About the Report
Base Year 2024KSA Rare Hemophilia Factors Market Overview
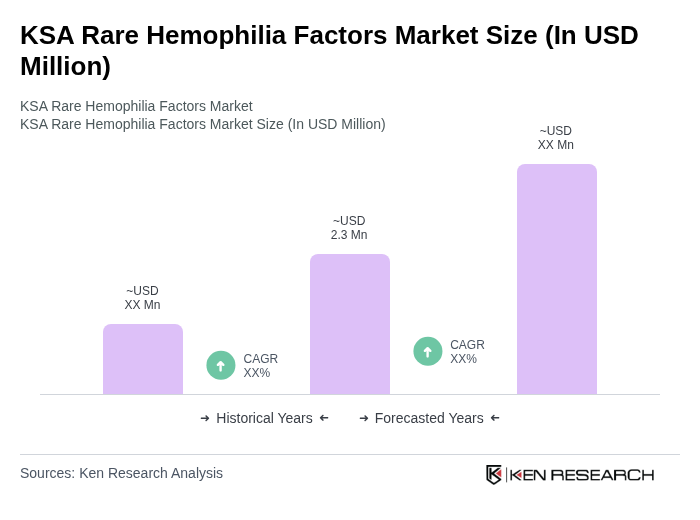
- The KSA Rare Hemophilia Factors Market is valued at USD 2.3 million, based on a five-year historical analysis. This growth is primarily driven by the increasing identification of rare hemophilia factor deficiencies, broader adoption of prophylactic treatment regimens, and rising healthcare expenditure in the kingdom. The demand for specialized therapies and factor concentrates has increased in line with improved diagnostic capabilities, newborn screening, and the expansion of tertiary hematology centers, reflecting growing awareness of these conditions among healthcare providers and patients.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the KSA Rare Hemophilia Factors Market due to their advanced healthcare infrastructure and concentration of specialized medical facilities. These urban centers host major tertiary hospitals and comprehensive hemophilia treatment centers that provide multidisciplinary care, genetic counseling, and access to factor replacement therapies and bypassing agents for patients with rare factor deficiencies.
- In 2023, the Saudi Food and Drug Authority (SFDA) strengthened the regulatory framework governing hemophilia and rare coagulation factor therapies through updated pharmacovigilance and biologics registration requirements, including for plasma-derived and recombinant coagulation factors. Instruments such as the Guideline on Registration of Biological Products issued by the Saudi Food and Drug Authority in 2023 set detailed standards for quality, safety, efficacy, and risk management plans, as well as ongoing post-marketing surveillance for factor concentrates and related therapies. These measures are designed to ensure that patients receive high-quality products aligned with international benchmarks, support traceability of batches supplied through national tenders, and ultimately improve patient outcomes while reinforcing confidence in the healthcare system.
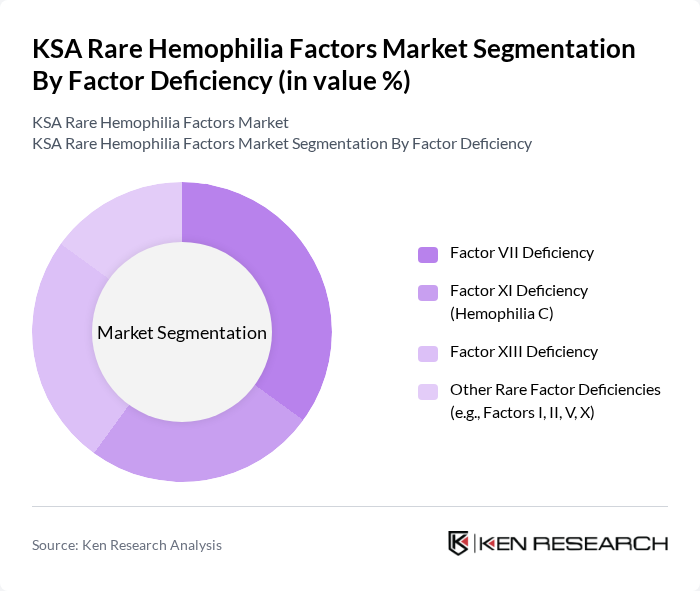
KSA Rare Hemophilia Factors Market Segmentation
By Factor Deficiency:The market is segmented based on the type of factor deficiency, which includes Factor VII Deficiency, Factor XI Deficiency (Hemophilia C), Factor XIII Deficiency, and Other Rare Factor Deficiencies (e.g., Factors I, II, V, X). Among these, Factor VII Deficiency is the largest contributor in terms of revenue share in Saudi Arabia, supported by established use of recombinant activated factor VII and factor VII concentrates for both congenital deficiency and inhibitor patients. The growing number of patients diagnosed through targeted coagulation testing and family screening has increased demand for these products, positioning Factor VII Deficiency as a key segment within the national rare hemophilia factors market.
By Product Type:The market is also segmented by product type, which includes Plasma-Derived Factor Concentrates, Recombinant Factor Concentrates, Bypassing Agents and Adjunct Therapies, and Others. Recombinant Factor Concentrates account for a growing share of the market due to their favorable safety profile regarding viral transmission, consistent batch quality, and suitability for prophylaxis programs. Increasing physician preference for recombinant and next-generation products, supported by national treatment guidelines and tender procurement, is driving the uptake of these therapies relative to traditional plasma-derived alternatives.

KSA Rare Hemophilia Factors Market Competitive Landscape
The KSA Rare Hemophilia Factors Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Bayer AG, CSL Behring AG, Novo Nordisk A/S, Pfizer Inc., Grifols, S.A., Octapharma AG, Sanofi S.A., Bio Products Laboratory Limited (BPL), BioMarin Pharmaceutical Inc., Swedish Orphan Biovitrum AB (Sobi), Kedrion Biopharma Inc., LFB S.A., Biogen Inc., Baxalta (a Takeda company) contribute to innovation, geographic expansion, and service delivery in this space.
KSA Rare Hemophilia Factors Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Rare Hemophilia Disorders:The Kingdom of Saudi Arabia (KSA) has reported approximately 1,500 cases of rare hemophilia disorders, with an annual increase of 5% in diagnoses. This rise is attributed to improved diagnostic capabilities and heightened awareness among healthcare professionals. The World Health Organization (WHO) estimates that around 1 in 10,000 individuals are affected by hemophilia, indicating a significant patient population that drives demand for specialized treatment options and healthcare services.
- Advancements in Treatment Options:The KSA healthcare sector has seen substantial investments in innovative therapies for rare hemophilia factors, with over 20 new treatment modalities introduced in the last five years. The Ministry of Health allocated SAR 1.5 billion (approximately USD 400 million) for rare disease research and treatment in future. These advancements not only improve patient outcomes but also stimulate market growth by attracting both local and international pharmaceutical companies to invest in the region.
- Rising Awareness and Diagnosis Rates:Increased public and professional awareness regarding rare hemophilia disorders has led to a 35% rise in diagnosis rates over the past three years. Educational campaigns funded by the government and NGOs have reached over 2.5 million people, significantly enhancing understanding of these conditions. This growing awareness is crucial for early diagnosis and treatment, ultimately contributing to a more robust market for rare hemophilia factors in KSA.
Market Challenges
- High Cost of Treatment:The average annual treatment cost for patients with rare hemophilia disorders in KSA can exceed SAR 600,000 (approximately USD 160,000). This high financial burden limits access to necessary therapies for many patients, particularly those without adequate insurance coverage. The economic implications of such costs pose a significant challenge to market growth, as many patients may forgo treatment due to affordability issues.
- Limited Availability of Specialized Healthcare:KSA currently has only 20 specialized hemophilia treatment centers, serving a population of over 40 million. This translates to approximately 1 center for every 2 million people, leading to significant accessibility issues for patients. The lack of specialized healthcare providers and facilities hampers timely diagnosis and treatment, creating a barrier to market expansion and patient care in the region.
KSA Rare Hemophilia Factors Market Future Outlook
The future of the KSA rare hemophilia factors market appears promising, driven by ongoing advancements in treatment technologies and increased government support for rare disease initiatives. As healthcare infrastructure expands, more patients will gain access to specialized care. Additionally, the integration of telemedicine and digital health solutions is expected to enhance patient monitoring and education, further improving treatment adherence and outcomes. These trends indicate a positive trajectory for the market, fostering innovation and collaboration among stakeholders.
Market Opportunities
- Development of Innovative Therapies:The KSA market presents significant opportunities for the development of innovative therapies, particularly gene therapies and personalized medicine. With an estimated SAR 400 million (approximately USD 107 million) allocated for R&D in future, pharmaceutical companies can explore novel treatment options that cater to the unique needs of hemophilia patients, potentially transforming care standards.
- Expansion of Healthcare Infrastructure:The Saudi Vision 2030 initiative aims to enhance healthcare infrastructure, with plans to establish 60 new healthcare facilities by future. This expansion will improve access to specialized care for rare hemophilia disorders, creating a favorable environment for market growth. Enhanced facilities will also facilitate better patient education and support, further driving demand for rare hemophilia factors.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Factor Deficiency | Factor VII Deficiency Factor XI Deficiency (Hemophilia C) Factor XIII Deficiency Other Rare Factor Deficiencies (e.g., Factors I, II, V, X) |
| By Product Type | Plasma-Derived Factor Concentrates Recombinant Factor Concentrates Bypassing Agents and Adjunct Therapies Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Distribution Channel | Hospital Pharmacies Specialty Pharmacies Tender-Based Institutional Supply Others |
| By Treatment Setting | Inpatient Outpatient Home Care Others |
| By Geographic Distribution | Central Region Eastern Region Western Region Southern Region Others |
| By Treatment Type | Prophylactic Treatment On-Demand Treatment Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers of Rare Hemophilia Factors
Distributors and Wholesalers of Pharmaceutical Products
Healthcare Providers and Hospitals
Pharmaceutical Supply Chain Managers
Industry Associations (e.g., Saudi Hemophilia Society)
Health Insurance Companies
Players Mentioned in the Report:
Takeda Pharmaceutical Company Limited
Bayer AG
CSL Behring AG
Novo Nordisk A/S
Pfizer Inc.
Grifols, S.A.
Octapharma AG
Sanofi S.A.
Bio Products Laboratory Limited (BPL)
BioMarin Pharmaceutical Inc.
Swedish Orphan Biovitrum AB (Sobi)
Kedrion Biopharma Inc.
LFB S.A.
Biogen Inc.
Baxalta (a Takeda company)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. KSA Rare Hemophilia Factors Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 KSA Rare Hemophilia Factors Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. KSA Rare Hemophilia Factors Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of rare hemophilia disorders
3.1.2 Advancements in treatment options
3.1.3 Rising awareness and diagnosis rates
3.1.4 Government support and funding for rare diseases
3.2 Market Challenges
3.2.1 High cost of treatment
3.2.2 Limited availability of specialized healthcare
3.2.3 Regulatory hurdles
3.2.4 Lack of patient education and support
3.3 Market Opportunities
3.3.1 Development of innovative therapies
3.3.2 Expansion of healthcare infrastructure
3.3.3 Collaborations with pharmaceutical companies
3.3.4 Increased investment in research and development
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Growth of telemedicine and remote monitoring
3.4.3 Emphasis on patient-centric care
3.4.4 Integration of digital health solutions
3.5 Government Regulation
3.5.1 Regulatory frameworks for rare disease treatments
3.5.2 Pricing and reimbursement policies
3.5.3 Guidelines for clinical trials
3.5.4 Quality control standards for pharmaceuticals
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. KSA Rare Hemophilia Factors Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. KSA Rare Hemophilia Factors Market Segmentation
8.1 By Factor Deficiency
8.1.1 Factor VII Deficiency
8.1.2 Factor XI Deficiency (Hemophilia C)
8.1.3 Factor XIII Deficiency
8.1.4 Other Rare Factor Deficiencies (e.g., Factors I, II, V, X)
8.2 By Product Type
8.2.1 Plasma-Derived Factor Concentrates
8.2.2 Recombinant Factor Concentrates
8.2.3 Bypassing Agents and Adjunct Therapies
8.2.4 Others
8.3 By Patient Demographics
8.3.1 Pediatric Patients
8.3.2 Adult Patients
8.3.3 Geriatric Patients
8.3.4 Others
8.4 By Distribution Channel
8.4.1 Hospital Pharmacies
8.4.2 Specialty Pharmacies
8.4.3 Tender-Based Institutional Supply
8.4.4 Others
8.5 By Treatment Setting
8.5.1 Inpatient
8.5.2 Outpatient
8.5.3 Home Care
8.5.4 Others
8.6 By Geographic Distribution
8.6.1 Central Region
8.6.2 Eastern Region
8.6.3 Western Region
8.6.4 Southern Region
8.6.5 Others
8.7 By Treatment Type
8.7.1 Prophylactic Treatment
8.7.2 On-Demand Treatment
8.7.3 Others
9. KSA Rare Hemophilia Factors Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Presence in KSA Rare Hemophilia Factors Portfolio (Yes/No, breadth of SKUs)
9.2.3 Revenue from Rare Hemophilia Factors in KSA
9.2.4 CAGR of Rare Hemophilia Factors Revenue in KSA, 2019-2024
9.2.5 Share of Rare Hemophilia Factors in Company Hematology Revenue (KSA)
9.2.6 Market Share in KSA Rare Hemophilia Factors Market
9.2.7 Average Tender Win Rate with KSA Public Buyers
9.2.8 Number of Approved SKUs by Factor Deficiency (VII, XI, XIII, Others)
9.2.9 R&D Investment in Rare Hemophilia Factors / Total R&D (%)
9.2.10 Coverage of KSA Regions (no. of hospitals/centers served)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Takeda Pharmaceutical Company Limited
9.5.2 Bayer AG
9.5.3 CSL Behring AG
9.5.4 Novo Nordisk A/S
9.5.5 Pfizer Inc.
9.5.6 Grifols, S.A.
9.5.7 Octapharma AG
9.5.8 Sanofi S.A.
9.5.9 Bio Products Laboratory Limited (BPL)
9.5.10 BioMarin Pharmaceutical Inc.
9.5.11 Swedish Orphan Biovitrum AB (Sobi)
9.5.12 Kedrion Biopharma Inc.
9.5.13 LFB S.A.
9.5.14 Biogen Inc.
9.5.15 Baxalta (a Takeda company)
10. KSA Rare Hemophilia Factors Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of National Guard Health Affairs
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Research and Development Funding
10.2.3 Public-Private Partnerships
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Specialty Clinics
10.3.3 Home Healthcare Providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training and Support Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcome Improvements
10.5.3 Scalability of Solutions
10.5.4 Others
11. KSA Rare Hemophilia Factors Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Tracking
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from health organizations and medical journals on rare hemophilia factors
- Review of market data from government health departments and regulatory bodies in KSA
- Examination of industry publications and white papers focusing on hemophilia treatment advancements
Primary Research
- Interviews with hematologists and specialists in hemophilia treatment across KSA
- Surveys with hospital administrators regarding procurement practices for rare hemophilia factors
- Focus groups with patients and caregivers to understand treatment experiences and needs
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and patient feedback
- Triangulation of data from clinical studies, market reports, and expert opinions
- Sanity checks through consultations with a panel of medical professionals and industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national health expenditure on rare diseases
- Segmentation of the market by treatment type, including factor replacement therapies and gene therapies
- Incorporation of demographic data to assess the prevalence of hemophilia in KSA
Bottom-up Modeling
- Collection of sales data from pharmaceutical distributors and manufacturers of hemophilia factors
- Estimation of treatment costs based on pricing models from healthcare providers
- Volume estimates derived from patient population data and treatment regimens
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as healthcare policy changes and advancements in treatment
- Scenario modeling based on potential market disruptions, including new therapies and regulatory changes
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hematology Clinics | 90 | Hematologists, Clinic Managers |
| Hospitals with Hemophilia Treatment Centers | 70 | Medical Directors, Procurement Officers |
| Patient Advocacy Groups | 50 | Patient Representatives, Caregivers |
| Pharmaceutical Distributors | 60 | Sales Managers, Product Managers |
| Health Insurance Providers | 40 | Policy Analysts, Claims Managers |
Frequently Asked Questions
What is the current value of the KSA Rare Hemophilia Factors Market?
The KSA Rare Hemophilia Factors Market is valued at approximately USD 2.3 million, reflecting a five-year historical analysis. This growth is driven by increased identification of rare hemophilia factor deficiencies and rising healthcare expenditures in the region.