About the Report
Base Year 2024Middle East Biological Safety Testing Products Services Market Overview
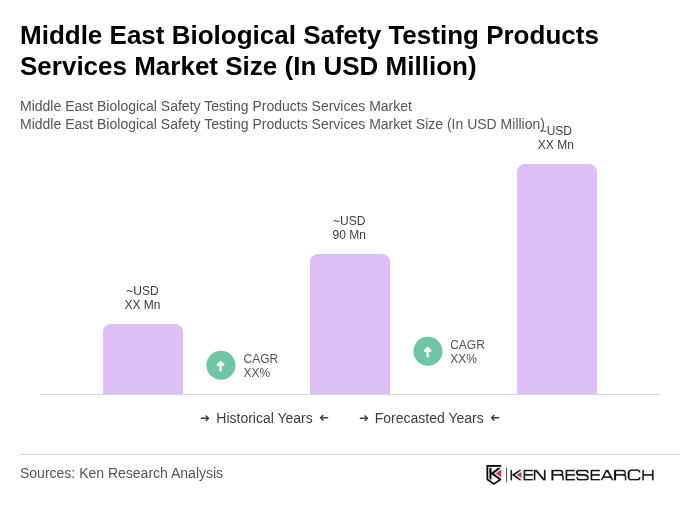
- The Middle East Biological Safety Testing Products Services Market is valued at USD 90 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for safety testing in biopharmaceuticals and biotechnology sectors, alongside rising regulatory requirements for product safety and efficacy. The market is also supported by advancements in testing technologies and methodologies, such as rapid microbial detection and automation, which enhance the reliability and speed of testing processes. The expansion of vaccine, biosimilar, and gene therapy pipelines in the region further accelerates market demand .
- Key players in this market include countries like the United Arab Emirates, Saudi Arabia, and Israel. These nations dominate the market due to their robust healthcare infrastructure, significant investments in biotechnology and pharmaceuticals, and a growing number of research institutions. The presence of leading global companies and a favorable regulatory environment further contribute to their market leadership .
- In 2023, the UAE government implemented new regulations mandating comprehensive biological safety testing for all biopharmaceutical products before market approval. This regulation, under the “Ministerial Resolution No. 28 of 2023 on the Regulation of Biological Products” issued by the UAE Ministry of Health and Prevention, requires all biopharmaceutical manufacturers and importers to conduct validated safety testing—including sterility, endotoxin, and viral clearance—prior to product registration and market entry. The regulation aligns UAE standards with international guidelines and has significantly increased demand for biological safety testing services across the region .
Middle East Biological Safety Testing Products Services Market Segmentation
By Type:The market is segmented into various types of biological safety testing services, which include Bioburden Testing Services, Endotoxin Testing Services, Sterility Testing Services, Viral Clearance and Validation Services, Cell Line Authentication and Characterization Tests, Residual Host Contamination Testing Services, and Adventitious Agent Detection Testing Services. Each of these services plays a crucial role in ensuring the safety and efficacy of biopharmaceutical products. Bioburden testing services represent the largest revenue share, while cell line authentication and characterization tests are among the fastest-growing segments due to increased focus on cell and gene therapies .

By End-User:The end-user segmentation includes Biopharmaceutical Companies, Pharmaceutical Companies, Biotechnology Firms, Academic and Research Institutions, and Contract Research Organizations (CROs). Each of these segments has unique requirements for biological safety testing, driven by their specific product development processes and regulatory compliance needs. Biopharmaceutical and pharmaceutical companies constitute the largest end-user segments, reflecting the region’s growing biologics and vaccine manufacturing capacity .

Middle East Biological Safety Testing Products Services Market Competitive Landscape
The Middle East Biological Safety Testing Products Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Charles River Laboratories, Eurofins Scientific, SGS S.A., Intertek Group plc, BioReliance (Merck KGaA), WuXi AppTec, Labcorp Drug Development, Medpace Holdings, Inc., Q2 Solutions, Nelson Labs (Sotera Health), Viroclinics Xplore, BioAgilytix Labs, Pacific BioLabs, NAMSA (North American Science Associates), and Toxikon Corporation contribute to innovation, geographic expansion, and service delivery in this space .
Middle East Biological Safety Testing Products Services Market Industry Analysis
Growth Drivers
- Increasing Demand for Safety Testing in Pharmaceuticals:The Middle East pharmaceutical market is projected to reach $36 billion in current market conditions, driven by a growing population and increased healthcare spending. This surge in demand for pharmaceuticals necessitates rigorous biological safety testing to ensure product efficacy and safety. The region's focus on enhancing healthcare quality and patient safety further propels the need for comprehensive testing services, creating a robust market for biological safety testing products and services.
- Rising Awareness of Biological Hazards:With the increasing incidence of infectious diseases and bioterrorism threats, awareness of biological hazards is on the rise in the Middle East. Reports indicate that the region experienced a 15% increase in reported infectious disease cases from 2020 to 2023. This heightened awareness drives demand for biological safety testing, as organizations seek to mitigate risks associated with biological agents, thereby fostering market growth for testing services and products.
- Stringent Regulatory Requirements:Regulatory bodies in the Middle East are implementing stricter guidelines for biological safety testing, particularly in pharmaceuticals and biotechnology. For instance, the UAE's Ministry of Health and Prevention has introduced new regulations that require comprehensive safety testing for all new drug approvals. This regulatory landscape compels companies to invest in biological safety testing services, thereby expanding the market and ensuring compliance with international safety standards.
Market Challenges
- High Costs of Testing Services:The biological safety testing market faces significant challenges due to the high costs associated with testing services. For instance, the average cost of comprehensive biological testing can range from $10,000 to $50,000 per project, depending on the complexity and scope. These costs can deter smaller companies and startups from accessing necessary testing services, limiting market growth and innovation in the region.
- Limited Availability of Skilled Professionals:The Middle East is experiencing a shortage of skilled professionals in the biological safety testing sector. According to the World Health Organization, the region has only 1.5 healthcare workers per 1,000 population, significantly below the global average of 2.5. This shortage hampers the ability of testing facilities to meet increasing demand, posing a challenge to the growth of the biological safety testing market in the region.
Middle East Biological Safety Testing Products Services Market Future Outlook
The future of the biological safety testing market in the Middle East appears promising, driven by advancements in technology and increasing investments in healthcare infrastructure. As biopharmaceutical companies expand, the demand for innovative testing solutions will rise. Additionally, the integration of artificial intelligence in testing processes is expected to enhance efficiency and accuracy, further propelling market growth. Collaborative research initiatives will also play a crucial role in addressing emerging biological threats and improving safety standards across the region.
Market Opportunities
- Expansion of Biopharmaceutical Companies:The biopharmaceutical sector in the Middle East is projected to grow significantly, with investments expected to exceed $10 billion in future. This expansion presents a substantial opportunity for biological safety testing services, as new products will require rigorous testing to meet regulatory standards and ensure patient safety.
- Technological Advancements in Testing Methods:Innovations in testing technologies, such as rapid testing and automation, are transforming the biological safety testing landscape. The adoption of these advanced methods can reduce testing times and costs, making it easier for companies to comply with safety regulations and enhancing the overall market potential for testing services in the region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Bioburden Testing Services Endotoxin Testing Services Sterility Testing Services Viral Clearance and Validation Services Cell Line Authentication and Characterization Tests Residual Host Contamination Testing Services Adventitious Agent Detection Testing Services |
| By End-User | Biopharmaceutical Companies Pharmaceutical Companies Biotechnology Firms Academic and Research Institutions Contract Research Organizations (CROs) |
| By Application | Vaccines & Therapeutics Cell and Gene Therapy Tissue & Tissue-based Products Blood & Blood-based Products Stem Cell Research |
| By Region | GCC Countries Levant Region North Africa Others |
| By Service Type | Testing Services Consulting Services Training Services Others |
| By Sample Type | Biological Samples Environmental Samples Product Samples Others |
| By Technology | Traditional Methods Molecular Techniques Automated Systems Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Biotechnology and Pharmaceutical Companies
Healthcare Providers and Hospitals
Laboratory Equipment Suppliers
Clinical Research Organizations (CROs)
Public Health Agencies (e.g., World Health Organization, Centers for Disease Control and Prevention)
Biological Safety Testing Laboratories
Players Mentioned in the Report:
Charles River Laboratories
Eurofins Scientific
SGS S.A.
Intertek Group plc
BioReliance (Merck KGaA)
WuXi AppTec
Labcorp Drug Development
Medpace Holdings, Inc.
Q2 Solutions
Nelson Labs (Sotera Health)
Viroclinics Xplore
BioAgilytix Labs
Pacific BioLabs
NAMSA (North American Science Associates)
Toxikon Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Biological Safety Testing Products Services Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Biological Safety Testing Products Services Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Biological Safety Testing Products Services Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for safety testing in pharmaceuticals
3.1.2 Rising awareness of biological hazards
3.1.3 Stringent regulatory requirements
3.1.4 Growth in biotechnology and pharmaceutical sectors
3.2 Market Challenges
3.2.1 High costs of testing services
3.2.2 Limited availability of skilled professionals
3.2.3 Variability in regulatory standards across countries
3.2.4 Competition from alternative testing methods
3.3 Market Opportunities
3.3.1 Expansion of biopharmaceutical companies
3.3.2 Technological advancements in testing methods
3.3.3 Increased investment in healthcare infrastructure
3.3.4 Collaborations and partnerships in research
3.4 Market Trends
3.4.1 Adoption of automation in testing processes
3.4.2 Growth of personalized medicine
3.4.3 Emphasis on environmental safety
3.4.4 Integration of AI in biological safety testing
3.5 Government Regulation
3.5.1 Implementation of Good Laboratory Practices (GLP)
3.5.2 Compliance with ISO standards
3.5.3 National health and safety regulations
3.5.4 Guidelines for biological waste management
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Biological Safety Testing Products Services Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Biological Safety Testing Products Services Market Segmentation
8.1 By Type
8.1.1 Bioburden Testing Services
8.1.2 Endotoxin Testing Services
8.1.3 Sterility Testing Services
8.1.4 Viral Clearance and Validation Services
8.1.5 Cell Line Authentication and Characterization Tests
8.1.6 Residual Host Contamination Testing Services
8.1.7 Adventitious Agent Detection Testing Services
8.2 By End-User
8.2.1 Biopharmaceutical Companies
8.2.2 Pharmaceutical Companies
8.2.3 Biotechnology Firms
8.2.4 Academic and Research Institutions
8.2.5 Contract Research Organizations (CROs)
8.3 By Application
8.3.1 Vaccines & Therapeutics
8.3.2 Cell and Gene Therapy
8.3.3 Tissue & Tissue-based Products
8.3.4 Blood & Blood-based Products
8.3.5 Stem Cell Research
8.4 By Region
8.4.1 GCC Countries
8.4.2 Levant Region
8.4.3 North Africa
8.4.4 Others
8.5 By Service Type
8.5.1 Testing Services
8.5.2 Consulting Services
8.5.3 Training Services
8.5.4 Others
8.6 By Sample Type
8.6.1 Biological Samples
8.6.2 Environmental Samples
8.6.3 Product Samples
8.6.4 Others
8.7 By Technology
8.7.1 Traditional Methods
8.7.2 Molecular Techniques
8.7.3 Automated Systems
8.7.4 Others
9. Middle East Biological Safety Testing Products Services Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Annual Revenue (USD Million)
9.2.4 Service Portfolio Breadth
9.2.5 Geographic Presence in Middle East
9.2.6 Testing Capacity and Turnaround Time
9.2.7 Regulatory Accreditations (ISO, FDA, EMA)
9.2.8 Market Penetration Rate
9.2.9 Customer Retention Rate
9.2.10 Pricing Strategy
9.2.11 Service Quality Index
9.2.12 Innovation Rate
9.2.13 Brand Recognition Score
9.2.14 Operational Efficiency Ratio
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Charles River Laboratories
9.5.2 Eurofins Scientific
9.5.3 SGS S.A.
9.5.4 Intertek Group plc
9.5.5 BioReliance (Merck KGaA)
9.5.6 WuXi AppTec
9.5.7 Labcorp Drug Development
9.5.8 Medpace Holdings, Inc.
9.5.9 Q2 Solutions
9.5.10 Nelson Labs (Sotera Health)
9.5.11 Viroclinics Xplore
9.5.12 BioAgilytix Labs
9.5.13 Pacific BioLabs
9.5.14 NAMSA (North American Science Associates)
9.5.15 Toxikon Corporation
10. Middle East Biological Safety Testing Products Services Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Budget Prioritization
10.2.3 Long-term Contracts
10.2.4 Cost Management Strategies
10.3 Pain Point Analysis by End-User Category
10.3.1 Regulatory Compliance Challenges
10.3.2 Quality Assurance Issues
10.3.3 Cost Constraints
10.3.4 Technology Adoption Barriers
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Infrastructure Readiness
10.4.3 Change Management
10.4.4 Support Services
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Feedback Mechanisms
10.5.3 Scalability Potential
10.5.4 Future Investment Plans
11. Middle East Biological Safety Testing Products Services Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
2.6 Customer Engagement Tactics
2.7 Performance Metrics
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Direct Sales Approaches
3.5 Partnership with Distributors
3.6 Logistics and Supply Chain Management
3.7 Performance Evaluation
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Discounting Strategies
4.7 Pricing Optimization Techniques
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends Identification
5.4 Customer Feedback Analysis
5.5 Market Research Insights
5.6 Future Demand Projections
5.7 Strategic Recommendations
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Support Strategies
6.4 Feedback Mechanisms
6.5 Relationship Management Tools
6.6 Performance Metrics
6.7 Continuous Improvement Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Approaches
7.4 Competitive Differentiation
7.5 Value-Added Services
7.6 Performance Metrics
7.7 Strategic Partnerships
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development
8.5 Market Research Activities
8.6 Performance Monitoring
8.7 Continuous Improvement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk Assessment
10.6 Strategic Fit Evaluation
10.7 Performance Metrics
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Financial Projections
11.4 Funding Sources
11.5 Risk Management Strategies
11.6 Performance Metrics
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
12.3 Control Mechanisms
12.4 Performance Metrics
12.5 Strategic Recommendations
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
13.3 Financial Health Indicators
13.4 Strategic Recommendations
13.5 Performance Metrics
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
14.4 Strategic Alliances
14.5 Performance Metrics
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for Key Activities
15.2.2 Performance Metrics
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from regional health authorities and biological safety organizations
- Review of scientific publications and journals focusing on biological safety testing methodologies
- Examination of industry white papers and case studies from leading biological safety testing firms
Primary Research
- Interviews with laboratory managers and directors in biological safety testing facilities
- Surveys with regulatory compliance officers in pharmaceutical and biotech companies
- Field interviews with researchers and scientists involved in biological safety testing
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and industry reports
- Triangulation of data from primary research and secondary sources to ensure accuracy
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and biological safety testing budgets
- Segmentation of the market by end-user industries such as pharmaceuticals, biotechnology, and research institutions
- Incorporation of government regulations and initiatives promoting biological safety standards
Bottom-up Modeling
- Collection of data on service pricing and volume from leading biological safety testing providers
- Estimation of operational costs based on laboratory setup and testing procedures
- Volume x cost analysis for various biological safety testing services offered
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors such as disease outbreaks and regulatory changes
- Scenario modeling based on potential advancements in biological safety technologies
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Services | 100 | Quality Assurance Managers, Regulatory Affairs Specialists |
| Biotechnology Safety Assessments | 75 | Laboratory Directors, Research Scientists |
| Clinical Research Organizations | 60 | Clinical Operations Managers, Compliance Officers |
| Environmental Safety Testing | 50 | Environmental Health Officers, Safety Compliance Managers |
| Academic Research Institutions | 80 | Research Professors, Laboratory Technicians |
Frequently Asked Questions
What is the current value of the Middle East Biological Safety Testing Products Services Market?
The Middle East Biological Safety Testing Products Services Market is valued at approximately USD 90 million, driven by increasing demand for safety testing in biopharmaceuticals and biotechnology sectors, along with rising regulatory requirements for product safety and efficacy.