Region:Middle East
Author(s):Rebecca
Product Code:KRAD5004
Pages:91
Published On:December 2025
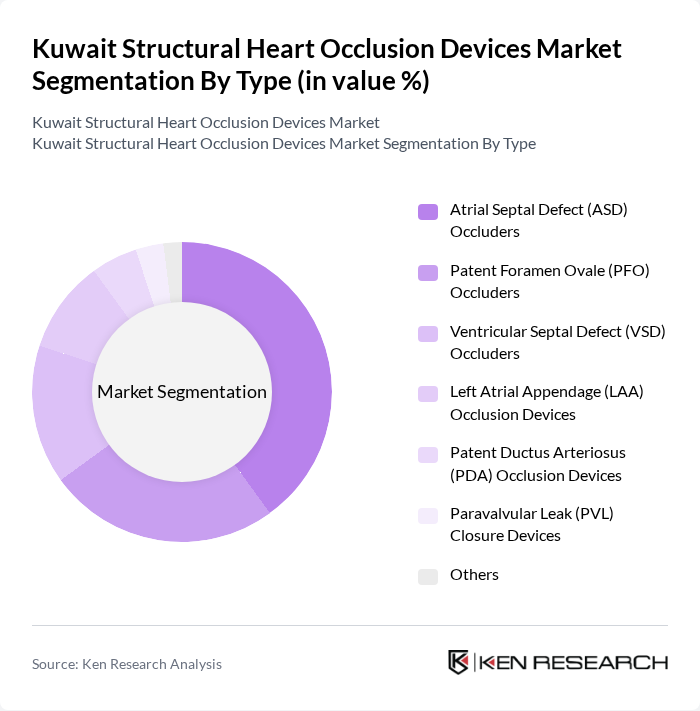
By Type:The market is segmented into various types of occlusion devices, including Atrial Septal Defect (ASD) Occluders, Patent Foramen Ovale (PFO) Occluders, Ventricular Septal Defect (VSD) Occluders, Left Atrial Appendage (LAA) Occlusion Devices, Patent Ductus Arteriosus (PDA) Occlusion Devices, Paravalvular Leak (PVL) Closure Devices, and Others. This segmentation is aligned with global structural heart occlusion device categorization, where ASD, PFO, VSD and LAA devices represent the core product groups. Among these, ASD Occluders are leading the market due to their widespread use in treating congenital atrial septal defects, which remain among the most frequently encountered congenital heart lesions in Gulf and Middle East pediatric and adult congenital programs. The increasing number of catheter-based ASD and PFO closure procedures, together with growing awareness of stroke prevention and embolic risk reduction among cardiologists, neurologists, and patients, contributes to their dominance in Kuwait.
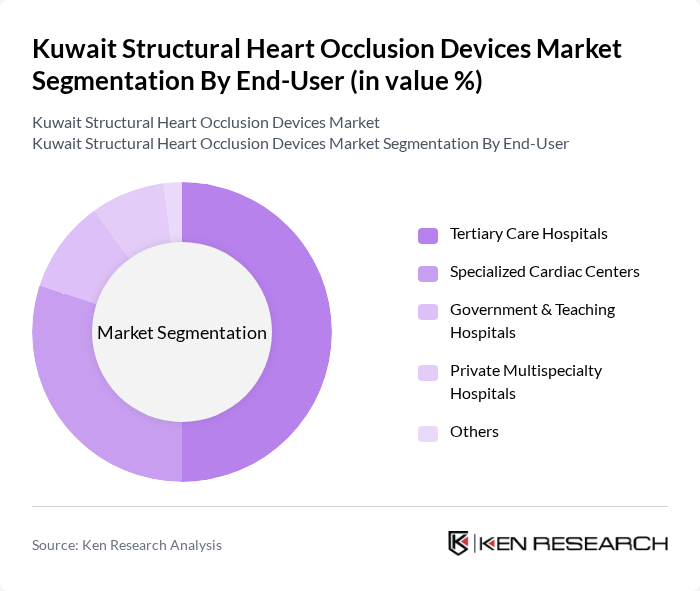
By End-User:The end-user segmentation includes Tertiary Care Hospitals, Specialized Cardiac Centers, Government & Teaching Hospitals, Private Multispecialty Hospitals, and Others. This structure is consistent with global and regional practice patterns in structural heart and occlusion procedures, where hospitals dominate usage. Tertiary Care Hospitals dominate this segment due to their advanced catheterization laboratories, hybrid operating rooms, and multidisciplinary heart teams capable of performing complex transcatheter ASD, PFO, VSD, LAA, PDA, and PVL closure procedures. The increasing number of patients requiring structural heart interventions in these hospitals, combined with centralized referral pathways from secondary and private facilities, drives the demand for occlusion devices, making them the leading end-user in the market.
The Kuwait Structural Heart Occlusion Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories (including legacy St. Jude Medical portfolio), Boston Scientific Corporation, Medtronic plc, Edwards Lifesciences Corporation, W. L. Gore & Associates, Inc. (Gore Medical), Occlutech Holding AG, Lifetech Scientific Corporation, Lepu Medical Technology (Beijing) Co., Ltd., Biotronik SE & Co. KG, LivaNova PLC, Cardia, Inc. (NobleStitch EL System), Cook Medical LLC, Terumo Corporation, Merit Medical Systems, Inc., Philips Healthcare (Imaging & Navigation Support for Structural Heart Interventions) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the structural heart occlusion devices market in Kuwait appears promising, driven by technological advancements and an increasing focus on patient-centric care. As healthcare providers adopt innovative solutions, the integration of artificial intelligence and machine learning in device development is expected to enhance treatment outcomes. Additionally, the expansion of healthcare infrastructure will facilitate better access to these devices, ultimately improving patient care and outcomes in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Atrial Septal Defect (ASD) Occluders Patent Foramen Ovale (PFO) Occluders Ventricular Septal Defect (VSD) Occluders Left Atrial Appendage (LAA) Occlusion Devices Patent Ductus Arteriosus (PDA) Occlusion Devices Paravalvular Leak (PVL) Closure Devices Others |
| By End-User | Tertiary Care Hospitals Specialized Cardiac Centers Government & Teaching Hospitals Private Multispecialty Hospitals Others |
| By Application | Congenital Heart Defect Closure (ASD, VSD, PDA) PFO Closure for Secondary Stroke Prevention LAA Occlusion for Atrial Fibrillation–Related Stroke Prevention Paravalvular Leak Closure Others |
| By Distribution Channel | Direct Sales to Hospitals Local Medical Device Distributors Group Purchasing Organizations (GPOs) / Tender-based Procurement Others |
| By Region | Capital Governorate (Kuwait City) Hawalli Governorate Al Ahmadi Governorate Farwaniya & Al Jahra Governorates |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients High-Risk / Comorbid Patients |
| By Technology | Nitinol-Based Self-Expanding Occluders Bioabsorbable Occluders Hybrid / Composite Material Occluders Imaging-Guided Delivery Systems |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologists in Private Practice | 120 | Interventional Cardiologists, Electrophysiologists |
| Hospital Procurement Departments | 90 | Procurement Managers, Supply Chain Coordinators |
| Medical Device Distributors | 70 | Sales Managers, Product Specialists |
| Healthcare Policy Makers | 60 | Health Economists, Regulatory Affairs Specialists |
| Clinical Research Coordinators | 80 | Clinical Trial Managers, Research Nurses |
The Kuwait Structural Heart Occlusion Devices Market is valued at approximately USD 45 million, reflecting a five-year historical analysis and triangulation from regional cardiology device spending and heart-related device expenditures in Kuwait.