Region:Middle East
Author(s):Dev
Product Code:KRAC8774
Pages:92
Published On:November 2025
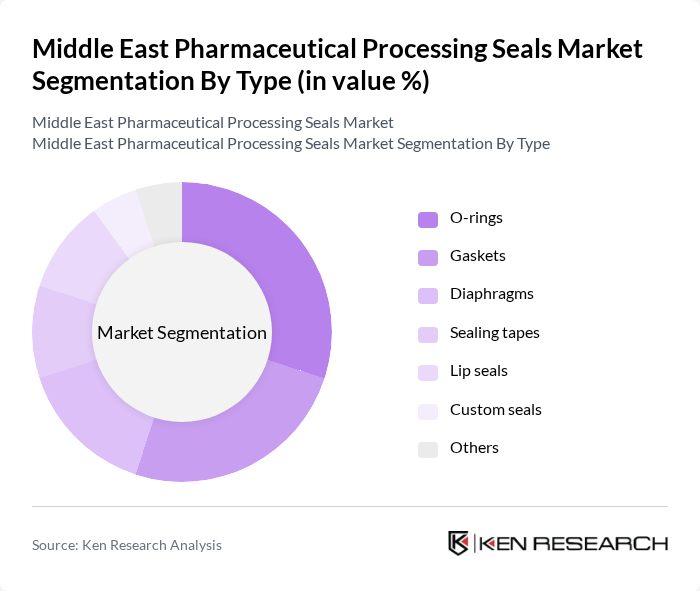
By Type:The market is segmented into various types of seals, including O-rings, gaskets, diaphragms, sealing tapes, lip seals, custom seals, and others. Among these, O-rings and gaskets are the most widely used due to their versatility and effectiveness in preventing leaks and maintaining sterile conditions in pharmaceutical applications. The demand for custom seals is also rising as manufacturers seek tailored solutions for specific equipment and process requirements. The adoption of advanced materials such as PTFE, silicone, and EPDM is increasing for enhanced chemical resistance and durability .
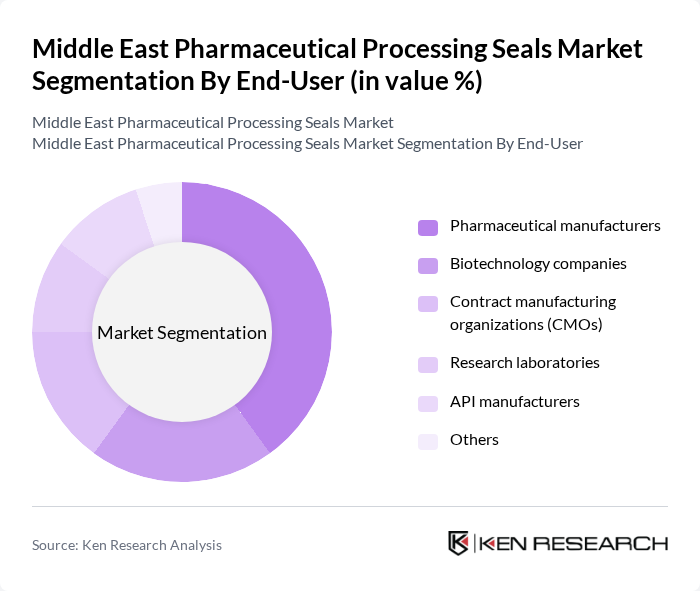
By End-User:The end-user segmentation includes pharmaceutical manufacturers, biotechnology companies, contract manufacturing organizations (CMOs), research laboratories, API manufacturers, and others. Pharmaceutical manufacturers are the largest segment, driven by the need for reliable sealing solutions in drug production processes. The biotechnology sector is also growing rapidly, increasing the demand for specialized seals that support sterile and high-purity manufacturing environments. The adoption of automation and digitalization in pharmaceutical production is further boosting demand for advanced sealing technologies .
The Middle East Pharmaceutical Processing Seals Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saint-Gobain, Parker Hannifin, Trelleborg AB, Freudenberg Sealing Technologies, A.W. Chesterton Company, SKF Group, John Crane (Smiths Group plc), SMC Corporation, Klinger Group, Garlock (Enpro Industries), W. L. Gore & Associates (GORE®), ElringKlinger AG, Flowserve Corporation, Dichtomatik (Freudenberg Group), M+W Products GmbH contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East pharmaceutical processing seals market appears promising, driven by increasing investments in healthcare infrastructure and a growing emphasis on biopharmaceutical production. As manufacturers adapt to stringent regulatory requirements, the demand for innovative sealing solutions will rise. Additionally, the integration of automation and IoT technologies in production processes is expected to enhance operational efficiency, leading to improved product quality and safety. This evolving landscape presents significant opportunities for growth and innovation in the sector.
| Segment | Sub-Segments |
|---|---|
| By Type | O-rings Gaskets Diaphragms Sealing tapes Lip seals Custom seals Others |
| By End-User | Pharmaceutical manufacturers Biotechnology companies Contract manufacturing organizations (CMOs) Research laboratories API manufacturers Others |
| By Material | Silicone PTFE (Polytetrafluoroethylene) EPDM (Ethylene Propylene Diene Monomer) FKM (Fluoroelastomer) Rubber Others |
| By Application | Liquid processing Solid processing Aseptic processing Packaging Filtration systems Others |
| By Region | GCC Countries Levant Region North Africa Others |
| By Compliance Standards | FDA regulations EMA guidelines ISO standards USP Class VI Others |
| By Distribution Channel | Direct sales Distributors Online sales Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturing | 100 | Production Managers, Quality Control Supervisors |
| Research & Development | 60 | R&D Directors, Product Development Scientists |
| Procurement & Supply Chain | 50 | Procurement Managers, Supply Chain Analysts |
| Regulatory Affairs | 40 | Regulatory Affairs Managers, Compliance Officers |
| Quality Assurance | 45 | Quality Assurance Managers, Validation Specialists |
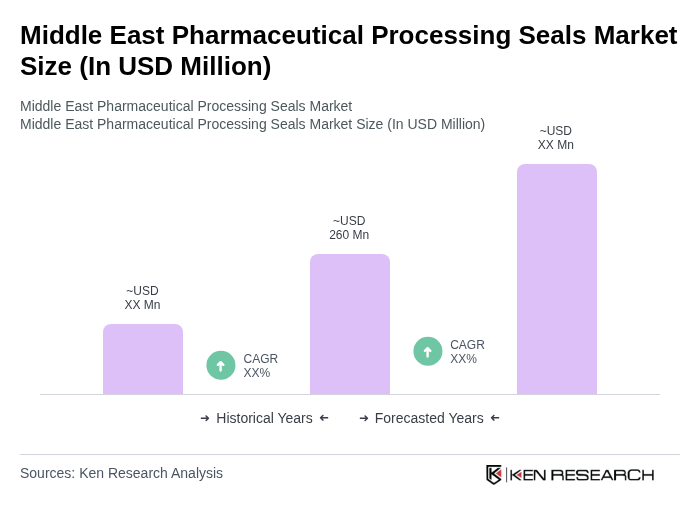
The Middle East Pharmaceutical Processing Seals Market is valued at approximately USD 260 million, reflecting a five-year historical analysis of regional shares within the global market. This growth is driven by increasing demand for high-quality pharmaceutical products and stringent regulatory requirements.