About the Report
Base Year 2024Kuwait Medical Device Regulatory Affairs Market Overview
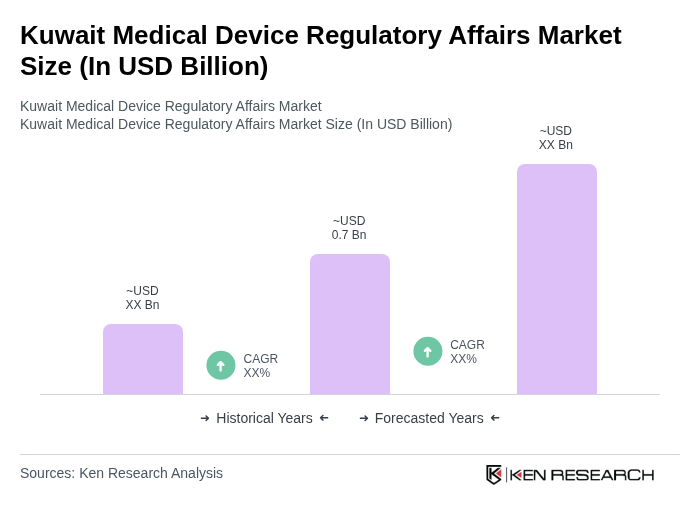
- The Kuwait Medical Device Regulatory Affairs Market is valued at USD 0.7 billion, based on a five-year historical analysis. This growth is primarily driven by substantial government investment in healthcare infrastructure, rising prevalence of chronic diseases, and expansion of private healthcare facilities. The market is further supported by robust adoption of advanced diagnostic and monitoring equipment, reflecting sustained demand from both public and private sectors. Major drivers include increased healthcare expenditure, technological advancements, and a rising demand for quality healthcare services, particularly as the aging population and chronic disease rates continue to climb.
- Kuwait City is the dominant hub in the Kuwait Medical Device Regulatory Affairs Market due to its advanced healthcare infrastructure and concentration of medical facilities. The presence of major hospitals and healthcare institutions in the capital facilitates the adoption of innovative medical devices. Additionally, government-led healthcare megaprojects and strategic development initiatives, such as those under Kuwait Vision 2035, further strengthen Kuwait City’s position as the market leader. Al Jahra and Hawalli regions also exhibit strong uptake due to flourishing private clinics and laboratories.
- The Kuwait Ministry of Health regulates medical device registration under the “Medical Devices Registration Guidelines, 2019” issued by the Ministry of Health, Kuwait. All medical devices must undergo a comprehensive evaluation process before market entry, including detailed assessment of safety, efficacy, and quality. The guidelines require submission of technical documentation, clinical data, and conformity certificates, and mandate that only compliant products are made available to healthcare providers and patients.
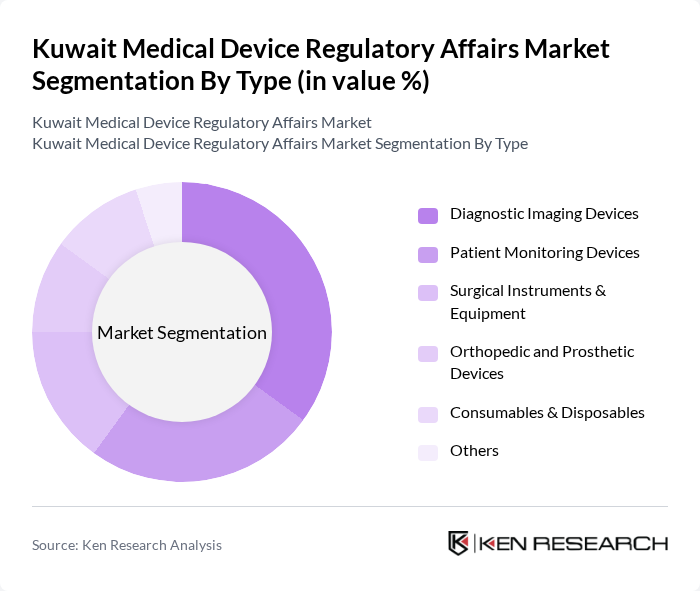
Kuwait Medical Device Regulatory Affairs Market Segmentation
By Type:The market is segmented into various types of medical devices, including diagnostic imaging devices, patient monitoring devices, surgical instruments and equipment, orthopedic and prosthetic devices, consumables and disposables, and others. Diagnostic imaging devices hold the largest share due to their critical role in early disease detection and management, supported by expanded utilization of MRI, CT scans, and ultrasound systems in both public and private hospitals. Patient monitoring devices are gaining traction as demand for remote and home-based monitoring solutions increases, driven by healthcare digitization and consumer awareness.
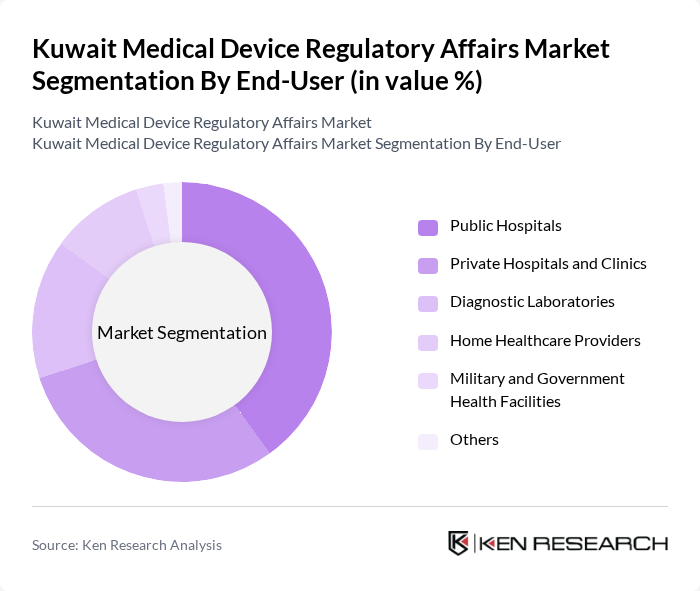
By End-User:The end-user segmentation includes public hospitals, private hospitals and clinics, diagnostic laboratories, home healthcare providers, military and government health facilities, and others. Public hospitals command the largest segment due to Kuwait’s considerable public healthcare infrastructure, significant capital deployment under national development plans, and comprehensive procurement programs. Private hospitals and clinics are also significant contributors, focusing on advanced medical technologies and specialized care. Diagnostic laboratories and home healthcare providers are expanding rapidly, reflecting increased demand for outpatient and remote care solutions.
Kuwait Medical Device Regulatory Affairs Market Competitive Landscape
The Kuwait Medical Device Regulatory Affairs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Kuwait Medical Devices Company, Al-Dar Medical Supplies, Warba Medical Supplies Company, Al-Mahaba Medical Company, Medtronic Middle East (Kuwait Operations), Siemens Healthineers Kuwait, GE Healthcare Kuwait, Philips Healthcare Kuwait, Johnson & Johnson Medical Devices Kuwait, Abbott Laboratories Kuwait, B. Braun Medical Kuwait, Stryker Kuwait, Boston Scientific Kuwait, Zimmer Biomet Kuwait, 3M Health Care Kuwait contribute to innovation, geographic expansion, and service delivery in this space.
Kuwait Medical Device Regulatory Affairs Market Industry Analysis
Growth Drivers
- Increasing Healthcare Expenditure:Kuwait's healthcare expenditure is projected to reach approximately KWD 2.6 billion in future, reflecting a significant increase from KWD 2.2 billion in future. This growth is driven by the government's commitment to enhancing healthcare services and infrastructure, which directly impacts the medical device sector. The rise in spending facilitates the acquisition of advanced medical technologies, thereby fostering a conducive environment for regulatory affairs in the medical device market.
- Rising Demand for Advanced Medical Technologies:The demand for advanced medical technologies in Kuwait is expected to surge, with an estimated increase in imports of medical devices reaching KWD 1.2 billion by future. This demand is fueled by the growing prevalence of chronic diseases and the need for innovative healthcare solutions. As healthcare providers seek to improve patient outcomes, the regulatory framework must adapt to accommodate these advancements, ensuring safety and efficacy in medical devices.
- Government Initiatives for Healthcare Improvement:The Kuwaiti government has launched several initiatives aimed at improving healthcare quality, with a budget allocation of KWD 350 million for health sector reforms in future. These initiatives include the establishment of new healthcare facilities and the enhancement of existing ones, which will increase the demand for compliant medical devices. Such government support is crucial for streamlining regulatory processes and encouraging investment in the medical device sector.
Market Challenges
- Complex Regulatory Processes:The regulatory landscape for medical devices in Kuwait is characterized by complex processes that can delay product approvals. The average time for device registration can exceed six months, which poses a significant challenge for manufacturers. This complexity is often attributed to the need for comprehensive documentation and compliance with both local and international standards, creating barriers for new entrants in the market.
- High Import Dependency:Kuwait's medical device market is heavily reliant on imports, with over 90% of devices sourced from foreign manufacturers. This dependency creates vulnerabilities in the supply chain, particularly in times of global disruptions. Additionally, the reliance on imports can lead to increased costs and longer lead times for healthcare providers, complicating the regulatory landscape as local adaptation becomes necessary for compliance.
Kuwait Medical Device Regulatory Affairs Market Future Outlook
The future of the Kuwait medical device regulatory affairs market is poised for transformation, driven by technological advancements and evolving healthcare needs. As the government continues to invest in healthcare infrastructure, the regulatory framework will likely adapt to facilitate faster approvals for innovative devices. Furthermore, the integration of digital health solutions and artificial intelligence in medical devices will necessitate updated regulations, ensuring safety while promoting innovation in the sector.
Market Opportunities
- Expansion of Telemedicine Services:The rise of telemedicine in Kuwait presents a significant opportunity for medical device manufacturers. With an estimated 35% increase in telehealth consultations expected in future, there is a growing need for devices that support remote patient monitoring and diagnostics, creating a favorable environment for regulatory advancements in this area.
- Development of Local Manufacturing Facilities:The Kuwaiti government is encouraging the establishment of local manufacturing facilities for medical devices, with a target of increasing local production by 30% by future. This initiative aims to reduce import dependency and enhance compliance with local regulations, providing a strategic opportunity for companies to invest in manufacturing capabilities within the country.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Diagnostic Imaging Devices Patient Monitoring Devices Surgical Instruments & Equipment Orthopedic and Prosthetic Devices Consumables & Disposables Others |
| By End-User | Public Hospitals Private Hospitals and Clinics Diagnostic Laboratories Home Healthcare Providers Military and Government Health Facilities Others |
| By Regulatory Classification | Class I Devices Class II Devices Class III Devices Others |
| By Distribution Channel | Direct Purchase Tender-Based Procurement Distributors Online Sales Others |
| By Application/Therapeutic Area | Cardiology Orthopedics Neurology Ophthalmology Diagnostic Imaging In Vitro Diagnostics (IVD) Dental Others |
| By Technology | Wearable Devices Implantable Devices Point-of-Care Testing Others |
| By Policy Support | Government Subsidies Tax Incentives Regulatory Support Programs Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Kuwait Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Medical Device Importers
Healthcare Providers and Hospitals
Industry Associations (e.g., Kuwait Medical Association)
Financial Institutions
Players Mentioned in the Report:
Kuwait Medical Devices Company
Al-Dar Medical Supplies
Warba Medical Supplies Company
Al-Mahaba Medical Company
Medtronic Middle East (Kuwait Operations)
Siemens Healthineers Kuwait
GE Healthcare Kuwait
Philips Healthcare Kuwait
Johnson & Johnson Medical Devices Kuwait
Abbott Laboratories Kuwait
B. Braun Medical Kuwait
Stryker Kuwait
Boston Scientific Kuwait
Zimmer Biomet Kuwait
3M Health Care Kuwait
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Kuwait Medical Device Regulatory Affairs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Kuwait Medical Device Regulatory Affairs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Kuwait Medical Device Regulatory Affairs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing healthcare expenditure
3.1.2 Rising demand for advanced medical technologies
3.1.3 Government initiatives for healthcare improvement
3.1.4 Growing aging population
3.2 Market Challenges
3.2.1 Complex regulatory processes
3.2.2 Limited local manufacturing capabilities
3.2.3 High import dependency
3.2.4 Stringent compliance requirements
3.3 Market Opportunities
3.3.1 Expansion of telemedicine services
3.3.2 Development of local manufacturing facilities
3.3.3 Partnerships with international firms
3.3.4 Investment in health technology startups
3.4 Market Trends
3.4.1 Shift towards digital health solutions
3.4.2 Increased focus on patient-centric devices
3.4.3 Adoption of AI in medical devices
3.4.4 Growing emphasis on sustainability in manufacturing
3.5 Government Regulation
3.5.1 New medical device registration guidelines
3.5.2 Enhanced post-market surveillance requirements
3.5.3 Streamlined approval processes for innovative devices
3.5.4 Compliance with international standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Kuwait Medical Device Regulatory Affairs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Kuwait Medical Device Regulatory Affairs Market Segmentation
8.1 By Type
8.1.1 Diagnostic Imaging Devices
8.1.2 Patient Monitoring Devices
8.1.3 Surgical Instruments & Equipment
8.1.4 Orthopedic and Prosthetic Devices
8.1.5 Consumables & Disposables
8.1.6 Others
8.2 By End-User
8.2.1 Public Hospitals
8.2.2 Private Hospitals and Clinics
8.2.3 Diagnostic Laboratories
8.2.4 Home Healthcare Providers
8.2.5 Military and Government Health Facilities
8.2.6 Others
8.3 By Regulatory Classification
8.3.1 Class I Devices
8.3.2 Class II Devices
8.3.3 Class III Devices
8.3.4 Others
8.4 By Distribution Channel
8.4.1 Direct Purchase
8.4.2 Tender-Based Procurement
8.4.3 Distributors
8.4.4 Online Sales
8.4.5 Others
8.5 By Application/Therapeutic Area
8.5.1 Cardiology
8.5.2 Orthopedics
8.5.3 Neurology
8.5.4 Ophthalmology
8.5.5 Diagnostic Imaging
8.5.6 In Vitro Diagnostics (IVD)
8.5.7 Dental
8.5.8 Others
8.6 By Technology
8.6.1 Wearable Devices
8.6.2 Implantable Devices
8.6.3 Point-of-Care Testing
8.6.4 Others
8.7 By Policy Support
8.7.1 Government Subsidies
8.7.2 Tax Incentives
8.7.3 Regulatory Support Programs
8.7.4 Others
9. Kuwait Medical Device Regulatory Affairs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Regulatory Approval Success Rate
9.2.3 Average Regulatory Approval Time (months)
9.2.4 Number of Product Registrations in Kuwait
9.2.5 Local Market Penetration Rate
9.2.6 Compliance Audit Pass Rate
9.2.7 Number of Regulatory Filings per Year
9.2.8 Frequency of Post-Market Surveillance Activities
9.2.9 Number of Regulatory Staff in Kuwait
9.2.10 Revenue from Kuwait Medical Device Market
9.2.11 Market Share Percentage
9.2.12 Customer Satisfaction Score (Regulatory Services)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Kuwait Medical Devices Company
9.5.2 Al-Dar Medical Supplies
9.5.3 Warba Medical Supplies Company
9.5.4 Al-Mahaba Medical Company
9.5.5 Medtronic Middle East (Kuwait Operations)
9.5.6 Siemens Healthineers Kuwait
9.5.7 GE Healthcare Kuwait
9.5.8 Philips Healthcare Kuwait
9.5.9 Johnson & Johnson Medical Devices Kuwait
9.5.10 Abbott Laboratories Kuwait
9.5.11 B. Braun Medical Kuwait
9.5.12 Stryker Kuwait
9.5.13 Boston Scientific Kuwait
9.5.14 Zimmer Biomet Kuwait
9.5.15 3M Health Care Kuwait
10. Kuwait Medical Device Regulatory Affairs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Defense
10.1.3 Ministry of Education
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.2.3 Technology Upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home Healthcare Providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Development
10.5.3 Long-term Sustainability Considerations
10.5.4 Others
11. Kuwait Medical Device Regulatory Affairs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of Kuwait's healthcare regulations and medical device laws from the Ministry of Health
- Review of market reports and publications from local health authorities and international organizations
- Examination of trade statistics and import/export data related to medical devices in Kuwait
Primary Research
- Interviews with regulatory affairs professionals in medical device companies operating in Kuwait
- Surveys targeting healthcare practitioners and hospital procurement managers
- Field interviews with compliance officers and quality assurance managers in the medical device sector
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government reports and industry publications
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panel discussions with industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on Kuwait's healthcare expenditure and medical device imports
- Segmentation of the market by device type, including diagnostic, therapeutic, and surgical devices
- Incorporation of government healthcare initiatives and funding allocations for medical technology
Bottom-up Modeling
- Collection of sales data from leading medical device distributors and manufacturers in Kuwait
- Estimation of market share based on product categories and sales volume
- Cost analysis of medical devices, including pricing strategies and reimbursement rates
Forecasting & Scenario Analysis
- Development of growth projections based on historical market trends and healthcare policy changes
- Scenario modeling considering factors such as population growth, aging demographics, and technological advancements
- Baseline, optimistic, and pessimistic forecasts through 2030, reflecting potential regulatory impacts
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Regulatory Compliance in Medical Devices | 50 | Regulatory Affairs Managers, Compliance Officers |
| Healthcare Procurement Strategies | 40 | Procurement Managers, Hospital Administrators |
| Market Trends in Diagnostic Devices | 40 | Product Managers, Medical Device Sales Representatives |
| Impact of Technology on Medical Devices | 40 | R&D Managers, Innovation Officers |
| Healthcare Policy and Medical Device Regulation | 40 | Health Policy Analysts, Medical Device Consultants |
Frequently Asked Questions
What is the current value of the Kuwait Medical Device Regulatory Affairs Market?
The Kuwait Medical Device Regulatory Affairs Market is valued at approximately USD 0.7 billion, reflecting growth driven by government investment in healthcare infrastructure, rising chronic disease prevalence, and the expansion of private healthcare facilities.