Region:Middle East
Author(s):Geetanshi
Product Code:KRAC3150
Pages:88
Published On:October 2025
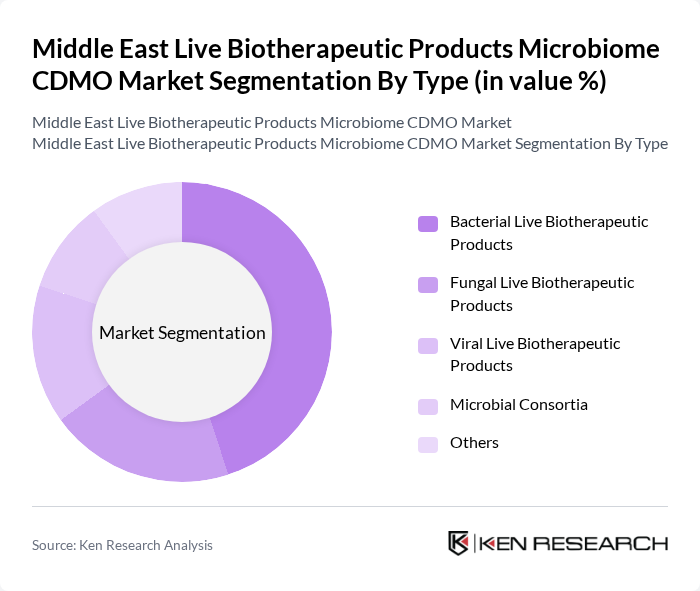
By Type:The market is segmented into various types of live biotherapeutic products, including bacterial, fungal, viral, microbial consortia, and others. Among these, bacterial live biotherapeutic products are the most dominant due to their extensive application in treating gastrointestinal disorders and their established efficacy in clinical settings. The increasing consumer preference for probiotic products further drives the demand for bacterial formulations.
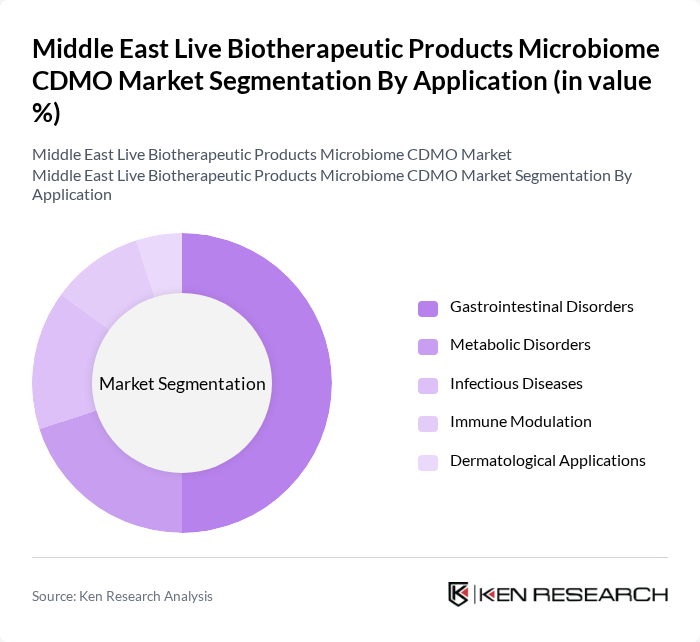
By Application:The applications of live biotherapeutic products include gastrointestinal disorders, metabolic disorders, infectious diseases, immune modulation, dermatological applications, and others. Gastrointestinal disorders are the leading application area, driven by the rising incidence of conditions such as irritable bowel syndrome (IBS) and Clostridium difficile infections. The growing consumer awareness regarding gut health and the efficacy of probiotics in managing these conditions further supports this trend.
The Middle East Live Biotherapeutic Products Microbiome CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lifera (Saudi Arabia), Biose Industrie, Lallemand Health Solutions, Lonza Group AG, Arranta Bio (a Recipharm company), Ferring Pharmaceuticals, Pharmabiome AG, Probi AB, 4D Pharma PLC, Seres Therapeutics, Inc., Enterome Bioscience SA, Eligo Bioscience, Symbiosis Pharmaceutical Services, Evolve BioSystems, Inc., BioCare Copenhagen A/S contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East Live Biotherapeutic Products Microbiome CDMO market appears promising, driven by increasing investments in biopharmaceuticals and a growing focus on personalized medicine. As consumer demand for gut health solutions rises, companies are likely to enhance their R&D efforts, leading to innovative product offerings. Furthermore, collaborations between CDMOs and research institutions are expected to foster advancements in microbiome therapies, creating a dynamic environment for growth and development in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Bacterial Live Biotherapeutic Products Fungal Live Biotherapeutic Products Viral Live Biotherapeutic Products Microbial Consortia Others |
| By Application | Gastrointestinal Disorders (e.g., C. difficile infection, IBD, IBS) Metabolic Disorders (e.g., Diabetes, Obesity) Infectious Diseases Immune Modulation Dermatological Applications Others |
| By Manufacturing Process | Fermentation Formulation & Fill-Finish Analytical & Quality Control Packaging Others |
| By End-User | Pharmaceutical & Biotechnology Companies Research Institutes & Academic Centers Hospitals & Clinics Others |
| By Service Type | Contract Development Contract Manufacturing Clinical Trial Manufacturing Commercial Manufacturing Others |
| By Country/Region | GCC Countries (Saudi Arabia, UAE, Qatar, etc.) Levant (Jordan, Lebanon, etc.) North Africa (Egypt, Morocco, etc.) Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Microbiome Research Institutions | 65 | Research Scientists, Lab Directors |
| Biotherapeutic Product Developers | 55 | Product Managers, R&D Heads |
| Regulatory Affairs in Biotherapeutics | 45 | Regulatory Managers, Compliance Officers |
| Healthcare Providers Utilizing Biotherapeutics | 50 | Clinical Practitioners, Pharmacists |
| Investors in Biotherapeutic Startups | 40 | Venture Capitalists, Angel Investors |
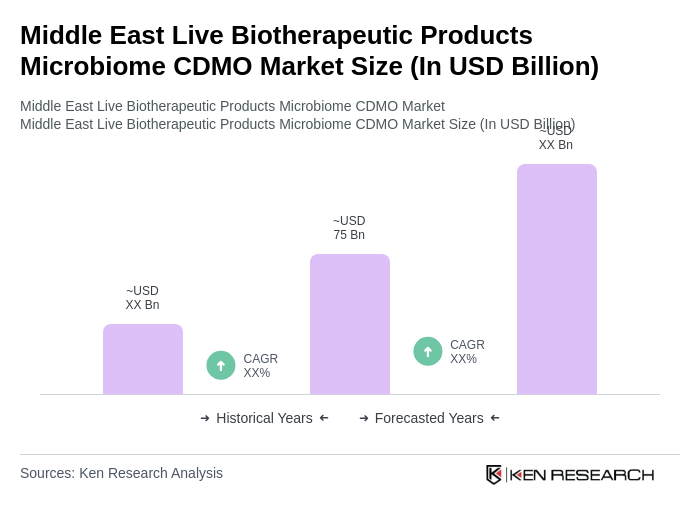
The Middle East Live Biotherapeutic Products Microbiome CDMO Market is valued at approximately USD 75 billion, reflecting significant growth driven by investments in biopharmaceuticals and increasing awareness of microbiome health benefits.