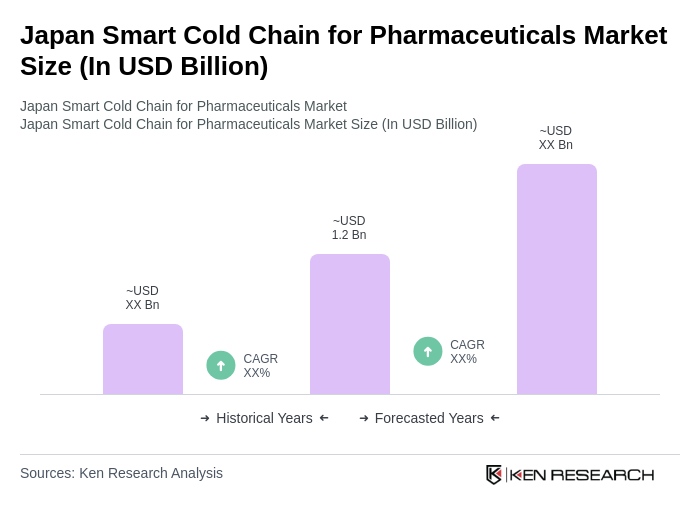
- The Japan Smart Cold Chain for Pharmaceuticals Market is valued at USD 1.2 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for temperature-sensitive pharmaceuticals, including vaccines and biologics, alongside the rising need for efficient logistics solutions in the healthcare sector. The market is also supported by advancements in technology that enhance monitoring and tracking capabilities, ensuring product integrity throughout the supply chain.
- Tokyo, Osaka, and Yokohama are the dominant cities in the Japan Smart Cold Chain for Pharmaceuticals Market due to their robust healthcare infrastructure, high concentration of pharmaceutical companies, and advanced logistics networks. These cities serve as key hubs for distribution and storage, facilitating efficient cold chain operations and meeting the stringent regulatory requirements for pharmaceutical transportation.
- In 2023, the Japanese government implemented the "Cold Chain Logistics Improvement Act," which mandates stricter temperature control measures for the transportation and storage of pharmaceuticals. This regulation aims to enhance the safety and efficacy of temperature-sensitive products, ensuring compliance with international standards and improving overall public health outcomes.
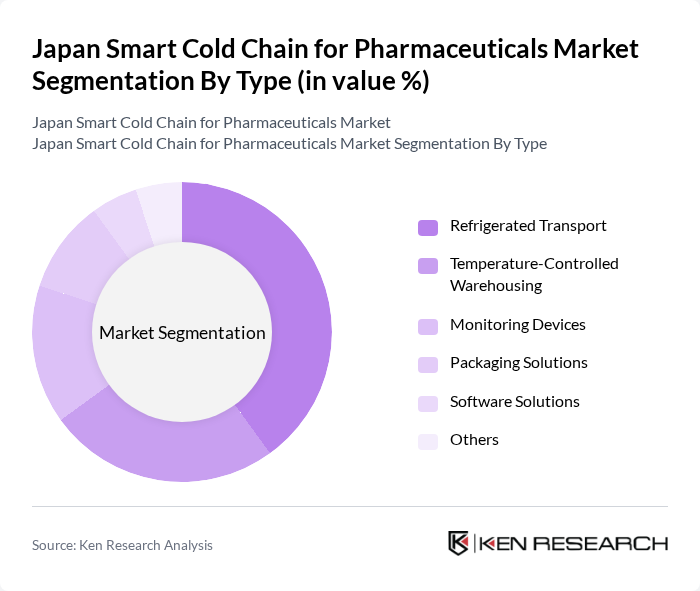
Japan Smart Cold Chain for Pharmaceuticals Market Segmentation
By Type:The market is segmented into various types, including Refrigerated Transport, Temperature-Controlled Warehousing, Monitoring Devices, Packaging Solutions, Software Solutions, and Others. Among these, Refrigerated Transport is the leading sub-segment due to the increasing demand for efficient and reliable transportation of temperature-sensitive pharmaceuticals. The rise in e-commerce and direct-to-consumer delivery models has further fueled the need for advanced refrigerated transport solutions.
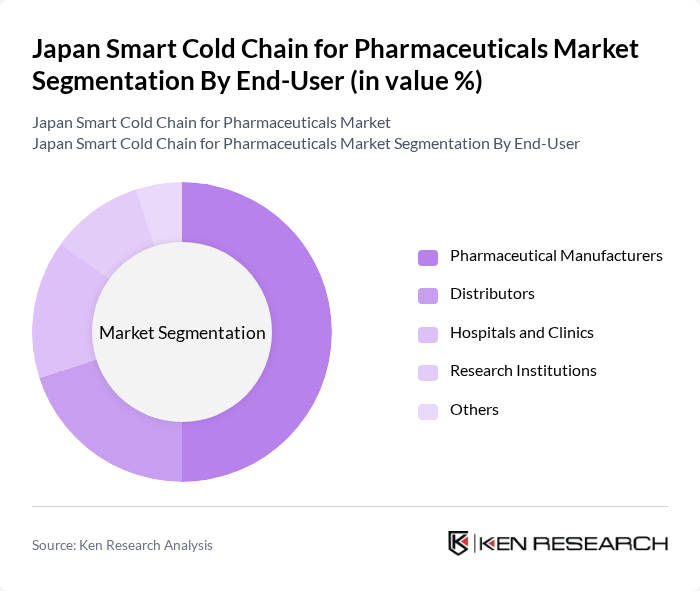
By End-User:The end-user segmentation includes Pharmaceutical Manufacturers, Distributors, Hospitals and Clinics, Research Institutions, and Others. Pharmaceutical Manufacturers dominate this segment as they require reliable cold chain solutions to maintain the efficacy of their products during transportation and storage. The increasing focus on biologics and personalized medicine further drives the demand for specialized cold chain services among manufacturers.
Japan Smart Cold Chain for Pharmaceuticals Market Competitive Landscape
The Japan Smart Cold Chain for Pharmaceuticals Market is characterized by a dynamic mix of regional and international players. Leading participants such as Mitsubishi Logistics Corporation, Nippon Express Co., Ltd., Kintetsu World Express, Inc., Yamato Holdings Co., Ltd., Seino Holdings Co., Ltd., Sagawa Express Co., Ltd., DB Schenker Japan, DHL Supply Chain Japan, FedEx Express Japan, UPS Japan, Takeda Pharmaceutical Company Limited, Astellas Pharma Inc., Daiichi Sankyo Company, Limited, Chugai Pharmaceutical Co., Ltd., Otsuka Pharmaceutical Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
Japan Smart Cold Chain for Pharmaceuticals Market Industry Analysis
Growth Drivers
- Increasing Demand for Temperature-Sensitive Pharmaceuticals:The Japanese pharmaceutical market is projected to reach ¥10 trillion (approximately $90 billion) in future, driven by a growing demand for temperature-sensitive products. With over 50% of new drugs requiring strict temperature control, the need for efficient cold chain logistics is paramount. This demand is further fueled by the rise in chronic diseases, necessitating the distribution of biologics and vaccines that must be maintained within specific temperature ranges to ensure efficacy and safety.
- Advancements in IoT and Monitoring Technologies:The integration of IoT technologies in cold chain logistics is transforming the pharmaceutical sector in Japan. By future, the IoT market in logistics is expected to exceed ¥1.5 trillion (around $13.5 billion), enhancing real-time monitoring capabilities. These advancements allow for precise temperature tracking and data analytics, reducing spoilage rates and ensuring compliance with stringent regulations. Consequently, pharmaceutical companies are increasingly investing in smart cold chain solutions to optimize their supply chains.
- Rising Regulatory Requirements for Drug Safety:Japan's regulatory landscape is becoming increasingly stringent, with the Pharmaceuticals and Medical Devices Agency (PMDA) enforcing rigorous standards for drug safety. By future, compliance costs are expected to rise by 20%, emphasizing the need for reliable cold chain solutions. This regulatory pressure drives pharmaceutical companies to invest in advanced cold chain technologies to ensure adherence to Good Distribution Practices (GDP) and maintain product integrity throughout the supply chain.
Market Challenges
- High Initial Investment Costs:The implementation of smart cold chain solutions requires significant upfront capital investment, often exceeding ¥500 million (approximately $4.5 million) for advanced systems. This financial barrier can deter smaller pharmaceutical companies from adopting necessary technologies. As a result, many firms struggle to balance the need for compliance and efficiency with the constraints of their budgets, potentially compromising the quality of their temperature-sensitive products.
- Complexity of Logistics and Supply Chain Management:The logistics of managing a smart cold chain are inherently complex, involving multiple stakeholders and stringent regulatory requirements. In future, the average lead time for temperature-sensitive shipments is projected to increase by 15%, complicating inventory management. This complexity can lead to inefficiencies and increased risk of product spoilage, challenging companies to streamline their operations while maintaining compliance with evolving regulations.
Japan Smart Cold Chain for Pharmaceuticals Market Future Outlook
The future of the smart cold chain for pharmaceuticals in Japan is poised for significant transformation, driven by technological advancements and regulatory pressures. As companies increasingly adopt AI and real-time data analytics, operational efficiencies are expected to improve, reducing waste and enhancing compliance. Furthermore, the growing emphasis on sustainability will likely lead to innovative practices in logistics, ensuring that the cold chain not only meets safety standards but also aligns with environmental goals, fostering a more resilient supply chain.
Market Opportunities
- Growth in E-commerce for Pharmaceuticals:The e-commerce sector for pharmaceuticals in Japan is projected to reach ¥1 trillion (approximately $9 billion) by future. This growth presents a significant opportunity for smart cold chain solutions, as online sales of temperature-sensitive products require robust logistics to ensure product integrity during transit, driving demand for advanced cold chain technologies.
- Adoption of Blockchain for Supply Chain Transparency:The implementation of blockchain technology in the pharmaceutical cold chain is gaining traction, with an expected market value of ¥200 billion (around $1.8 billion) by future. This technology enhances traceability and transparency, allowing stakeholders to verify the integrity of temperature-sensitive products throughout the supply chain, thereby increasing consumer trust and regulatory compliance.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Refrigerated Transport Temperature-Controlled Warehousing Monitoring Devices Packaging Solutions Software Solutions Others |
| By End-User | Pharmaceutical Manufacturers Distributors Hospitals and Clinics Research Institutions Others |
| By Application | Vaccines Biologics Blood Products Clinical Trials Others |
| By Distribution Mode | Direct Distribution Third-Party Logistics E-commerce Platforms Others |
| By Packaging Type | Insulated Containers Refrigerated Boxes Active Packaging Passive Packaging Others |
| By Temperature Range | 8°C °C °C Others |
| By Service Type | Consulting Services Maintenance Services Training Services Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Labour and Welfare; Pharmaceuticals and Medical Devices Agency)
Pharmaceutical Manufacturers
Cold Chain Logistics Providers
Healthcare Providers and Hospitals
Biotechnology Companies
Pharmaceutical Wholesalers
Supply Chain Management Firms
Players Mentioned in the Report:
Mitsubishi Logistics Corporation
Nippon Express Co., Ltd.
Kintetsu World Express, Inc.
Yamato Holdings Co., Ltd.
Seino Holdings Co., Ltd.
Sagawa Express Co., Ltd.
DB Schenker Japan
DHL Supply Chain Japan
FedEx Express Japan
UPS Japan
Takeda Pharmaceutical Company Limited
Astellas Pharma Inc.
Daiichi Sankyo Company, Limited
Chugai Pharmaceutical Co., Ltd.
Otsuka Pharmaceutical Co., Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan Smart Cold Chain for Pharmaceuticals Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan Smart Cold Chain for Pharmaceuticals Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan Smart Cold Chain for Pharmaceuticals Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for temperature-sensitive pharmaceuticals
3.1.2 Advancements in IoT and monitoring technologies
3.1.3 Rising regulatory requirements for drug safety
3.1.4 Expansion of biopharmaceuticals and personalized medicine
3.2 Market Challenges
3.2.1 High initial investment costs
3.2.2 Complexity of logistics and supply chain management
3.2.3 Limited infrastructure in rural areas
3.2.4 Stringent regulatory compliance requirements
3.3 Market Opportunities
3.3.1 Growth in e-commerce for pharmaceuticals
3.3.2 Adoption of blockchain for supply chain transparency
3.3.3 Increasing focus on sustainability in logistics
3.3.4 Collaborations with technology providers
3.4 Market Trends
3.4.1 Integration of AI in cold chain management
3.4.2 Use of real-time data analytics
3.4.3 Shift towards automated cold chain solutions
3.4.4 Growing emphasis on end-to-end visibility
3.5 Government Regulation
3.5.1 Compliance with Good Distribution Practice (GDP)
3.5.2 Regulations on temperature monitoring and reporting
3.5.3 Guidelines for transportation of biological products
3.5.4 Standards for packaging and labeling of pharmaceuticals
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan Smart Cold Chain for Pharmaceuticals Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan Smart Cold Chain for Pharmaceuticals Market Segmentation
8.1 By Type
8.1.1 Refrigerated Transport
8.1.2 Temperature-Controlled Warehousing
8.1.3 Monitoring Devices
8.1.4 Packaging Solutions
8.1.5 Software Solutions
8.1.6 Others
8.2 By End-User
8.2.1 Pharmaceutical Manufacturers
8.2.2 Distributors
8.2.3 Hospitals and Clinics
8.2.4 Research Institutions
8.2.5 Others
8.3 By Application
8.3.1 Vaccines
8.3.2 Biologics
8.3.3 Blood Products
8.3.4 Clinical Trials
8.3.5 Others
8.4 By Distribution Mode
8.4.1 Direct Distribution
8.4.2 Third-Party Logistics
8.4.3 E-commerce Platforms
8.4.4 Others
8.5 By Packaging Type
8.5.1 Insulated Containers
8.5.2 Refrigerated Boxes
8.5.3 Active Packaging
8.5.4 Passive Packaging
8.5.5 Others
8.6 By Temperature Range
8.6.1 2-8°C
8.6.2 -20°C
8.6.3 -80°C
8.6.4 Others
8.7 By Service Type
8.7.1 Consulting Services
8.7.2 Maintenance Services
8.7.3 Training Services
8.7.4 Others
9. Japan Smart Cold Chain for Pharmaceuticals Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Operational Efficiency
9.2.7 Pricing Strategy
9.2.8 Supply Chain Efficiency
9.2.9 Innovation Rate
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Mitsubishi Logistics Corporation
9.5.2 Nippon Express Co., Ltd.
9.5.3 Kintetsu World Express, Inc.
9.5.4 Yamato Holdings Co., Ltd.
9.5.5 Seino Holdings Co., Ltd.
9.5.6 Sagawa Express Co., Ltd.
9.5.7 DB Schenker Japan
9.5.8 DHL Supply Chain Japan
9.5.9 FedEx Express Japan
9.5.10 UPS Japan
9.5.11 Takeda Pharmaceutical Company Limited
9.5.12 Astellas Pharma Inc.
9.5.13 Daiichi Sankyo Company, Limited
9.5.14 Chugai Pharmaceutical Co., Ltd.
9.5.15 Otsuka Pharmaceutical Co., Ltd.
10. Japan Smart Cold Chain for Pharmaceuticals Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health, Labour and Welfare
10.1.2 Ministry of Economy, Trade and Industry
10.1.3 Ministry of Agriculture, Forestry and Fisheries
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Cold Chain Infrastructure
10.2.2 Energy Efficiency Initiatives
10.2.3 Budget Allocation for Technology Upgrades
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Manufacturers
10.3.2 Distributors
10.3.3 Healthcare Providers
10.4 User Readiness for Adoption
10.4.1 Awareness of Smart Cold Chain Solutions
10.4.2 Training and Skill Development Needs
10.4.3 Financial Readiness for Investment
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Cost Savings
10.5.2 Expansion of Service Offerings
10.5.3 Long-term Sustainability Benefits
11. Japan Smart Cold Chain for Pharmaceuticals Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Model Structuring
1.4 Customer Segmentation
1.5 Key Partnerships
1.6 Cost Structure Analysis
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Direct-to-Consumer Channels
3.4 Partnerships with Logistics Providers
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends Identification
5.4 Future Needs Assessment
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Engagement Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Cost Efficiency
7.4 Customer-Centric Solutions
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Innovations
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Management Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Identification
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from pharmaceutical logistics associations in Japan
- Review of government publications on cold chain regulations and standards
- Examination of market trends through academic journals and white papers on pharmaceutical supply chains
Primary Research
- Interviews with logistics managers at major pharmaceutical companies
- Surveys with cold chain technology providers and equipment manufacturers
- Field interviews with regulatory compliance officers in the pharmaceutical sector
Validation & Triangulation
- Cross-validation of data through multiple industry sources and expert opinions
- Triangulation of findings from desk research and primary interviews
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total pharmaceutical market size in Japan as a baseline
- Segmentation of cold chain logistics by pharmaceutical categories (e.g., vaccines, biologics)
- Incorporation of growth rates from historical data and future projections
Bottom-up Modeling
- Collection of operational data from leading cold chain service providers
- Estimation of costs associated with temperature-controlled transportation and storage
- Volume and cost analysis based on specific pharmaceutical products and their distribution needs
Forecasting & Scenario Analysis
- Development of forecasting models based on market drivers such as aging population and increased healthcare spending
- Scenario analysis considering potential regulatory changes and technological advancements
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Distribution Logistics | 150 | Logistics Managers, Supply Chain Analysts |
| Cold Chain Technology Providers | 100 | Product Managers, Technical Directors |
| Regulatory Compliance in Pharmaceuticals | 80 | Compliance Officers, Quality Assurance Managers |
| Pharmaceutical Retail Sector | 70 | Pharmacy Managers, Inventory Control Specialists |
| Healthcare Logistics Innovations | 90 | Research & Development Managers, Operations Executives |
Frequently Asked Questions
What is the current value of the Japan Smart Cold Chain for Pharmaceuticals Market?
The Japan Smart Cold Chain for Pharmaceuticals Market is valued at approximately USD 1.2 billion, driven by the increasing demand for temperature-sensitive pharmaceuticals, including vaccines and biologics, and the need for efficient logistics solutions in the healthcare sector.