About the Report
Base Year 2024Middle East Remicade Market Overview
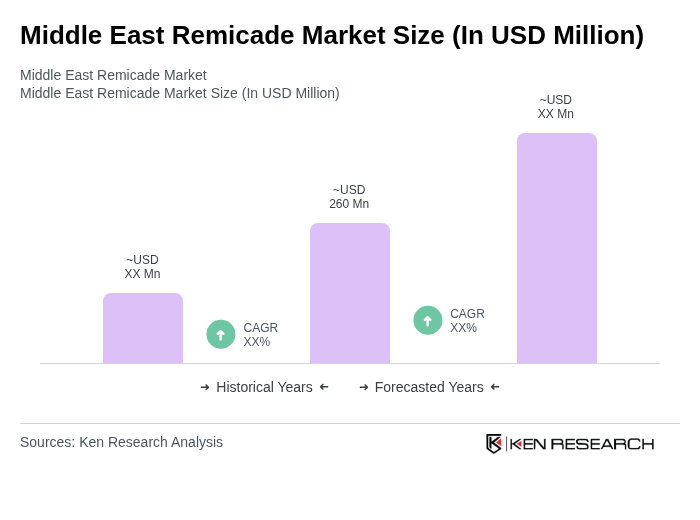
- The Middle East Remicade Market is valued at USD 260 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of autoimmune diseases, rising healthcare expenditure, and advancements in biologic therapies. The demand for Remicade, a leading treatment for conditions such as rheumatoid arthritis and Crohn's disease, has significantly contributed to the market's expansion. Recent trends include the rapid adoption of biosimilars, expansion of telemedicine and e-pharmacy platforms, and increased government funding for biologic therapies, particularly in Saudi Arabia and the United Arab Emirates .
- Key players in this market include Saudi Arabia, the United Arab Emirates, and Egypt. These countries dominate the market due to their robust healthcare infrastructure, increasing investments in healthcare technology, and a growing population with rising healthcare needs. The presence of well-established pharmaceutical companies and an evolving regulatory environment further enhance their market position. Government initiatives such as Saudi Arabia’s Vision 2030 and the UAE’s healthcare digitalization programs are supporting the expansion of biologics infrastructure and access .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Guideline for Registration of Biological Products”, issued by the Saudi Food and Drug Authority, 2023. This regulation streamlines the approval process for biologics, including Remicade, by introducing accelerated review pathways for innovative therapies and biosimilars. The guideline mandates comprehensive clinical and quality data submission, ensures pharmacovigilance, and establishes clear timelines for regulatory decisions, thereby enhancing patient access and fostering a more competitive market landscape .
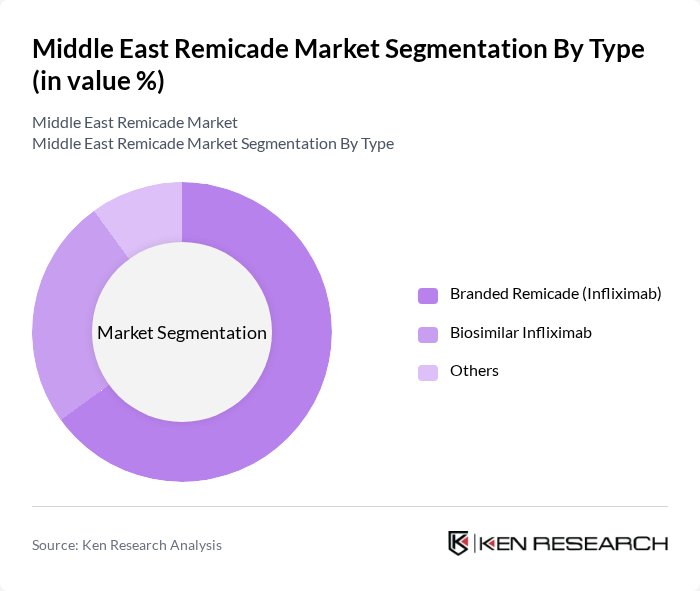
Middle East Remicade Market Segmentation
By Type:The market is segmented into three main types: Branded Remicade (Infliximab), Biosimilar Infliximab, and Others. The Branded Remicade segment remains the dominant player due to its established reputation and extensive clinical data supporting its efficacy. However, biosimilars are rapidly gaining traction as cost-effective alternatives, supported by government initiatives to increase affordability and access. The "Others" category includes alternative formulations and delivery methods, which are gradually expanding as new entrants and local manufacturers introduce differentiated products .
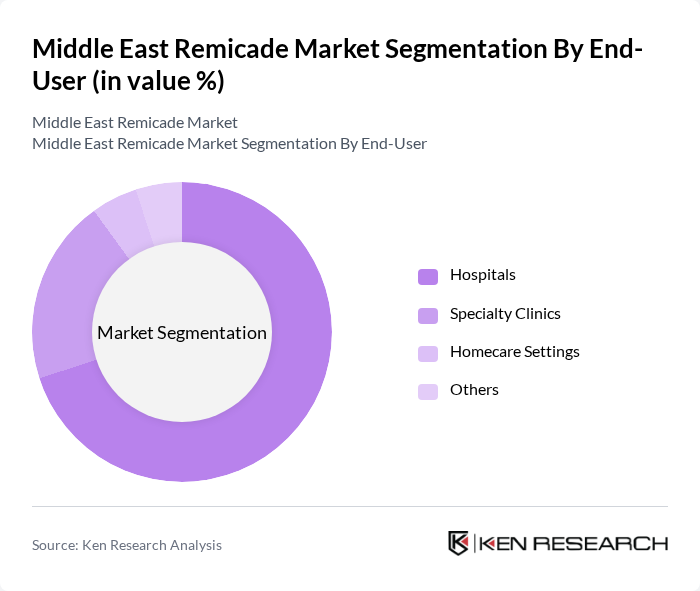
By End-User:The market is categorized into Hospitals, Specialty Clinics, Homecare Settings, and Others. Hospitals are the leading end-users due to their capacity to provide comprehensive treatment and monitoring for patients with complex autoimmune conditions. Specialty clinics play a significant role, focusing on disease-specific management and biologic infusion services. Homecare settings are emerging as a convenient option for patients requiring ongoing treatment, supported by the growth of home infusion services and telehealth platforms .
Middle East Remicade Market Competitive Landscape
The Middle East Remicade Market is characterized by a dynamic mix of regional and international players. Leading participants such as Johnson & Johnson (Janssen Biotech), Merck & Co., Inc. (MSD), AbbVie Inc., Amgen Inc., Pfizer Inc., Sandoz International GmbH (Novartis Group), Viatris Inc. (formerly Mylan N.V.), Teva Pharmaceutical Industries Ltd., Biogen Inc., UCB S.A., Fresenius Kabi AG, Hikma Pharmaceuticals PLC, EMD Serono (Merck KGaA), Celltrion Healthcare Co., Ltd., Samsung Bioepis Co., Ltd., Julphar (Gulf Pharmaceutical Industries), Tabuk Pharmaceuticals Manufacturing Co., SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Pharco Pharmaceuticals, and Eva Pharma contribute to innovation, geographic expansion, and service delivery in this space .
Middle East Remicade Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Autoimmune Diseases:The Middle East has witnessed a significant rise in autoimmune diseases, with an estimated 1.5 million people affected by conditions such as rheumatoid arthritis and Crohn's disease. According to the World Health Organization, the prevalence of these diseases is expected to increase by 20% in future. This growing patient population drives demand for effective treatments like Remicade, which is crucial for managing these chronic conditions and improving patient outcomes.
- Rising Healthcare Expenditure:Healthcare spending in the Middle East is projected to reach $135 billion in future, reflecting a compound annual growth rate of 7.5%. This increase is driven by government initiatives to enhance healthcare infrastructure and services. As healthcare budgets expand, more resources are allocated to innovative treatments, including biologics like Remicade, which are essential for managing complex autoimmune diseases, thereby boosting market growth.
- Expanding Access to Healthcare Facilities:The number of healthcare facilities in the Middle East is expected to grow by 15% in future, with investments in both public and private sectors. This expansion enhances access to advanced medical treatments, including Remicade. Improved healthcare infrastructure facilitates timely diagnosis and treatment of autoimmune diseases, contributing to increased patient enrollment in treatment programs and ultimately driving market growth.
Market Challenges
- High Cost of Treatment:The cost of Remicade treatment can exceed $28,000 annually per patient, posing a significant financial burden on healthcare systems and patients. This high cost limits accessibility, particularly in lower-income regions of the Middle East, where healthcare budgets are constrained. Consequently, many patients may not receive timely treatment, hindering overall market growth and patient outcomes.
- Regulatory Hurdles:The regulatory landscape in the Middle East presents challenges for the approval and distribution of Remicade. Each country has distinct regulatory requirements, leading to delays in product launches. For instance, the average time for drug approval can range from 12 to 24 months, which can impede market entry and limit the availability of essential treatments for patients suffering from autoimmune diseases.
Middle East Remicade Market Future Outlook
The Middle East Remicade market is poised for significant growth, driven by increasing awareness of autoimmune diseases and advancements in treatment options. The integration of digital health solutions is expected to enhance patient engagement and adherence to treatment protocols. Additionally, the shift towards personalized medicine will likely lead to more tailored therapeutic approaches, improving patient outcomes. As healthcare systems evolve, the demand for innovative therapies like Remicade will continue to rise, fostering a more robust market environment.
Market Opportunities
- Development of Biosimilars:The introduction of biosimilars presents a significant opportunity in the Middle East, potentially reducing treatment costs by up to 25%. This could enhance patient access to effective therapies for autoimmune diseases, driving market growth and increasing competition among manufacturers.
- Expansion into Emerging Markets:Emerging markets in the Middle East, such as Iraq and Yemen, are experiencing rapid healthcare development. By establishing a presence in these regions, companies can tap into a growing patient base, increasing the adoption of Remicade and other biologics, thereby enhancing overall market potential.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Branded Remicade (Infliximab) Biosimilar Infliximab Others |
| By End-User | Hospitals Specialty Clinics Homecare Settings Others |
| By Indication | Rheumatoid Arthritis Crohn's Disease Ulcerative Colitis Ankylosing Spondylitis Psoriasis Others |
| By Distribution Channel | Direct Sales Distributors Online Pharmacies Hospital Pharmacies Others |
| By Region | GCC Countries (Saudi Arabia, UAE, Kuwait, Qatar, Bahrain, Oman) Levant Region (Jordan, Lebanon, Syria, Iraq, Palestine) North Africa (Egypt, Morocco, Algeria, Tunisia) Others |
| By Patient Demographics | Age Group (Pediatric, Adult, Geriatric) Gender Others |
| By Treatment Setting | Inpatient Outpatient Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Food and Drug Administration)
Pharmaceutical Manufacturers and Producers
Distributors and Wholesalers
Healthcare Providers and Hospitals
Health Insurance Companies
Pharmacy Chains and Retail Pharmacies
Biotechnology Firms
Players Mentioned in the Report:
Johnson & Johnson (Janssen Biotech)
Merck & Co., Inc. (MSD)
AbbVie Inc.
Amgen Inc.
Pfizer Inc.
Sandoz International GmbH (Novartis Group)
Viatris Inc. (formerly Mylan N.V.)
Teva Pharmaceutical Industries Ltd.
Biogen Inc.
UCB S.A.
Fresenius Kabi AG
Hikma Pharmaceuticals PLC
EMD Serono (Merck KGaA)
Celltrion Healthcare Co., Ltd.
Samsung Bioepis Co., Ltd.
Julphar (Gulf Pharmaceutical Industries)
Tabuk Pharmaceuticals Manufacturing Co.
SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
Pharco Pharmaceuticals
Eva Pharma
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Remicade Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Remicade Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Remicade Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of autoimmune diseases
3.1.2 Rising healthcare expenditure
3.1.3 Expanding access to healthcare facilities
3.1.4 Growing awareness of treatment options
3.2 Market Challenges
3.2.1 High cost of treatment
3.2.2 Regulatory hurdles
3.2.3 Limited availability of biosimilars
3.2.4 Variability in healthcare policies across countries
3.3 Market Opportunities
3.3.1 Development of biosimilars
3.3.2 Expansion into emerging markets
3.3.3 Collaborations with healthcare providers
3.3.4 Increasing investment in R&D
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Integration of digital health solutions
3.4.3 Focus on patient-centric care
3.4.4 Rising demand for home healthcare services
3.5 Government Regulation
3.5.1 Pricing controls on pharmaceuticals
3.5.2 Approval processes for new treatments
3.5.3 Policies promoting biosimilar adoption
3.5.4 Regulations on clinical trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Remicade Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Remicade Market Segmentation
8.1 By Type
8.1.1 Branded Remicade (Infliximab)
8.1.2 Biosimilar Infliximab
8.1.3 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Specialty Clinics
8.2.3 Homecare Settings
8.2.4 Others
8.3 By Indication
8.3.1 Rheumatoid Arthritis
8.3.2 Crohn's Disease
8.3.3 Ulcerative Colitis
8.3.4 Ankylosing Spondylitis
8.3.5 Psoriasis
8.3.6 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Pharmacies
8.4.4 Hospital Pharmacies
8.4.5 Others
8.5 By Region
8.5.1 GCC Countries (Saudi Arabia, UAE, Kuwait, Qatar, Bahrain, Oman)
8.5.2 Levant Region (Jordan, Lebanon, Syria, Iraq, Palestine)
8.5.3 North Africa (Egypt, Morocco, Algeria, Tunisia)
8.5.4 Others
8.6 By Patient Demographics
8.6.1 Age Group (Pediatric, Adult, Geriatric)
8.6.2 Gender
8.6.3 Others
8.7 By Treatment Setting
8.7.1 Inpatient
8.7.2 Outpatient
8.7.3 Others
9. Middle East Remicade Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Middle East Remicade/Biosimilar Segment)
9.2.4 Market Penetration Rate (Share of Remicade/Biosimilar in Middle East Biologics Market)
9.2.5 Regulatory Approval Success Rate (MOH, SFDA, etc.)
9.2.6 Pricing Strategy (Branded vs. Biosimilar)
9.2.7 Product Portfolio Breadth (Number of Indications/Variants)
9.2.8 Distribution Network Coverage (Number of Countries/Channels)
9.2.9 Sales per Medical Representative (Middle East Region)
9.2.10 Customer Satisfaction Score (Healthcare Provider/Patient Feedback)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Johnson & Johnson (Janssen Biotech)
9.5.2 Merck & Co., Inc. (MSD)
9.5.3 AbbVie Inc.
9.5.4 Amgen Inc.
9.5.5 Pfizer Inc.
9.5.6 Sandoz International GmbH (Novartis Group)
9.5.7 Viatris Inc. (formerly Mylan N.V.)
9.5.8 Teva Pharmaceutical Industries Ltd.
9.5.9 Biogen Inc.
9.5.10 UCB S.A.
9.5.11 Fresenius Kabi AG
9.5.12 Hikma Pharmaceuticals PLC
9.5.13 EMD Serono (Merck KGaA)
9.5.14 Celltrion Healthcare Co., Ltd.
9.5.15 Samsung Bioepis Co., Ltd.
9.5.16 Julphar (Gulf Pharmaceutical Industries)
9.5.17 Tabuk Pharmaceuticals Manufacturing Co.
9.5.18 SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation)
9.5.19 Pharco Pharmaceuticals
9.5.20 Eva Pharma
10. Middle East Remicade Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Biologics
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Infrastructure
10.2.2 Spending on Biologics
10.2.3 Budgeting for Clinical Trials
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Treatment
10.3.2 Affordability Issues
10.3.3 Availability of Specialists
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Treatment Options
10.4.2 Training Needs for Healthcare Providers
10.4.3 Patient Education Requirements
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Treatment Outcomes
10.5.2 Cost-Benefit Analysis
10.5.3 Expansion into New Indications
10.5.4 Others
11. Middle East Remicade Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare organizations and pharmaceutical associations in the Middle East
- Review of published articles and white papers on Remicade usage and market trends
- Examination of regulatory frameworks and approval processes from local health authorities
Primary Research
- Interviews with oncologists and rheumatologists to understand prescribing patterns
- Surveys with hospital procurement managers regarding Remicade purchasing decisions
- Field interviews with pharmacists to gather insights on patient access and medication adherence
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and clinical studies
- Triangulation of insights from healthcare professionals, market analysts, and patient advocacy groups
- Sanity checks through expert panel reviews to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total market size based on national healthcare expenditure and pharmaceutical spending
- Segmentation by therapeutic area, including autoimmune diseases and oncology
- Incorporation of demographic data to assess patient population eligible for Remicade treatment
Bottom-up Modeling
- Volume estimates derived from patient treatment regimens and average dosage of Remicade
- Cost analysis based on pricing structures from hospitals and outpatient clinics
- Calculation of market size using patient volume multiplied by average treatment costs
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as disease prevalence and healthcare policy changes
- Scenario modeling based on potential market disruptions, including new entrants and biosimilars
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Treatment Centers | 100 | Oncologists, Clinical Pharmacists |
| Rheumatology Clinics | 80 | Rheumatologists, Nurse Practitioners |
| Hospital Procurement Departments | 70 | Procurement Managers, Financial Officers |
| Patient Advocacy Groups | 50 | Patient Representatives, Healthcare Advocates |
| Pharmacy Chains | 60 | Pharmacy Managers, Medication Therapists |
Frequently Asked Questions
What is the current value of the Middle East Remicade Market?
The Middle East Remicade Market is valued at approximately USD 260 million, driven by the rising prevalence of autoimmune diseases, increased healthcare expenditure, and advancements in biologic therapies, particularly in countries like Saudi Arabia and the UAE.