About the Report
Base Year 2024Middle East Clinical Trial Management System (CTMS) Market Overview
- The Middle East Clinical Trial Management System (CTMS) Market is valued at USD 2.17 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing number of clinical trials, rising investments in research and development, and the growing demand for efficient management of clinical data. The market is also supported by advancements in technology, the adoption of cloud-based and AI-enhanced platforms, and the need for regulatory compliance in clinical research. The region is witnessing a surge in digital and decentralized clinical trials, leveraging telemedicine, wearable devices, and mobile applications to improve patient recruitment and retention .
- Key players in this market include the UAE, Saudi Arabia, and Israel, which dominate due to their robust healthcare infrastructure, significant investments in biotechnology, and a growing number of pharmaceutical companies. These countries have established themselves as hubs for clinical research, attracting global players and fostering innovation in clinical trial management. Saudi Arabia, in particular, is expanding rapidly due to strong government initiatives, healthcare investments, and supportive regulatory frameworks .
- In 2023, the UAE government implemented new regulations aimed at streamlining the clinical trial approval process. This initiative includes a digital platform for submission and review, reducing the time required for approvals and enhancing transparency. The regulation is designed to encourage more clinical trials in the region, thereby boosting the overall market for clinical trial management systems. The UAE’s regulatory reforms are part of a broader regional trend toward digitalization and efficiency in clinical research .
 Market.png)
Middle East Clinical Trial Management System (CTMS) Market Segmentation
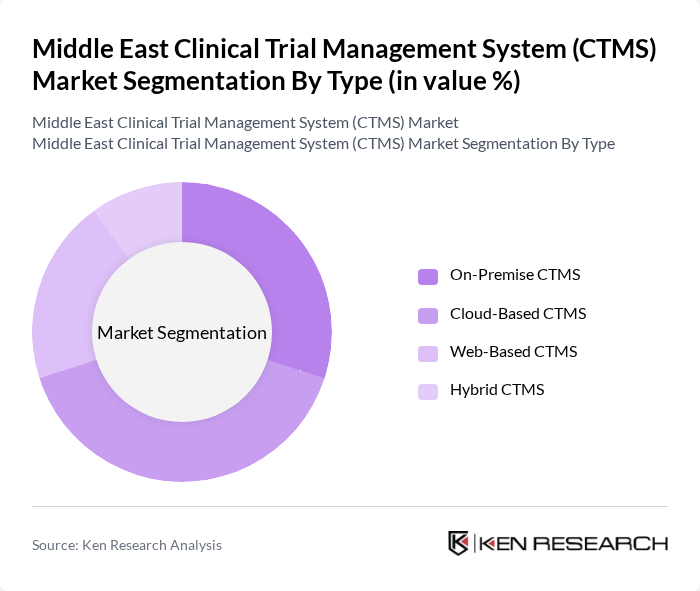
By Type:The market is segmented into four types: On-Premise CTMS, Cloud-Based CTMS, Web-Based CTMS, and Hybrid CTMS. Each type serves different operational needs and preferences of organizations involved in clinical trials. The On-Premise CTMS is preferred by larger organizations that require complete control over their data, while Cloud-Based CTMS is gaining traction due to its flexibility and cost-effectiveness. Web-Based CTMS offers ease of access, and Hybrid CTMS combines the benefits of both on-premise and cloud solutions.
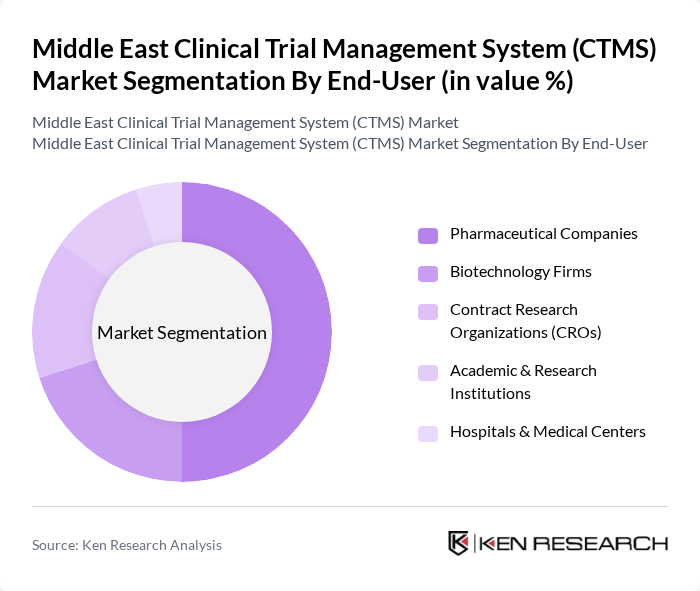
By End-User:This segmentation includes Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Academic & Research Institutions, Hospitals & Medical Centers, and Others. Pharmaceutical companies are the largest end-users due to their extensive clinical trial activities and need for robust data management systems. CROs are also significant players, as they manage trials on behalf of pharmaceutical companies, driving demand for CTMS solutions.
Middle East Clinical Trial Management System (CTMS) Market Competitive Landscape
The Middle East Clinical Trial Management System (CTMS) Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions, Oracle Corporation, Veeva Systems, Parexel International, Labcorp (formerly Covance), BioClinica (now part of Clario), Medpace, eClinical Solutions, ArisGlobal, Clinical Ink, TrialSpark, Celerion, Syneos Health, WCG Clinical, PPD (part of Thermo Fisher Scientific) contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Clinical Trial Management System (CTMS) Market Industry Analysis
Growth Drivers
- Increasing Demand for Efficient Clinical Trial Processes:The Middle East is witnessing a surge in clinical trials, with over 1,200 trials registered in future alone, reflecting a 15% increase from the previous period. This demand is driven by the need for faster drug development and improved patient outcomes. Efficient CTMS solutions streamline processes, reduce timelines, and enhance data management, making them essential for stakeholders aiming to meet regulatory requirements and patient needs effectively.
- Rising Investment in Healthcare Infrastructure:The Middle East's healthcare expenditure is projected to reach $135 billion in future, driven by government initiatives and private investments. Countries like Saudi Arabia and the UAE are investing heavily in healthcare infrastructure, including research facilities and clinical trial sites. This investment fosters an environment conducive to clinical research, thereby increasing the adoption of CTMS solutions to manage the growing number of trials efficiently.
- Enhanced Regulatory Support for Clinical Research:Regulatory bodies in the Middle East are increasingly supportive of clinical research, with the UAE's Ministry of Health and Prevention approving 300 clinical trials in future, a 20% increase from the previous period. This regulatory backing not only accelerates trial approvals but also encourages the adoption of CTMS solutions, as organizations seek to comply with evolving regulations and ensure data integrity throughout the trial process.
Market Challenges
- Limited Awareness of CTMS Benefits:Despite the growing demand for CTMS, awareness remains low among many stakeholders in the Middle East. A survey conducted in future indicated that only 40% of clinical trial sponsors were familiar with CTMS functionalities. This lack of understanding hampers adoption rates, as organizations may not recognize the potential efficiencies and cost savings that a robust CTMS can provide in managing clinical trials.
- High Implementation Costs:The initial investment required for implementing a CTMS can be substantial, often exceeding $100,000 for mid-sized organizations. This financial barrier is particularly challenging for smaller clinical research organizations and startups, which may struggle to allocate sufficient budgets for technology adoption. Consequently, high implementation costs can deter potential users from transitioning to more efficient systems, limiting market growth.
Middle East Clinical Trial Management System (CTMS) Market Future Outlook
The future of the Middle East CTMS market appears promising, driven by technological advancements and increasing collaboration among stakeholders. The integration of AI and machine learning is expected to enhance data analytics capabilities, improving trial efficiency and patient outcomes. Additionally, the rise of telemedicine and remote monitoring will likely reshape clinical trial methodologies, making them more patient-centric and accessible, thus fostering further growth in the CTMS sector.
Market Opportunities
- Expansion of Telemedicine and Remote Monitoring:The telemedicine market in the Middle East is projected to reach $2.5 billion in future, creating opportunities for CTMS providers to integrate remote monitoring capabilities. This integration can enhance patient engagement and data collection, making clinical trials more efficient and accessible, particularly in remote areas.
- Collaborations with Pharmaceutical Companies:With over 50 pharmaceutical companies operating in the region, partnerships between CTMS providers and these firms can lead to innovative solutions tailored to specific trial needs. Such collaborations can enhance the development of customized CTMS features, driving adoption and improving trial outcomes across the Middle East.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | On-Premise CTMS Cloud-Based CTMS Web-Based CTMS Hybrid CTMS |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Academic & Research Institutions Hospitals & Medical Centers Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Rare Diseases Others |
| By Deployment Mode | Public Cloud Private Cloud Hybrid Cloud On-Premise |
| By Functionality | Study Management Site Management Patient Management Data Management Reporting & Analytics Financial Management Regulatory & Compliance Management Others |
| By Region | GCC Countries (Saudi Arabia, UAE, Kuwait, Qatar, Oman, Bahrain) Levant Region (Jordan, Lebanon, Syria, Palestine, Iraq) North Africa (Egypt, Morocco, Algeria, Tunisia, Libya) Others |
| By Pricing Model | Subscription-Based Perpetual License Pay-Per-Use Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Pharmaceutical Companies
Biotechnology Firms
Clinical Research Organizations (CROs)
Healthcare Providers and Hospitals
Technology Providers and Software Developers
Insurance Companies and Payers
Players Mentioned in the Report:
Medidata Solutions
Oracle Corporation
Veeva Systems
Parexel International
Labcorp (formerly Covance)
BioClinica (now part of Clario)
Medpace
eClinical Solutions
ArisGlobal
Clinical Ink
TrialSpark
Celerion
Syneos Health
WCG Clinical
PPD (part of Thermo Fisher Scientific)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Clinical Trial Management System (CTMS) Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Clinical Trial Management System (CTMS) Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Clinical Trial Management System (CTMS) Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for efficient clinical trial processes
3.1.2 Rising investment in healthcare infrastructure
3.1.3 Growing number of clinical trials in the region
3.1.4 Enhanced regulatory support for clinical research
3.2 Market Challenges
3.2.1 Limited awareness of CTMS benefits
3.2.2 High implementation costs
3.2.3 Data privacy and security concerns
3.2.4 Fragmented market with varying regulations
3.3 Market Opportunities
3.3.1 Expansion of telemedicine and remote monitoring
3.3.2 Collaborations with pharmaceutical companies
3.3.3 Adoption of AI and machine learning in CTMS
3.3.4 Increasing focus on patient-centric trials
3.4 Market Trends
3.4.1 Shift towards cloud-based CTMS solutions
3.4.2 Integration of mobile technologies in clinical trials
3.4.3 Emphasis on real-time data analytics
3.4.4 Growing importance of patient engagement tools
3.5 Government Regulation
3.5.1 Stricter guidelines for clinical trial approvals
3.5.2 Enhanced monitoring of clinical trial data
3.5.3 Supportive policies for research and development
3.5.4 Incentives for local clinical trial conductors
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Clinical Trial Management System (CTMS) Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Clinical Trial Management System (CTMS) Market Segmentation
8.1 By Type
8.1.1 On-Premise CTMS
8.1.2 Cloud-Based CTMS
8.1.3 Web-Based CTMS
8.1.4 Hybrid CTMS
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Contract Research Organizations (CROs)
8.2.4 Academic & Research Institutions
8.2.5 Hospitals & Medical Centers
8.2.6 Others
8.3 By Therapeutic Area
8.3.1 Oncology
8.3.2 Cardiovascular
8.3.3 Neurology
8.3.4 Infectious Diseases
8.3.5 Rare Diseases
8.3.6 Others
8.4 By Deployment Mode
8.4.1 Public Cloud
8.4.2 Private Cloud
8.4.3 Hybrid Cloud
8.4.4 On-Premise
8.5 By Functionality
8.5.1 Study Management
8.5.2 Site Management
8.5.3 Patient Management
8.5.4 Data Management
8.5.5 Reporting & Analytics
8.5.6 Financial Management
8.5.7 Regulatory & Compliance Management
8.5.8 Others
8.6 By Region
8.6.1 GCC Countries (Saudi Arabia, UAE, Kuwait, Qatar, Oman, Bahrain)
8.6.2 Levant Region (Jordan, Lebanon, Syria, Palestine, Iraq)
8.6.3 North Africa (Egypt, Morocco, Algeria, Tunisia, Libya)
8.6.4 Others
8.7 By Pricing Model
8.7.1 Subscription-Based
8.7.2 Perpetual License
8.7.3 Pay-Per-Use
8.7.4 Others
9. Middle East Clinical Trial Management System (CTMS) Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Regional Market Share
9.2.4 Revenue Growth Rate (Middle East)
9.2.5 Number of Clinical Trials Supported
9.2.6 Customer Retention Rate
9.2.7 Market Penetration Rate
9.2.8 Pricing Strategy
9.2.9 Product Innovation Index (e.g., frequency of new feature releases)
9.2.10 Regulatory Compliance Certifications (e.g., ICH-GCP, local MOH)
9.2.11 Customer Satisfaction Score (NPS or equivalent)
9.2.12 Average Deal Size
9.2.13 Local Partnership/Presence (offices, data centers, local support)
9.2.14 Implementation Timeframe
9.2.15 Sales Conversion Rate
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medidata Solutions
9.5.2 Oracle Corporation
9.5.3 Veeva Systems
9.5.4 Parexel International
9.5.5 Labcorp (formerly Covance)
9.5.6 BioClinica (now part of Clario)
9.5.7 Medpace
9.5.8 eClinical Solutions
9.5.9 ArisGlobal
9.5.10 Clinical Ink
9.5.11 TrialSpark
9.5.12 Celerion
9.5.13 Syneos Health
9.5.14 WCG Clinical
9.5.15 PPD (part of Thermo Fisher Scientific)
10. Middle East Clinical Trial Management System (CTMS) Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Vendors
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Spending Patterns
10.2.3 Funding Sources
10.3 Pain Point Analysis by End-User Category
10.3.1 Common Challenges Faced
10.3.2 Impact of Regulatory Changes
10.3.3 Technology Adoption Barriers
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Technology Familiarity
10.4.3 Support Requirements
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success
10.5.2 Future Use Cases
10.5.3 Feedback Mechanisms
11. Middle East Clinical Trial Management System (CTMS) Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare and clinical trial organizations in the Middle East
- Review of government publications and healthcare regulations impacting clinical trials
- Examination of market trends and forecasts from reputable healthcare analytics firms
Primary Research
- Interviews with clinical trial managers and coordinators across leading hospitals and research institutions
- Surveys targeting pharmaceutical companies and CROs (Contract Research Organizations) operating in the region
- Focus groups with regulatory bodies and ethics committees involved in clinical trial approvals
Validation & Triangulation
- Cross-validation of findings through multiple data sources including academic journals and industry publications
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panels comprising industry veterans and regulatory experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on healthcare expenditure in the Middle East
- Segmentation of the market by therapeutic areas and types of clinical trials
- Incorporation of regional healthcare initiatives and funding for clinical research
Bottom-up Modeling
- Collection of data on the number of active clinical trials and their respective budgets
- Estimation of costs associated with trial management software and services
- Analysis of growth rates based on historical data from clinical trial registrations
Forecasting & Scenario Analysis
- Multi-variable forecasting using factors such as technological advancements and regulatory changes
- Scenario analysis based on potential shifts in healthcare policies and funding
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 120 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Medical Device Trials | 90 | Product Development Managers, Clinical Research Associates |
| Oncology Trials | 75 | Oncologists, Research Coordinators |
| Cardiovascular Trials | 65 | Cardiologists, Clinical Operations Managers |
| Regulatory Compliance in Trials | 55 | Compliance Officers, Ethics Committee Members |
Frequently Asked Questions
What is the current value of the Middle East Clinical Trial Management System (CTMS) Market?
The Middle East Clinical Trial Management System (CTMS) Market is valued at approximately USD 2.17 billion, reflecting significant growth driven by an increase in clinical trials and investments in research and development across the region.