Region:Middle East
Author(s):Dev
Product Code:KRAC0974
Pages:82
Published On:December 2025
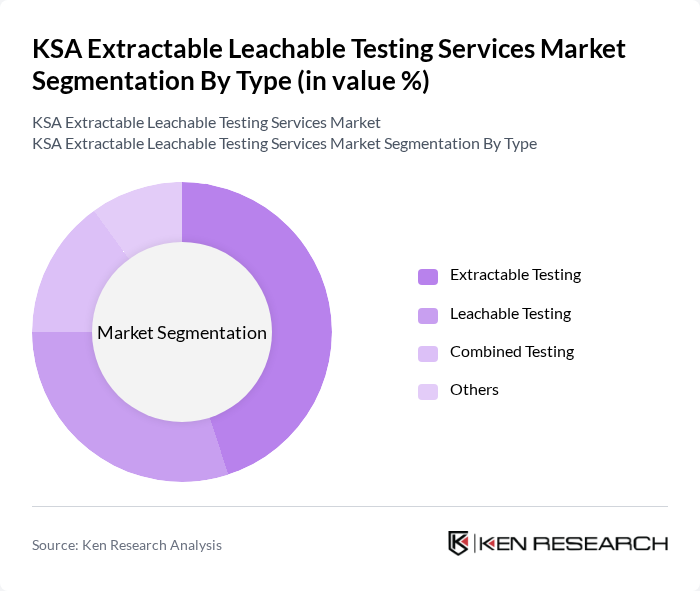
By Type:The market is segmented into Extractable Testing, Leachable Testing, Combined Testing, and Others. Among these, Extractable Testing is the leading sub-segment due to its critical role in ensuring the safety and compliance of pharmaceutical products. The increasing focus on product safety and regulatory compliance drives the demand for this testing type, as manufacturers seek to mitigate risks associated with extractable substances.
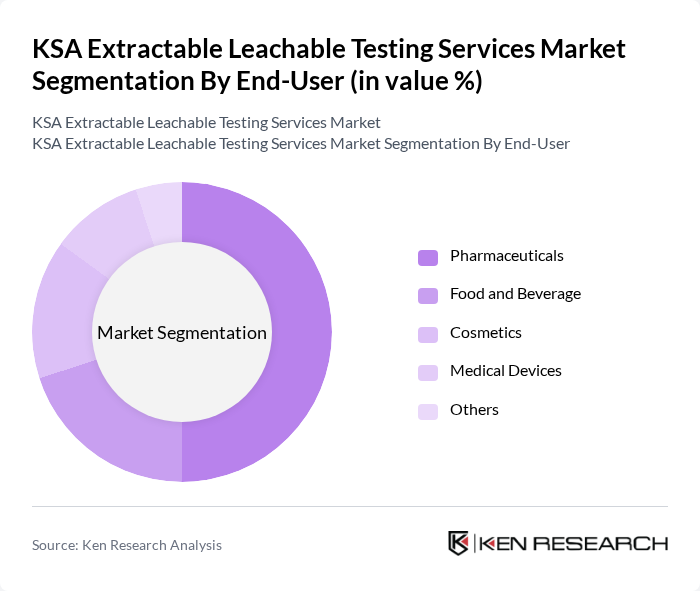
By End-User:The end-user segmentation includes Pharmaceuticals, Food and Beverage, Cosmetics, Medical Devices, and Others. The Pharmaceuticals segment dominates the market, driven by the stringent regulatory requirements for drug safety and efficacy. Pharmaceutical companies are increasingly investing in testing services to ensure compliance with local and international standards, thereby enhancing product safety and consumer trust.
The KSA Extractable Leachable Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as SGS SA, Eurofins Scientific, Intertek Group plc, Bureau Veritas, ALS Limited, TUV SUD, Merck KGaA, Charles Lawrence International, Nelson Labs, Labcorp, Pace Analytical Services, Q Laboratories, Ametek, Inc., Chemours Company, Thermo Fisher Scientific contribute to innovation, geographic expansion, and service delivery in this space.
The future of the KSA extractable leachable testing services market appears promising, driven by digital transformation and a focus on sustainability. The expansion of national data infrastructure is likely to enhance the adoption of digital lab information management systems and AI-based analytics, improving testing accuracy and efficiency. Additionally, the emphasis on local content and eco-friendly practices under Vision 2030 will likely drive demand for compliant testing services, ensuring consumer safety and regulatory adherence in various sectors.
| Segment | Sub-Segments |
|---|---|
| By Type | Extractable Testing Leachable Testing Combined Testing Others |
| By End-User | Pharmaceuticals Food and Beverage Cosmetics Medical Devices Others |
| By Industry Application | Healthcare Manufacturing Research and Development Others |
| By Testing Method | Laboratory Testing Field Testing Remote Testing Others |
| By Geographic Distribution | Central Region Eastern Region Western Region Southern Region |
| By Service Provider Type | Independent Testing Labs In-House Testing Facilities Third-Party Testing Services Others |
| By Certification Type | ISO Certified GMP Certified FDA Approved Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Services | 100 | Quality Control Managers, Regulatory Affairs Specialists |
| Food Packaging Compliance | 80 | Food Safety Officers, Packaging Engineers |
| Consumer Goods Testing | 70 | Product Development Managers, Compliance Officers |
| Environmental Testing Services | 60 | Environmental Compliance Managers, Laboratory Directors |
| Industrial Manufacturing Testing | 90 | Manufacturing Engineers, Quality Assurance Managers |
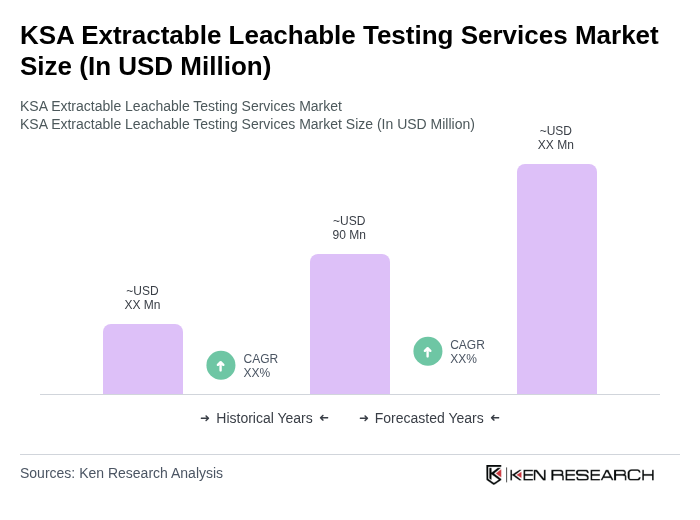
The KSA Extractable Leachable Testing Services Market is valued at approximately USD 90 million, reflecting a significant growth driven by increased demand for pharmaceutical analytical testing and regulatory compliance in the Kingdom.