Middle East Human Papilloma Virus Testing Market Overview
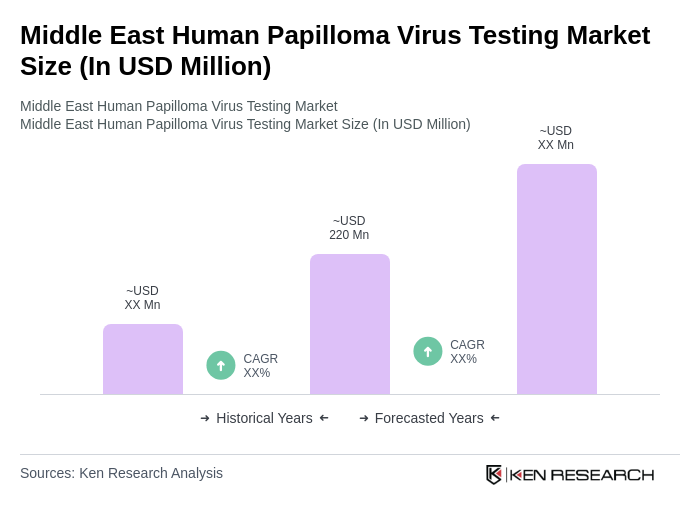
- The Middle East Human Papilloma Virus Testing Market is valued at USD 220 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of HPV-related health issues, rising incidences of cervical cancer, and the implementation of national screening programs across various countries in the region. The demand for effective testing solutions has surged as healthcare providers focus on preventive measures and early detection.
- Key players in this market include Saudi Arabia, the United Arab Emirates, and Egypt, which dominate due to their advanced healthcare infrastructure, government initiatives promoting HPV vaccination and screening, and a growing population that is increasingly health-conscious. These countries have established comprehensive healthcare policies that support HPV testing and awareness campaigns, further solidifying their market leadership.
- In 2023, the Saudi Arabian government introduced the "National Cervical Cancer Screening Program Guidelines" issued by the Ministry of Health, mandating HPV testing as part of routine cervical cancer screening for women aged 30 and above. This initiative requires healthcare providers to offer HPV DNA testing alongside cytology for eligible women, aiming to enhance early detection rates and reduce cervical cancer mortality, reflecting the government's commitment to improving women's health and increasing access to preventive healthcare services.
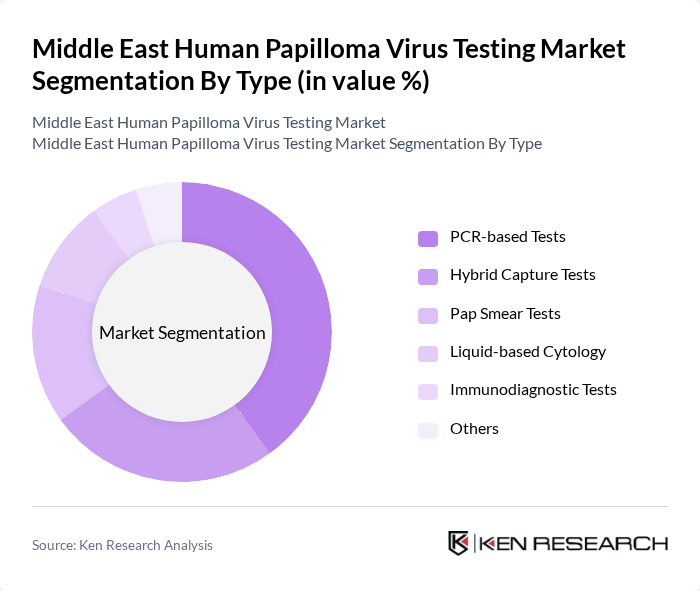
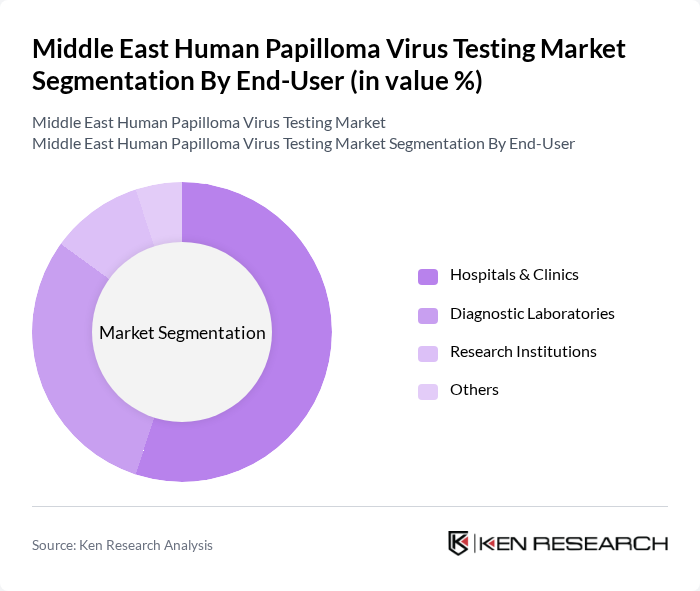
Middle East Human Papilloma Virus Testing Market Segmentation
By Type:The market is segmented into various testing types, including PCR-based Tests, Hybrid Capture Tests, Pap Smear Tests, Liquid-based Cytology, Immunodiagnostic Tests, and Others. Among these, PCR-based Tests are gaining traction due to their high sensitivity and specificity, making them the preferred choice for accurate HPV detection. The increasing adoption of advanced molecular diagnostics in healthcare facilities is further propelling the demand for PCR-based Tests.
By End-User:The market is categorized into Hospitals & Clinics, Diagnostic Laboratories, Research Institutions, and Others. Hospitals & Clinics are the leading end-users, driven by the increasing number of HPV tests conducted as part of routine health check-ups and cancer screening programs. The growing emphasis on preventive healthcare and the integration of HPV testing into women's health services are key factors contributing to the dominance of this segment.
Middle East Human Papilloma Virus Testing Market Competitive Landscape
The Middle East Human Papilloma Virus Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Hologic, Inc., Qiagen N.V., Becton, Dickinson and Company, Siemens Healthineers, Cepheid, Inc., BioMérieux S.A., Thermo Fisher Scientific Inc., Agilent Technologies, Inc., PerkinElmer, Inc., F. Hoffmann-La Roche Ltd., Merck KGaA, Illumina, Inc., Seegene Inc., Bio-Rad Laboratories, Inc., Trinity Biotech plc, Al Borg Diagnostics, National Reference Laboratory (UAE), King Faisal Specialist Hospital & Research Centre (Saudi Arabia) contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Human Papilloma Virus Testing Market Industry Analysis
Growth Drivers
- Increasing Awareness of HPV-Related Health Issues:The Middle East has seen a significant rise in awareness regarding HPV-related health issues, with educational campaigns reaching over 10 million individuals in future. This heightened awareness is crucial, as studies indicate that approximately 70% of cervical cancer cases are linked to HPV. The World Health Organization reported that in future, cervical cancer is projected to affect around 12,000 women in the region, emphasizing the need for effective testing solutions.
- Government Initiatives for Cancer Screening:Governments across the Middle East are increasingly prioritizing cancer screening initiatives. For instance, the UAE allocated $50 million in future to enhance cancer screening programs, including HPV testing. This funding aims to increase screening rates by 30% in future, addressing the rising incidence of HPV-related cancers. Such initiatives are expected to significantly boost the demand for HPV testing services in the region.
- Advancements in Testing Technologies:The Middle East is witnessing rapid advancements in HPV testing technologies, with new molecular testing methods being introduced. In future, the introduction of a new HPV test reduced testing time by 50%, allowing for quicker diagnosis. Additionally, the market for HPV testing kits is projected to grow by 20% annually, driven by innovations that enhance accuracy and reduce costs, making testing more accessible to the population.
Market Challenges
- Limited Access to Healthcare Facilities:Access to healthcare facilities remains a significant challenge in the Middle East, particularly in rural areas. According to the World Bank, approximately 30% of the population lives more than 30 kilometers from the nearest healthcare facility. This geographical barrier limits access to HPV testing, contributing to lower screening rates and delayed diagnoses, which can exacerbate health outcomes for affected individuals.
- Cultural Stigmas Surrounding HPV Testing:Cultural stigmas associated with HPV and its testing pose a considerable challenge in the Middle East. Surveys indicate that nearly 40% of women avoid HPV testing due to fear of social repercussions. This stigma is compounded by a lack of public education on HPV, leading to misconceptions that hinder testing uptake. Addressing these cultural barriers is essential for improving screening rates and overall public health.
Middle East Human Papilloma Virus Testing Market Future Outlook
The future of the HPV testing market in the Middle East appears promising, driven by increasing government support and technological advancements. As awareness campaigns continue to educate the public, the demand for HPV testing is expected to rise significantly. Furthermore, the integration of telemedicine and digital health solutions will facilitate easier access to testing services, particularly in underserved areas. These trends indicate a shift towards more proactive healthcare measures, ultimately improving health outcomes across the region.
Market Opportunities
- Expansion of Telemedicine Services:The growth of telemedicine services presents a unique opportunity for HPV testing in the Middle East. With an estimated 15 million telehealth consultations conducted in future, integrating HPV testing into these services can enhance accessibility, particularly for women in remote areas. This approach can significantly increase testing rates and early detection of HPV-related conditions.
- Development of Cost-Effective Testing Kits:There is a growing opportunity for the development of cost-effective HPV testing kits tailored for the Middle Eastern market. With testing costs averaging $100 per procedure, affordable alternatives could increase accessibility. By future, the introduction of kits priced below $50 could potentially double the number of tests conducted annually, making a substantial impact on public health.